Abstract
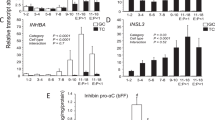
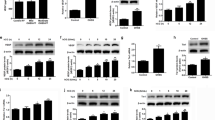
In this study, we have investigated the expression of inhibin subunits and activin receptors (ActRs) in normal and malignant ovarian cells. Each product of the inhibin subunits (α, βa, βb) and activin receptors (ActRs) amplified by reverse transcription polymerase chain reaction were detected as a single band in human granulosa cells, surface epithelial cells (OSE), and the ovarian cancer cell lines OVCAR 3 and SKOV 3. Western blot analysis was performed using polyclonal antibodies against ActR IIa or IIb peptides based on 13 COOH-terminal amino acids; cultured human granulosa cells were used as a positive control. Using ActR IIa antibody, one major band corresponding to approximately 80 kDa and one minor band corresponding to 105 kDa were observed in the samples. One single band at approximately 60 kDa was detected in OVCAR 3 and a 50 kDa band was detected with ActR IIb antibody in cultured granulosa cell, OSE and SKOV 3. Although no detectable change was induced in Smad 4 mRNA in OVCAR 3, Smad 2 mRNA levels were increased during 48 h treatment with activin A (50 ng ml−1). These data provide a better understanding as the first step in the mechanism of action of the activin in the epithelial ovarian carcinoma. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Attisano L and Wrana JL (1996) Signal transduction by members of the transforming growth factor-beta superfamily. Cytokine Growth Factor Rev 7: 327–339
Attisano L, Carcamo J, Ventura F, Weis FM, Massague J and Wrana JL (1993) Identification of human activin and TGF beta type I receptors that form heteromeric kinase complexes with type II receptors. Cell 75: 671–680
Berchuck A, Rodriguez G, Olt G, Whitaker R, Boente MP, Arrick BA, Clarke Pearson DL and Bast RC Jr (1992) Regulation of growth of normal ovarian epithelial cells and ovarian cancer cell lines by transforming growth factor-beta. Am J Obstet Gynecol 166: 676–684
Bernstein JR, Crowley WF Jr and Schneyer AL (1990) An improved method of purifying inhibin radioligand for radioimmunoassay. Biol Reprod 43: 492–496
Chen X, Rubock MJ and Whitman M (1996) A transcriptional partner for MAD proteins in TGF-beta signalling. Nature 383: 691–696
Chomczynski P and Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159
Derynck R and Feng XH (1997) TGF-beta receptor signaling. Biochim Biophys Acta 1333: F105–150
Di Simone N, Crowley WF Jr, Wang QF, Sluss PM and Schneyer AL (1996) Characterization of inhibin/activin subunit, follistatin, and activin type II receptors in human ovarian cancer cell lines: a potential role in autocrine growth regulation. Endocrinology 137: 486–494
Eramaa M, Hilden K, Tuuri T and Ritvos O (1995) Regulation of inhibin/activin subunit messenger ribonucleic acids (mRNAs) by activin A and expression of activin receptor mRNAs in cultured human granulosa-luteal cells. Endocrinology 136: 4382–4389
Fukuda J, Ito I, Tanaka T and Leung PC (1998) Cell survival effect of activin against heat shock stress on OVCAR3. Life Sci 63: 2209–2220
Fynan TM and Reiss M (1993) Resistance to inhibition of cell growth by transforming growth factor-beta and its role in oncogenesis. Crit Rev Oncog 4: 493–540
Hahn SA, Schutte M, Hoque AT, Moskaluk CA, da Costa LT, Rozenblum E, Weinstein CL, Fischer A, Yeo CJ, Hruban RH and Kern SE (1996) DPC4, a candidate tumor suppressor gene at human chromosome 18q21.1. Science 271: 350–353
Hamilton TC, Young RC, McKoy WM, Grotzinger KR, Green JA, Chu EW, Whang Peng J, Rogan AM, Green WR and Ozols RF (1983) Characterization of a human ovarian carcinoma cell line (NIH: OVCAR-3) with androgen and estrogen receptors. Cancer Res 43: 5379–5389
Heldin CH, Miyazono K and ten Dijke P (1997) TGF-β signalling from cell membrane to nucleus through SMAD proteins. Nature 390: 465–471
Hillier SG (1991) Regulatory functions for inhibin and activin in human ovaries. J Endocrinol 131: 171–175
Hino M, Tojo A, Miyazono K, Miura Y, Chiba S, Eto Y, Shibai H and Takaku F (1989) Characterization of cellular receptors for erythroid differentiation factor on murine erythroleukemia cells. J Biol Chem 264: 10309–10314
Hurteau J, Rodriguez GC, Whitaker RS, Shah S, Mills G, Bast RC and Berchuck A (1994) Transforming growth factor-beta inhibits proliferation of human ovarian cancer cells obtained from ascites. Cancer 74: 93–99
Kondo S, Hashimoto M, Etoh Y, Murata M, Shibai H and Muramatsu M (1989) Identification of the two types of specific receptor for activin/EDF expressed on Friend leukemia and embryonal carcinoma cells. Biochem Biophys Res Commun 161: 1267–1272
Kruk PA, Maines Bandiera SL and Auersperg N (1990) A simplified method to culture human ovarian surface epithelium. Lab Invest 63: 132–136
Li M, Li J, Hoodless PA, Tzukazaki T, Wrana JL, Attisano L and Tsang BK (1997) Mothers against decapentaplegic-related protein 2 expression in avian granulosa cells is up-regulated by transforming growth factor beta during ovarian follicular development. Endocrinology 138: 3659–3665
Li W, Yuen BH and Leung PC (1992) Inhibition of progestin accumulation by activin-A in human granulosa cells. J Clin Endocrinol Metab 75: 285–289
Mason AJ, Niall HD and Seeburg PH (1986) Structure of two human ovarian inhibins. Biochem Biophys Res Commun 135: 957–964
Mason AJ, Berkemeier LM, Schmelzer CH and Schwall RH (1989) Activin B: precursor sequences, genomic structure and in vitro activities. Mol Endocrinol 3: 1352–1358
Massague J (1996) TGFbeta signaling: receptors, transducers, and Mad proteins. Cell 85: 947–950
Mathews LS (1994) Activin receptors and cellular signaling by the receptor serine kinase family. Endocr Rev 15: 310–325
Matzuk MM, Finegold MJ, Su JG, Hsueh AJ and Bradley A (1992) Alpha-inhibin is a tumour-suppressor gene with gonadal specificity in mice. Nature 360: 313–319
Roberts AB, Anzano MA, Wakefield LM, Roche NS, Stern DF and Sporn MB (1985) Type beta transforming growth factor: a bifunctional regulator of cellular growth. Proc Natl Acad Sci USA 82: 119–123
Robertson DM, Foulds LM, Leversha L, Morgan FJ, Hearn MT, Burger HG, Wettenhall RE and de Kretser DM (1985) Isolation of inhibin from bovine follicular fluid. Biochem Biophys Res Commun 126: 220–226
Siemens CH and Auersperg N (1988) Serial propagation of human ovarian surface epithelium in tissue culture. J Cell Physiol 134: 347–356
Tanimoto K, Handa S, Ueno N, Murakami K and Fukamizu A (1991) Structure and sequence analysis of the human activin beta A subunit gene. DNA Seq 2: 103–110
ten Dijke P, Yamashita H, Ichijo H, Franzen P, Laiho M, Miyazono K and Heldin CH (1994) Characterization of type I receptors for transforming growth factor-beta and activin. Science 264: 101–104
Vale W, Rivier J, Vaughan J, McClintock R, Corrigan A, Woo W, Karr D and Spiess J (1986) Purification and characterization of an FSH releasing protein from porcine ovarian follicular fluid. Nature 321: 776–779
Vale W, Rivier C, Hsueh A, Campen C, Meunier H, Bicsak T, Vaughan J, Corrigan A, Bardin W, Sawchenko P, Petraglia F, Yu J, Plotsky P, Spiess J and Rivier J (1988) Chemical and biological characterization of the inhibin family of protein hormones. Recent Prog Horm Res 44: 1–34
Welt CK, Lambert Messerlian G, Zheng W, Crowley WF Jr and Schneyer AL (1997) Presence of activin, inhibin, and follistatin in epithelial ovarian carcinoma. J Clin Endocrinol Metab 82: 3720–3727
Willis SA, Zimmerman CM, Li LI and Mathews LS (1996) Formation and activation by phosphorylation of activin receptor complexes. Mol Endocrinol 10: 367–379
Xu J, Matsuzaki K, McKeehan K, Wang F, Kan M and McKeehan WL (1994) Genomic structure and cloned cDNAs predict that four variants in the kinase domain of serine/threonine kinase receptors arise by alternative splicing and poly(A) addition. Proc Natl Acad Sci USA 91: 7957–7961
Ying SY (1988) Inhibins, activins, and follistatins: gonadal proteins modulating the secretion of follicle-stimulating hormone. Endocr Rev 9: 267–293
Zhang Y, Feng X, We R and Derynck R (1996) Receptor-associated Mad homologues synergize as effectors of the TGF-beta response. Nature 383: 168–172
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Ito, I., Minegishi, T., Fukuda, J. et al. Presence of activin signal transduction in normal ovarian cells and epithelial ovarian carcinoma. Br J Cancer 82, 1415–1420 (2000). https://doi.org/10.1054/bjoc.1999.1127
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.1999.1127