Abstract
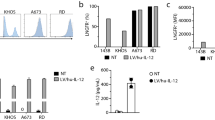
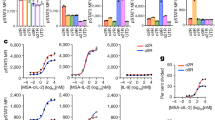
Various studies have emphasized an immunodepression state observed at the tumour site. To reverse this defect and based upon animal studies, we initiated a phase I clinical trial of gene therapy in which various doses of xenogeneic monkey fibroblasts (Vero cells) genetically engineered to produce human IL-2 were administered intratumorally in 8 patients with metastatic solid tumours. No severe adverse effect was observed in the 8 patients analysed during this clinical trial even in the highest dose (5 ¥ 107 cells) group. This absence of toxicity seems to be associated with rapid elimination of Vero-IL-2 cells from the organism. Indeed, exogenous IL-2 mRNA could no longer be detected in the peripheral whole blood 48 hours after Vero-IL-2 cell administration. In addition, we did not find any expression of exogenous IL-2 mRNA in post-therapeutic lesions removed 29 days after the start of therapy. A major finding of this trial concerns the two histological responses of two treated subcutaneous nodules not associated with an apparent clinical response. The relationship between local treatment and tumour regression was supported by replacement of tumour cells by inflammatory cells in regressing lesions and marked induction of T and natural killer cell derived cytokines (IL-2, IL-4, IFNg …) in post-therapeutic lesions analysed 28 days after the start of Vero-IL-2 administration. Gene therapy using xenogeneic cells as vehicle may therefore present certain advantages over other vectors, such as its complete absence of toxicity. Furthermore, the in vivo biological effect of immunostimulatory genes, i.e IL-2-, may be potentiated by the xenogeneic rejection reaction. © 2000 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Addison CL, Braciak T, Ralston R, Muller WJ, Gauldie J and Graham FL (1995 Intratumoral injection of an adenovirus expressing interleukin 2 induces regression and immunity in a murine breast cancer model. Proc Natl Acad Sci USA 92: 8522–8526
Aruga A, Aruga E and Chang AE (1997 Reduced efficacy of allogeneic versus syngeneic fibroblasts modified to secrete cytokines as a tumor vaccine adjuvant. Cancer Res 57: 3230–3237
Berd D, Kairys J, Dunton C, Mastrangelo MJ, Sato T and Maguire HC Jr (1998 Autologous, hapten-modified vaccine as a treatment for human cancers. Semin Oncol 25: 646–653
Bowman LC, Grossmann M, Rill D, Brown M, Zhong WY, Alexander B, Leimig T, Coustan-Smith E, Campana D, Jenkins J, Woods D and Brenner M (1998 Interleukin-2 gene-modified allogeneic tumor cells for treatment of relapsed neuroblastoma. Human Gene Ther 9: 1303–1311
Bubenik J, Voitenok NN, Kieler J, Prassolov VS, Chumakov PM, Bubenikova D, Simova J and Jandlova T (1988 Local administration of cells containing an inserted IL-2 gene and producing IL-2 inhibits growth of human tumours in nu/nu mice. Immunol Letters 19: 79–282
Chen L (1998 Immunological ignorance of silent antigens as an explanation of tumor evasion. Immunol Today 19: 27–30
Chouaib S, Asselin-Paturel C, Mami-Chouaib F, Caignard A and Blay JY (1997 The host-tumor immune conflict: from immunosuppression to resistance and destruction. Immunol Today 18: 493–497
Clary BM, Coveney EC, Philip R, Blazer DG 3rd, Morse M, Gilboa E and Lyerly HK (1997 Inhibition of established pancreatic cancers following specific active immunotherapy with interleukin-2 gene-transduced tumor cells. Cancer Gene Ther 4: 97–104
Cordier L, Duffour MT, Sabourin JC, Lee MG, Cabannes J, Ragot T and Perricaudet M Haddada (1995 Complete recovery of mice from a pre-established tumor by direct intratumoral delivery of an adenovirus vector harboring the murine IL-2 gene. Gene Ther 2: 16–21
Cressent M, Pidoux E, Cohen R, Modigliani E and Roth C (1995 Interleukin-2 and interleukin-4 display potent antitumour activity on rat medullary thyroid carcinoma cells. Eur J Cancer 31A: 2379–2384
Dorval T, Mathiot C, Chosidow O, Revuz J, Avril MF, Guillaume JC, Tursz T, Brandely M, Pouillart P and Fridman WH (1992 IL-2 phase II trial in metastatic melanoma: analysis of clinical and immunological parameters. Biotec Therap 3: 63–79
Dow W, Elmslie RE, Willson AP, Roche L, Gorman C and Potter TA (1998 In vivo tumor transfection with superantigen plus cytokine genes induces tumor regression and prolongs survival in dogs with malignant melanoma. J Clin Invest 101: 2406–2414
Dranoff G, Jaffee E, Lazenby A, Golumbek P, Levitsky H, Brose K, Jackson V, Hamada H, Pardoll D and Mulligan RC (1993 Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci USA 90: 3539–3543
Elkins KL, Ennist DL, Winegar RK and Weir JP (1994 In vivo delivery of interleukin-4 by a recombinant vaccinia virus prevents tumor development in mice. Human. Gene. Ther 5: 809–820
Fakhrai H, Shawler DL, Gjerset R, Naviaux RK, Koziol J, Royston I and Sobol RE (1995 Cytokine gene therapy with interleukin-2-transduced fibroblasts: effects of IL-2 dose on anti-tumor immunity. Human Gene Ther 6: 591–601
Fearon ER, Itaya T, Hunt B, Vogelstein B and Frost P (1988 Induction in a murine tumor of immunogenic tumor variants by transfection with a foreign gene. Cancer Res 48: 2975–2980
Fearon ER, Pardoll DM, Itaya T, Golumbek P, Levitsky HI, Simons JW, Karasuyama H, Vogelstein B and Frost P (1990 Interleukin-2 production by tumor cells bypasses T helper function in the generation of an antitumor response. Cell 60: 397–403
Gansbacher B, Zier K, Daniels B, Cronin K, Bannerji R and Gilboa E (1990 Interleukin 2 gene transfer into tumor cells abrogates tumorigenicity and induces protective immunity. J Exp Med 172: 1217–1224
Gemlo BT, Palladino MA Jr, Jaffe HS, Espevik TP and Rayner AA (1988 Circulating cytokines in patients with metastatic cancer treated with recombinant interleukin 2 and lymphokine-activated killer cells. Cancer Res 48: 5864–5867
Ghosh P, Komschlies KL, Cippitelli M, Longo DL, Subleski J, Ye J, Sica A, Young HA, Wiltrout RH and Ochoa AC (1995 Gradual loss of T-helper 1 populations in spleen of mice during progressive tumor growth. J Natl Cancer Inst 87: 1478–1483
Glick RP, Lichtor T, Mogharbel A, Taylor CA and Cohen EP (1997 Intracerebral versus subcutaneous immunization with allogeneic fibroblasts genetically engineered to secrete interleukin-2 in the treatment of central nervous system glioma and melanoma. Neurosurgery 41: 898–906
Golumbek PT, Lazenby AJ, Levitsky HI, Jaffee LM, Karasuyama H, Baker M and Pardoll DM (1991 Treatment of established renal cancer by tumor cells engineered to secrete interleukin-4. Science 254: 713–716
Golumbek PT, Azhari R, Jaffee EM, Levitsky HI, Lazenby A, Leong K and Pardoll DM (1993 Controlled release, biodegradable cytokine depots: a new approach in cancer vaccine design. Cancer Res 53: 5841–5844
Haddada H, Ragot T, Cordier L, Duffour MT and Perricaudet M (1993 Adenoviral interleukin-2 gene transfer into P815 tumor cells abrogates tumorigenicity and induces antitumoral immunity in mice. Human. Gene Ther 4: 703–711
Heo DS, Yoon SJ, Kim WS, Lee KH, Seol JG, Lee SG, Jung CW, Cho EK, Kim CW, Park MH, Sung MW, Kim KH, Bang YJ and Kim NK (1998 Locoregional response and increased natural killer activity after intratumoral injection of HLA-B7/beta2 microglobulin gene in patients with cancer. Human Gene Ther 9: 2031–2038
Janeway CA Jr and Bottomly K (1994 Signals and signs for lymphocyte responses. Cell 76: 275–285
Keilholz U, Scheibenbogen C, Stoelben E, Saege RHD and Hunstein W (1994 Immunotherapy of metastatic melanoma with interferon-alpha and interleukin-2: pattern of progression in responders and patients with stable disease with or without resection of residual lesions. Eur J Cancer 30A: 955–958
Konrad MW, Hemstreet G, Hersh EM, Mansell PW, Mertelsmann R, Kolitz JE and Bradley EC (1990 Pharmacokinetics of recombinant interleukin 2 in humans. Cancer Res 50: 2009–2017
Koppenhagen FJ, Kupcu Z, Wallner G, Crommelin DJ, Wagner E, Storm G and Kircheis R (1998 Sustained cytokine delivery for anticancer vaccination: liposomes as alternative for gene-transfected tumor cells. Clin Cancer Res 4: 1881–1886
Leroy P, Slos P, Homann H, Erbs P, Poitevin Y, Regulier E, Colonna FQ, Devauchelle P, Roth C, Pavirani A and Mehtali M (1998 Cancer immunotherapy by direct in vivo transfer of immunomodulatory genes. Res Immunol 149: 681–684
Lindenmann J and Klein PA (1967 Viral oncolysis: increased immunogenicity of host cell antigen associated with influenza virus. J Exp Med 126: 93–108
Lotze MT, Custer MC, Sharrow SO, Rubin LA, Nelson DL and Rosenberg SA (1987 In vivo administration of purified human interleukin-2 to patients with cancer: development of interleukin-2 receptor positive cells and circulating soluble interleukin-2 receptors following interleukin-2 administration. Cancer Res 47: 2188–2195
Maass G, Schmidt W, Berger M, Schilcher F, Koszik F, Schneeberger A, Stingl G, Birnstiel ML and Schweighoffer T (1995 Priming of tumor-specific T cells in the draining lymph nodes after immunization with interleukin 2-secreting tumor cells: three consecutive stages may be required for successful tumor vaccination. Proc Natl Acad Sci USA 92: 5540–5544
Mir LM, Roth C, Orlowski S, Quintin-Colonna F, Fradelizi D, Belehradek J Jr and Kourilsky P (1995 Systemic antitumor effects of electrochemotherapy combined with histoincompatible cells secreting interleukin-2. J Immunother 17: 30–38
Mukherji B and Chakraborty NG (1995 Immunobiology and immunotherapy of melanoma. Curr Opin Oncol 7: 175–184
Pardoll DM (1995 Paracrine cytokine adjuvants in cancer immunotherapy. Annu Rev Immunol 13: 399–415
Plautz GE, Yang ZY, Wu BY, Gao X, Huang L and Nabel GJ (1993 Immunotherapy of malignancy by in vivo gene transfer into tumors. Proc Natl Acad Sci US 90: 4645–4649
Quintin-Colonna F, Devauchelle P, Fradelizi D, Mourot B, Faure F, Kourilsky P, Roth C and Mehtali M (1996 Gene therapy of spontaneous canine melanoma and feline fibrosarcoma by intratumoral administration of histoincompatible cells expressing human interleukin-2. Gene Ther 3: 1104–1112
Rochlitz C, Jantscheff P, Bongartz G, Dietrich PY, Quiquerez AL, Schatz C, Mehtali M, Courtney M, Tartour E, Dorval T, Fridman WH and Herrmann R (1999 Gene therapy study of cytokine-transfected xenogeneic cells (Vero-interleukin-2) in patients with metastatic solid tumors. Cancer Gene Ther 6: 271–281
Roth C, Mir LM, Cressent M, Quintin-Colonna F, Ley V, Fradelizi D and Kourilsky P (1992 Inhibition of tumor growth by histoincompatible cells expressing interleukin-2. Int Immunol 4: 1429–1436
Schaafsma MR, Falkenburg JH, Landegent JE, Duinkerken N, Osanto S, Ralph P, Kaushansky K, Wagemaker G, Van Damme J, Willemze R and Fibbe WE (1991 In vivo production of interleukin-5, granulocyte-macrophage colony-stimulating factor, macrophages colony-stimulating factor, and interleukin-6 during intravenous administration of high-dose interleukin-2 in cancer patients. Blood 78: 1981–1987
Schmidt-Wolf GD and Schmidt-Wolf IG (1995 Cytokines and gene therapy. Immunol Today 16: 173–175
Schuler T, Qin Z, Ibe S, Noben-Trauth N and Blankenstein T (1999 T helper cell type 1-associated and cytotoxic T lymphocyte-mediated tumor immunity is impaired in interleukin 4-deficient mice. J Exp Med 189: 803–810
Speiser DE, Miranda R, Zakarian A, Bachmann MF, McKall-Faienza K, Odermatt B, Hanahan D, Zinkernagel RM and Ohashi PS (1997 Self antigens expressed by solid tumors Do not efficiently stimulate naive or activated T cells: implications for immunotherapy. J Exp Med 186: 645–653
Stopeck AT, Hersh EM, Akporiaye ET, Harris DT, Grogan T, Unger E, Warneke J, Schluter SF and Stahl S (1997 Phase I study of direct gene transfer of an allogeneic histocompatibility antigen, HLA-B7, in patients with metastatic melanoma. J Clin Oncol 15: 341–349
Tahara H, Zeh HJ 3rd, Storkus WJ, Pappo I, Watkins SC, Gubler U, Wolf SF, Robbins PD and Lotze MT (1994 Fibroblasts genetically engineered to secrete interleukin 12 can suppress tumor growth and induce antitumor immunity to a murine melanoma in vivo. Cancer Res 54: 182–189
Tartour E and Fridman WH (1998 Cytokines and cancer. Int Rev Immunol 16: 683–704
Tartour E, Mathiot C and Fridman WH (1992 Current status of interleukin-2 therapy in cancer. Biomed Pharmacother 46: 473–484
Tartour E, Gey A, Sastre-Garau X, Lombard Surin I, Mosseri V and Fridman WH (1998 Prognostic value of intratumoral interferon gamma messenger RNA expression in invasive cervical carcinomas. J Natl Cancer Inst 90: 287–294
Tepper RI, Pattengale PK and Leder P (1989 Murine interleukin-4 displays potent anti-tumor activity in vivo. Cell 57: 503–512
Tsai SC, Gansbacher B, Tait L, Miller FR and Heppner GH (1993 Induction of antitumor immunity by interleukin-2 gene-transduced mouse mammary tumor cells versus transduced mammary stromal fibroblasts. J Natl Cancer Inst 85: 546–553
Veelken H, Mackensen A, Lahn M, Kohler G, Becker D, Franke B, Brennscheidt U, Kulmburg P, Rosenthal FM, Keller H, Hasse J, Schultze-Seemann W, Farthmann EH, Mertelsmann R and Lindemann A (1997 A phase-I clinical study of autologous tumor cells plus interleukin-2-gene-transfected allogeneic fibroblasts as a vaccine in patients with cancer. Int J Cancer 70: 269–277
Vowels BR, Lessin SR, Cassin M, Jaworsky C, Benoit B, Wolfe JT and Rook AH (1994 Th2 cytokine mRNA expression in skin in cutaneous T-cell lymphoma. J Invest Dermatol 103: 669–673
Wang Q, Stanley J, Kudoh S, Myles J, Kolenko V, Yi T, Tubbs R, Bukowski R and Finke J (1995 T cells infiltrating non-Hodgkin’s B cell lymphomas show altered tyrosine phosphorylation pattern even though T cell receptor/CD3-associated kinases are present. J Immunol 155: 1382–1392
Zatloukal K, Schneeberger A, Berger M, Schmidt W, Koszik F, Kutil R, Cotten M, Wagner E, Buschle M, Maass G, Payer E, Stingl G and Birnstiel ML (1995 Elicitation of a systemic and protective anti-melanoma immune response by an IL-2-based vaccine. Assessment of critical cellular and molecular parameters. J Immunol 154: 3406–3419
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Tartour, E., Mehtali, M., Sastre-Garau, X. et al. Phase I clinical trial with IL-2-transfected xenogeneic cells administered in subcutaneous metastatic tumours: clinical and immunological findings. Br J Cancer 83, 1454–1461 (2000). https://doi.org/10.1054/bjoc.2000.1492
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1492
Keywords
This article is cited by
-
Cell-based Delivery of Oncolytic Viruses: A New Strategic Alliance for a Biological Strike Against Cancer
Molecular Therapy (2007)
-
Dendritic cells infected with a vaccinia virus interleukin-2 vector secrete high levels of IL-2 and can become efficient antigen presenting cells that secrete high levels of the immunostimulatory cytokine IL-12
Cancer Gene Therapy (2003)
-
Immunotherapy of metastatic melanoma by intratumoral injections of Vero cells producing human IL-2: Phase II randomized study comparing two dose levels
Cancer Gene Therapy (2002)