Abstract
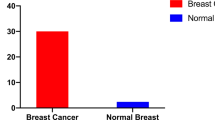
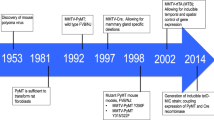
Incidence of human breast cancer (HBC) varies geographically, but to date no environmental factor has explained this variation. Previously, we reported a 44% reduction in the incidence of breast cancer in women fully immunosuppressed following organ transplantation (Stewart et al (1995) Lancet 346: 796–798). In mice infected with the mouse mammary tumour virus (MMTV), immunosuppression also reduces the incidence of mammary tumours. DNA with 95% identity to MMTV is detected in 40% of human breast tumours (Wang et al (1995) Cancer Res 55: 5173–5179). These findings led us to ask whether the incidence of HBC could be correlated with the natural ranges of different species of wild mice. We found that the highest incidence of HBC worldwide occurs in lands where Mus domesticus is the resident native or introduced species of house mouse. Given the similar responses of humans and mice to immunosuppression, the near identity between human and mouse MTV DNA sequences, and the close association between HBC incidence and mouse ranges, we propose that humans acquire MMTV from mice. This zoonotic theory for a mouse-viral cause of HBC allows testable predictions and has potential importance in prevention. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Auffray J-C, Tchernov E and Nevo E (1988) Origine du commensalisme de la souris domestique (Mus musculus domesticus) vis-à-vis de l’homme. Comptes Rendus Acad Sci 307: 517–522
Ayotte P, Dewailly E, Ryan JJ, Bruneau S and Lebel G (1997) PCBs and dioxin-like compounds in plasma of adult Inuit living in Nunavik (Arctic Quebec). Chemosphere 34: 1459–1468
Blank RD, Campbell GR and D’Eustachio P (1986) Possible derivation of the laboratory mouse genome from multiple wild Mus species. Genetics 114: 1257–1269
Boursot P, Auffray JC, Britton-Davidian J and Bonhomme F (1993) The evolution of house mice. Annu Rev Ecol Syst 24: 119–152
Brandt-Carlson C, Butel JS and Wheeler D (1993) Phylogenetic and structural analyses of MMTV LTR ORF sequences of exogenous and endogenous origins. Virology 193: 171–185
Bui TD, Rankin J, Smith K, Huguet EL, Ruben S, Strachan T, Harris AL and Lindsay S (1997) A novel human Wnt gene, WNT10B, maps to 12q13 and is expressed in human breast carcinomas. Oncogene 14: 1249–1253
Callahan R (1996) MMTV-induced mutations in mouse mammary tumors: their potential relevance to human breast cancer. Breast Cancer Res Treat 39: 33–44
Callahan R, Drohan W, Gallahan D, D’Hoostelaere L and Potter M (1982) Novel class of mouse mammary tumor virus-related DNA sequences found in all species of Mus, including mice lacking the virus proviral genome. Proc Natl Acad Sci USA 79: 4113–4117
Callahan R, Gallahan D, D’Hoostelaere LA and Potter M (1986) Endogenous MMTV proviral genomes in feral. Mus musculus domesticus. Curr Top Microbiol Immunol 127: 362–370
Chie WC, Chen CF, Chen CJ, Chang CL, Liaw YP and Lin RS (1995) Geographic variation of breast cancer in Taiwan: international and migrant comparison. Anticancer Res 15: 2745–2749
Cohen JC and Varmus HE (1979) Endogenous mammary tumour virus DNA varies among wild mice and segregates during inbreeding. Nature 278: 418–423
Day NK, Witkin SS, Sarkar NH, Kinne D, Jussawalla DJ, Levin A, Hsia CC, Geller N and Good RA (1981) Antibodies reactive with murine mammary tumor virus in sera of patients with breast cancer: geographic and family studies. Proc Natl Acad Sci USA 78: 2483–2487
Dion AS, Girardi AJ, Williams CC and Pomenti AA (1986) Serologic responses to murine mammary tumor virus (MuMTV) in MuMTV-exposed laboratory personnel. J Natl Cancer Inst 76: 611–619
Dmochowski L, Seman G and Gallager HS (1969) Viruses as possible etiologic factors in human breast cancer. Cancer 24: 1241–1249
Doll R (1969) The geographical distribution of cancer. Br J Cancer 23: 1–8
Doolittle RF, Feng DF, Johnson MS and McClure MA (1989) Origins and evolutionary relationships of retroviruses. Q Rev Biol 64: 1–30
Forsyth A (1985) Mammals of the Canadian Wild. Camden House Publishing: London
Fukui K, Noma T, Takeuchi K, Kobayashi N, Hatanaka M and Honjo T (1983) Origin of adult T cell leukemia virus: implication for its zoonosis. Mol Biol Med 1: 447–456
Gecan JS, Thrasher JL, Eisenberg W and Brickey PM (1980) Rodent excreta contamination and insect damage of wheat. J Food Protection 43: 203–204
Golovkina TV, Jaffe AB and Ross SR (1994) Coexpression of exogenous and endogenous mouse mammary tumor virus RNA in vivo results in viral recombination and broadens the virus host range. J Virol 68: 5019–5026
Haga S, Imai S, Okumoto M, Mori N, Nagano K, Nishino T, Yamamoto H, Nishio M, Sasaki IM, Enami J and Sarkar NH (1994) Detection of mouse mammary tumor virus related sequence in spontaneous human mammary tumor tissue. Int J Oncol 5: 368
Holder WD Jr, Peer GW, Bolognesi DP and Wells SA Jr (1976) Antibody reacting with mouse mammary tumor virus in serum of breast carcinoma patients. Surg Forum 27: 102–104
Holmes MD, Hunter DJ, Colditz GA, Stampfer MJ, Hankinson SE, Speizer FE, Rosner B and Willett WC (1999) Association of dietary intake of fat and fatty acids with risk of breast cancer. JAMA 281: 914–920
Howard DK and Schlom J (1980) Isolation of a series of novel variants of murine mammary tumor viruses with broadened host ranges. Int J Cancer 25: 647–654
Huguet EL, McMahon JA, McMahon AP, Bicknell R and Harris AL (1994) Differential expression of human Wnt genes 2, 3, 4, and 7B in human breast cell lines and normal and disease states of human breast tissue. Cancer Res 54: 2615–2621
Hunter DJ, Spiegelman D, Adami HO, Beeson L, van den Brandt PA, Folsom AR, Fraser GE, Goldbohm RA, Graham S and Howe GR (1996) Cohort studies of fat intake and the risk of breast cancer – a pooled analysis. N Engl J Med 334: 356–361
Hunter DJ, Hankinson SE, Laden F, Colditz GA, Manson JE, Willett WC, Speizer FE and Wolff MS (1997) Plasma organochlorine levels and the risk of breast cancer. N Engl J Med 337: 1253–1258
Imai S (1996) Mouse mammary tumor virus and mammary tumorigenesis in wild mice. Pathol Int 46: 919–932
Imai S, Okumoto M, Iwai M, Haga S, Mori N, Miyashita N, Moriwaki K, Hilgers J and Sarkar NH (1994) Distribution of mouse mammary tumor virus in Asian wild mice. J Virol 68: 3437–3442
Iscovich J and Howe GR (1998) Cancer incidence patterns (1972–91) among migrants from the Soviet Union to Israel. Cancer Causes Control 9: 29–36
Jouvin-Marche E, Cazenave PA, Voegtle D and Marche PN (1992) V β 17 T-cell deletion by endogenous mammary tumor virus in wild-type-derived mouse strain. Proc Natl Acad Sci USA 89: 3232–3235
Kozak C, Peters G, Pauley R, Morris V, Michalides R, Dudley J, Green M, Davisson M, Prakash O and Vaidya A (1987) A standardized nomenclature for endogenous mouse mammary tumor viruses. J Virol 61: 1651–1654
Labrecque N, McGrath H, Subramanyam M, Huber BT and Sekaly RP (1993) Human T cells respond to mouse mammary tumor virus-encoded superantigen: V β restriction and conserved evolutionary features. J Exp Med 177: 1735–1743
Lasfargues EY, Lasfargues JC, Dion AS, Greene AE and Moore DH (1976 a) Experimental infection of a cat kidney cell line with the mouse mammary tumor virus. Cancer Res 36: 67–72
Lasfargues EY, Vaidya AB, Lasfargues JC and Moore DH (1976 b) In vitro susceptibility of mink lung cells to the mouse mammary tumor virus. J Natl Cancer Inst 57: 447–449
Lasfargues EY, Coutinho WG and Dion AS (1979) A human breast tumor cell line (BT-474) that supports mouse mammary tumor virus replication. In Vitro 15: 723–729
Liu B, Wang Y, Holland JF and Pogo BGT (1999) Sequences homologous to mouse mammary tumor virus in human breast cancer. Proc Am Assoc Cancer Res 40: 444 (Abstract 2934)
Lopez DM, Parks WP, Silverman MA and Distasio JA (1981) Lymphoproliferative responses to mouse mammary tumor virus in lymphocyte subsets of breast cancer patients. J Natl Cancer Inst 67: 353–358
Lushnikova AA, Kriukova IN, Malivanova TF, Makhov PB and Polevaia EB (1998) Correlation between expression of antigen immunologically related to gp52 MMTV and transcription of homologous ENV MMTV DNA sequences in peripheral blood lymphocytes from breast cancer patients [in Russian]. Mol Gen Mikrobiol Virusol 3: 33–36
Miller AB and Gaudette LA (1996) Breast cancer in Circumpolar Inuit 1969–1988. Acta Oncol 35: 577–580
Mills JN and Childs JE (1998) Ecologic studies of rodent reservoirs: their relevance for human health. Emerg Infect Dis 4: 529–537
Nishio M, Xu L, Sasaki M, Haga S, Okumoto M, Mori N, Sarkar NH, Acha-Orbea H, Enami J and Imai S (1994) Complete nucleotide sequence of mouse mammary tumor virus from JYG Chinese wild mice: absence of bacterial insertion sequences in the cloned viral gag gene. Breast Cancer 1: 89–94
Nusse R (1991) Insertional mutagenesis in mouse mammary tumorigenesis. Curr Top Microbiol Immunol 171: 43–65
Parkin DM, Whelan SL, Raymond L and Young J (1997). Cancer Incidence in Five Continents. Vol. VII. IARC Scientific Publications: Lyon
Pogossiantz H (1956) Some data on the experimental studies of the nature of mammary cancer in mice carried out in the Soviet Union. Acta Unio internationalis contra Cancrum 12: 690–700
Poon MC, Tomana M and Niedermeier W (1983) Serum antibodies against mouse mammary tumor-virus-associated antigen detected nine months before appearance of a breast carcinoma. Ann Intern Med 98: 937–938
Prager EM, Sage RD, Gyllensten U, Thomas WK, Hübner R, Jones CS, Noble L, Searle JB and Wilson AC (1993) Mitochondrial DNA sequence diversity and the colonization of Scandinavia by house mice from East Holstein. Biol J Linn Soc 50: 85–122
Ringold GM, Cardiff RD, Varmus HE and Yamamoto KR (1977) Infection of cultured rat hepatoma cells by mouse mammary tumor virus. Cell 10: 11–18
Rongey RW, Hlavackova A, Lara S, Estes J and Gardner MB (1973) Types B and C RNA virus in breast tissue and milk of wild mice. J Natl Cancer Inst 50: 1581–1589
Sage RD, Atchley WR and Capanna E (1993) House mice as models in systematic biology. Syst Biol 42: 523–561
Stewart T, Tsai SC, Grayson H, Henderson R and Opelz G (1995) Incidence of de-novo breast cancer in women chronically immunosuppressed after organ transplantation. Lancet 346: 796–798
Stewart THM and Heppner GH (1997) Immunological enhancement of breast cancer. Parasitology 115: S141–153
Szabo CI and King MC (1997) Population genetics of BRCA1 and BRCA2. Am J Hum Genet 60: 1013–1020
Tajima K and Cartier L (1995) Epidemiological features of HTLV-I and adult T cell leukemia. Intervirology 38: 238–246
Titus-Ernstoff L, Egan KM, Newcomb PA, Baron JA, Stampfer M, Greenberg ER, Cole BF, Ding J, Willett W and Trichopoulos D (1998) Exposure to breast milk in infancy and adult breast cancer risk. J Natl Cancer Inst 90: 921–924
Traina-Dorge V and Cohen JC (1983) Molecular genetics of mouse mammary tumor virus. Curr Top Microbiol Immunol 106: 35–56
Wang Y, Holland JF, Bleiweiss IJ, Melana S, Liu X, Pelisson I, Cantarella A, Stellrecht K, Mani S and Pogo BG (1995) Detection of mammary tumor virus env gene-like sequences in human breast cancer. Cancer Res 55: 5173–5179
Wei WZ, Gill RF and Wang H (1993) Mouse mammary tumor virus associated antigens and super antigens – immuno-molecular correlates of neoplastic progression. Semin Cancer Biol 4: 205–213
Winter H, Cheng KK, Cummins C, Maric R, Silcocks P and Varghese C (1999) Cancer incidence in the south Asian population of England (1990–1992). Br J Cancer 79: 645–654
Wiseman CL, Bowen JM, Davis JW, Hersh EM, Brown BW and Blumenschein GR (1980) Human lymphocyte blastogenesis responses to mouse mammary tumor virus. J Natl Cancer Inst 64: 425–430
Ziegler RG, Hoover RN, Pike MC, Hildesheim A, Nomura AM, West DW, Wu-Williams AH, Kolonel LN, Horn-Ross PL and Rosenthal JF (1993) Migration patterns and breast cancer risk in Asian-American women. J Natl Cancer Inst 85: 1819–1827
Zotter S (1983) Widespread occurrence in mammals of antibodies reactive to intracytoplasmic A particles of the mouse mammary tumor virus. Arch Geschwulstforsch 53: 315–319
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Stewart, T., Sage, R., Stewart, A. et al. Breast cancer incidence highest in the range of one species of house mouse, Mus domesticus . Br J Cancer 82, 446–451 (2000). https://doi.org/10.1054/bjoc.1999.0941
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.1999.0941
Keywords
This article is cited by
-
Prevalence of MMTV-like sequences in breast cancer samples in Romanian patients-there is a geographic difference compared to the Western world
Infectious Agents and Cancer (2023)
-
Exploring the link between viruses and cancer in companion animals: a comprehensive and comparative analysis
Infectious Agents and Cancer (2023)
-
Prevalence of Human Papillomavirus and Mouse Mammary Tumor Virus Like DNAs in Tumors from Moroccan Breast Cancer Patients
Indian Journal of Gynecologic Oncology (2022)
-
Prevalence and characteristics of mouse mammary tumor virus-like virus associated breast cancer in China
Infectious Agents and Cancer (2021)
-
Evidence for a causal role by mouse mammary tumour-like virus in human breast cancer
npj Breast Cancer (2019)