Abstract
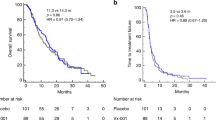
The cancer vaccine 105AD7 is an anti-idiotypic monoclonal antibody that mimics the tumour-associated antigen 791T/gp72 (CD55, Decay Accelerating Factor) on colorectal cancer cells. Phase I studies in patients with advanced disease confirmed that 105AD7 is non-toxic, and that T cell responses could be generated. A prospective, randomized, double-blind, placebo-controlled survival study in patients with advanced colorectal cancer was performed. 162 patients were enrolled between April 1994 and October 1996. Patients attended at trial entry, and at 6 and 12 weeks, where they received 105AD7 or placebo. Study groups were comparable in terms of patient demographics, and time from diagnosis of advanced colorectal cancer (277.1 v 278.6 days). Baseline disease was similar, with 50% of patients having malignancy in at least 2 anatomic sites. Compliance with treatment was poor, with only 50% of patients receiving 3 planned vaccinations. Median survival from randomization date was 124 and 184 days in 105AD7 and placebo arms respectively (P=0.38), and 456 and 486 days from the date of diagnosis of advanced disease (P=0.82). 105AD7 vaccination does not prolong survival in patients with advanced colorectal cancer. The reasons for lack of efficacy are unclear, but may reflect the high tumour burden in the patient population, and poor compliance with immunization. Further vaccine studies should concentrate on patients with minimal residual disease. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Amin S, Robins RA, Maxwell-Armstrong CA, Galvin A, Scholefield JH and Durrant LG (2000) Vaccine induced apoptosis – A novel clinical trial end point?. Cancer Res 60: 3132–3136
Austin EB, Robins RA and Durrant LG (1989) Human monoclonal anti-idiotypic antibody to the tumour-associated antibody 791T/36. Immunology 67: 525–530
Buckley TJD, Robins RA and Durrant LG (1995) Clinical evidence that the human monoclonal anti-idiotypic antibody 105AD7 delays tumour growth by stimulating anti-tumor T cell responses. Hum Antibod Hybridomas 6: 68–72
Denton GWL, Durrant LG and Hardcastle JD (1994) Clinical outcome of colorectal cancer patients treated with human monoclonal anti-idiotypic antibody. Int J Cancer 57: 10–14
Durrant LG, Buckley TJD and Denton GWL (1994) Enhanced cell-mediated killing in patients immunized with a human monoclonal anti-idiotypic antibody 105AD7. Cancer Res 54: 4837–4840
Durrant LG, Buckley DJ and Robins RA (2000a) 105AD7 cancer vaccine stimulates anti-tumour helper and cytotoxic T cell responses in colorectal cancer patients but repeated immunisations are required to maintain these responses. Int J Cancer 85: 87–92
Durrant LG, Maxwell-Armstrong CA and Buckley D (2000b) A neoadjuvant clinical trial in colorectal cancer patients of the human anti-idiotypic antibody 105AD7, that mimics CD55. Clinical Cancer Research 6: 422–430
Jacob L, Somasundaram R and Smith W (1997) Cytotoxic T cell clone against rectal carcinoma induced by stimulation of a patients peripheral blood mononuclear cells with autologous cultured tumour cells. Int J Cancer 71: 325–332
Maxwell-Armstrong CA and Durrant LG et al Robins RA et al (1999) Increased activation of lymphocytes infiltrating primary colorectal cancers following immunisation with the anti-idiotypic monoclonal antibody 105AD7. Gut 45: 593–598
Robins RA, Denton GWL and Hardcastle JD (1991) Antitumor immune response and Interleukin-2 production induced in colorectal cancer patienta by immunisation with human monoclonal anti-idiotypic antibody. Cancer Res 51: 5425–5429
Spendlove I, Li L and Carmichael J (1999) Decay Accelerating Factor (CD55): A Target for Cancer Vaccines. Cancer Res 59: 2282–2286
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Maxwell-Armstrong, C., Durrant, L., Buckley, T. et al. Randomized double-blind phase II survival study comparing immunization with the anti-idiotypic monoclonal antibody 105AD7 against placebo in advanced colorectal cancer. Br J Cancer 84, 1443–1446 (2001). https://doi.org/10.1054/bjoc.2001.1725
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.1725
Keywords
This article is cited by
-
Cancer vaccines as a therapeutic modality: the long trek
Cancer Immunology, Immunotherapy (2006)
-
Immune responses to the 105AD7 human anti-idiotypic vaccine after intensive chemotherapy, for osteosarcoma
British Journal of Cancer (2005)
-
Immune responses in advanced colorectal cancer following repeated intradermal vaccination with the anti-CEA murine monoclonal antibody, PR1A3: results of a phase I study
International Journal of Colorectal Disease (2005)
-
Decay accelerating factor and colorectal cancer
Chinese Journal of Cancer Research (2004)
-
The Inhibitory Effect of CD46, CD55, and CD59 on Complement Activation After Immunotherapeutic Treatment of Cervical Carcinoma Cells with Monoclonal Antibodies or Bispecific Monoclonal Antibodies
Laboratory Investigation (2002)