Abstract
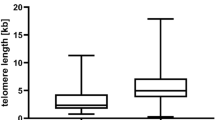
Telomere length maintenance, in the vast majority of cases executed by telomerase, is a prerequisite for long-term proliferation. Most malignant tumours, including lymphomas, are telomerase-positive and this activity is a potential target for future therapeutic interventions since inhibition of telomerase has been shown to result in telomere shortening and cell death in vitro. One prerequisite for the suitability of anti-telomerase drugs in treating cancer is that tumours exhibit shortened telomeres compared to telomerase-positive stem cells. A scenario is envisioned where the tumour burden is reduced using conventional therapy whereafter remaining tumour cells are treated with telomerase inhibitors. In evaluating the realism of such an approach it is essential to know the effects on telomere status by traditional therapeutic regimens. We have studied the telomere lengths in 47 diagnostic lymphomas and a significant telomere shortening was observed compared to benign lymphoid tissues. In addition, telomere length and telomerase activity were studied in consecutive samples from patients with relapsing non-Hodgkin's lymphomas. Shortened, unchanged and elongated telomere lengths were observed in the relapse samples. The telomere length alterations found in the relapsing lymphomas appeared to be independent of telomerase and rather represented clonal selection random at the telomere length level. These data indicate that anti-telomerase therapy would be suitable in only a fraction of malignant lymphomas. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Allsopp RC, Chang E, Kashefi-Aazam M, Rogaev EI, Piatyszek MA, Shay JW and Harley CB (1995) Telomere shortening is associated with cell division in vitro and in vivo. Exp Cell Res 220: 194–200
Bodnar AG, Ouellette M, Frolkis M, Holt SE, Chiu C-P, Morin GB, Harley CB, Shay JW, Lichtsteiner S and Wright WE (1998) Extension of life-span by introduction of telomerase into normal human cells. Science 279: 349–352
Bryan TM and Reddel RR (1997) Telomere dynamics and telomerase activity in in vitro immortalised human cells. Eur J Cancer 33: 767–773
Bryan TM, Englezou A, Dalla-Pozza L, Dunham MA and Reddel RR (1997) Evidence for an alternative mechanism for maintaining telomere length in human tumours and tumour-derived cell lines. Nat Med 3: 1271–1274
Feng J, Funk WD, Wang S-S, Weinrich SL, Avilion AA, Chiu C-P, Adams RR, Chang E, Allsopp RC, Yu J, Le S, West MD, Harley CB, Andrews WH, Greide CW and Villeponteau B (1995) The RNA component of human telomerase. Science 269: 1236–1241
Gravel S, Larriveé M, Labrecque P and Wellinger RJ (1998) Yeast Ku as a regulator of chromosomal DNA end structure. Science 280: 741–744
Harris NL, Jaffe ES, Stein H, Banks PM, Chan JKC, Cleary ML, Delsol G, De Wolf-Peeters C, Falini B, Gatter KC, Grogan TM, Isaacson PG, Knowles DM, Mason DY, Muller-Hermelink H-K, Pileri SA, Piris MA, Ralfkiaer E and Warnke RA (1994) A revised European–American classification of lymphoid neoplasms: a proposal from the international lymphoma study group. Blood 84: 1361–1392
Hiyama E, Yokoyhama T, Hiyama K, Yamakido M, Santo T, Kodama T, Ichikawa T and Matsuura Y (1995 a) Alteration of telomeric repeat length in adult and childhood solid neoplasias. Int J Oncol 6: 13–16
Hiyama K, Hirai Y, Kyoizumi S, Akiyama M, Hiyama E, Piatyszek MA, Shay JW, Ishioka S and Yamakido M (1995 b) Activation of telomerase in human lymphocytes and hematopoietic progenitor cells. J Immunol 155: 3711–3715
Holt SE, Norton JC, Wright WE and Shay JW (1996) Comparison of the telomeric repeat amplification protocol (TRAP) to the new TRAP-eze telomerase detection kit. Methods Cell Sci 18: 237–248
Härle-Bachor C and Boukamp P (1996) Telomerase activity in the regenerative basal layer of the epidermis in human skin and in immortal and carcinoma-derived skin keratinocytes. Proc Natl Acad Sci USA 93: 6476–6481
Jean-Louis M and Claude H (1998) G-quadruplex DNA: a target for drug design. Nat Med 4: 1366–1367
Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PLC, Coviello GM, Wright WE, Weinrich SL and Shay JW (1994) Specific association of human telomerase activity with immortal cells and cancer. Science 266: 2011–2015
LaBranche H, Dupuis S, Ben-David Y, Bani M-R, Wellinger RJ and Chabot B (1998) Telomere elongation by hnRNP A1 and a derivative that interacts with telomeric repeats and telomerase. Nat Gen 19: 199–202
Levy MZ, Allsopp RC, Futcher AB, Greider CW and Harley CB (1992) Telomere end-replication problem and cell aging. J Mol Biol 225: 951–960
Mehle C, Mieczyslaw AP, Ljungberg B, Shay JW and Roos G (1996) Telomerase activity in human renal cell carcinoma. Oncogene 13: 161–166
Meyerson M, Counter CM, Eaton EN, Ellisen LW, Steiner P, Caddle SD, Ziaugra L, Beijersbergen RL, Davidoff MJ, Liu Q, Bacchetti S, Haber DA and Weinberg RA (1997) hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumour cells and during immortalization. Cell 90: 785–795
Morales CP, Holt SE, Ouellette M, Kaur KJ, Yan Y, Wilson KS, White MA, Wright WE and Shay JW (1999) Absence of cancer-associated changes in human fibroblasts immortalized with telomerase. Nat Genet 21: 115–118
Morin GB (1989) The human telomere terminal transferase enzyme is a ribonucleoprotein that synthesizes TTAGGG repeats. Cell 59: 521–529
Norrback K-F and Roos G (1997) Telomeres and telomerase in normal and malignant haematopoietic cells. Eur J Cancer 33: 774–780
Norrback K-F, Dahlenborg K, Carlsson R and Roos G (1996) Telomerase activation in normal B lymphocytes and non-Hodgkin's lymphomas. Blood 88: 222–229
Norrback K-F, Enblad G, Erlanson M, Sundström C and Roos G (1998) Telomerase activity in Hodgkin's disease. Blood 92: 567–573
Rosenquist R (1998) The immunoglobulin heavy chain gene as a clonal marker in lymphoma and leukemia. Umeå University medical dissertations, new series No 558-ISSN 0346–6612
Slagboom PE, Droog S and Boomsma DI (1994) Genetic determination of telomere size: a twin study of three age groups. Am J Hum Genet 55: 876–882
Strahl C and Blackburn EH (1996) Effects of reverse transcriptase inhibitors on telomere length and telomerase activity in two immortalized human cell lines. Mol Cell Biol 16: 53–65
van Steensel B and de Lange T (1997) Control of telomere length by the human telomeric protein TRF 1. Nature 385: 740–743
Vaziri H, Schächter F, Uchida I, Wei L, Zhu X, Effros R, Cohen D and Harley CB (1993) Loss of telomeric DNA during aging of normal and trisomy 21 human lymphocytes. Am J Hum Genet 52: 661–667
Vaziri H, Dragowska W, Allsopp RC, Thomas TE, Harley CB and Lansdorp PM (1994) Evidence for a mitotic clock in human hematopoietic stem cells: loss of telomeric DNA with age. Proc Natl Acad Sci USA 91: 9857–9860
Weng N-P, Levine BL, June CH and Hodes RJ (1995) Human naive and memory T lymphocytes differ in telomeric length and replicative potential. Proc Natl Acad Sci USA 92: 11091–11094
Weng N-P, Granger L and Hodes RJ (1997) Telomere lengthening and telomerase activation during human B cell differentiation. Proc Natl Acad Sci USA 94(20): 10827–10832
Wright WE, Shay JW and Piatyszek MA (1995) Modifications of a telomeric repeat amplification protocol (TRAP) result in increased reliability, linearity and sensitivity. Nucleic Acids Res 23: 3794–3795
Wynford-Thomas D (1997) Proliferative lifespan checkpoints: cell-type specificity and influence on tumour biology. Eur J Cancer 33: 716–726
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Remes, K., Norrback, KF., Rosenquist, R. et al. Telomere length and telomerase activity in malignant lymphomas at diagnosis and relapse. Br J Cancer 82, 601–607 (2000). https://doi.org/10.1054/bjoc.1999.0970
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.1999.0970
Keywords
This article is cited by
-
Telomere length identifies two different prognostic subgroups among VH-unmutated B-cell chronic lymphocytic leukemia patients
Leukemia (2007)
-
Telomere size and telomerase activity in Epstein-Barr virus (EBV)-positive and EBV-negative Burkitt’s lymphoma cell lines
Archives of Virology (2005)
-
Telomere dynamics in childhood leukemia and solid tumors: a follow-up study
Leukemia (2003)
-
Telomerase activity in B-cell non-Hodgkin lymphomas is regulated by hTERT transcription and correlated with telomere-binding protein expression but uncoupled from proliferation
British Journal of Cancer (2003)
-
Telomere length shortening in non-Hodgkin's lymphoma patients undergoing chemotherapy
Annals of Hematology (2003)