Abstract
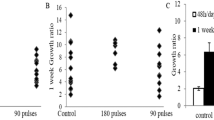
The Auger electron emitting agent 5-[125I]iodo-2′-deoxyuridine (i.e.[125I]IUdR) holds promise for the treatment of residual glioma after surgery because this thymidine analogue kills only proliferating cells. However, malignant cells which are not synthesizing DNA during exposure to the radiopharmaceutical will be spared. To determine whether tumour incorporation of [125I]IUdR could be enhanced by protracted administration, we used a C6 cell line, growing in the brains of Wistar rats, as a glioma model and compared three methods of intracerebral delivery of [125I]IUdR. Twenty-four hours after administration of drug, autoradiography of brain sections demonstrated nuclear uptake of the radiopharmaceutical in cells throughout tumour while normal brain cells remained free of radioactivity. The [125I]IUdR labelling indices (% ± s.e.m.) achieved were 6.2 (0.4) by single injection, 22.5 (4.1) using a sustained release polymer implant (poly(lactide-co-glycolide)) and 34.3 (2.0) by mini-osmotic pump. These results emphasize the need for a sustained delivery system as a prerequisite for effective treatment. These findings are also encouraging for the development of a sustained release system for radiolabelled IUdR for use in the treatment of intracranial tumours, particularly in the immediate postoperative setting. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Baranowska-Kortylewicz, J, Makrigiorgos GM, Van Den Abbeele AD, Berman RM, Adelstein SJ and Kassis AI (1991) 5[123I]iodo-2′-deoxyuridine in the radiotherapy of an early ascites tumor model. Int J Radiat Oncol Biol Phys 21: 1541–1551
Beutler AS, Banck MS, Wedekind D and Hedrich HJ (1999) Tumor gene therapy made easy: allogeneic major histocompatibility complex in the C6 rat glioma model. Human Gene Ther 10: 95–101
Brem H, Piantadosi S, Burger PC, Walker M, Selker R, Vick NA, Black K, Sisti M, Brem S, Mohr G, Muller P, Morawetz R and Schold SC (1995) Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. Lancet 345: 1008–1012
Cruickshank GS (1997) The use of SPECT in the analysis of brain tumours. SPECT Imaging of the Brain, Duncan, R (ed), pp. 161–178. Kluwer Academic: Dodrecht
Cunningham SH, Mairs RJ, Wheldon TE, Welsh PC, Vaidyanathan G and Zalutsky MR (1998) Radiotoxicity to neuroblastoma cells and spheroids of beta-, alpha- and Auger electron-emitting conjugates of benzylguanidine. Br J Cancer 77: 2061–2068
Kassis AI, Fayad F, Kinsey BM, Sastry KSR, Taube RA and Adelstein SJ (1987) Radiotoxicity of125I in mammalian cells. Radiat Res 111: 305–318
Kassis AI, Wen PW, Van den Abbeele AD, Baranowska-Kortylewicz J, Makrigiorgos GM, Metz KR, Matalka KZ, Cook CU, Sahu SK, Black PM and Adelstein SJ (1998) 5-[125I]iodo-2′-deoxyuridine in the radiotherapy of brain tumors in rats. J Nucl Med 39: 1148–1154
Kassis, AI and Adelstein SJ (1996) 5-[125I]iodo-2′-deoxyuridine in the radiotherapy of solid CNS tumors in rats. Acta Oncol 35: 935–939
Kassis AI, Tumeh SS, Wen PYC, Baranowska-Kortylewicz J, Van Den Abbeele AD, Zimmerman RE, Carvalho PA, Garada BM, Desisto WC, Bailey NO, Castronovo FP, Mariani G, Black PM and Adelstein SJ (1996) Intratumoral administration of 5[123I]iodo-2′-deoxyuridine in a patient with a brain tumor. J Nucl Med 37: 19S–22S
Klecker RW, Jr, Jenkins JF, Kinsella TJ, Fine RL, Strong JM and Collins JM (1985) Clinical pharmacology of 5-iodo-2′-deoxyuridine and 5-iodo-uracil and endogenous pyrimidine modulation. Clin Pharmacol Ther 38: 45–51
Larsen RH, Vaidyanathan G and Zalutsky MR (1997) Cytotoxicity of α-particle-emitter 5-[211At]astato-2′-deoxyuridine in human cancer cells. Int J Radiat Biol 72: 79–90
Martin RF and Haseltine WA (1981) Range of radiochemical damage to DNA with decay of iodine-125. Science 213: 896–898
Menei P, Boisdron-Celle M, Croue A, Guy G and Benoit JP (1996) Effect of stereotaxic implantation of biodegradable 5-fluorouracil-loaded microspheres in healthy and C6 glioma-bearing rats. Neurosurgery 39: 117–123
Neshasteh-Riz A, Angerson WJ, Reeves JR, Smith G, Rampling R and Mairs RJ (1997) Incorporation of iododeoxyuridine in multicellular glioma spheroids: implications for DNA-targeted radiotherapy using Auger electron emitters. Br J Cancer 75: 493–499
Neshasteh-Riz A, Mairs RJ, Angerson WJ, Stanton PD, Reeves JR, Rampling R, Owens J and Wheldon TE (1998) Differential cytotoxicity of [123I]IUdR, [125I]IUdR and [131I]IUdR to human glioma cells in monolayer or spheroid culture: effect of proliferative heterogeneity and radiation cross-fire. Br J Cancer 77: 385–390
O'Donoghue JA and Wheldon TE (1996) Targeted radiotherapy using Auger electron emitters. Phys Med Biol 41: 1973–1979
Olivi A, Ewend MG, Utsuki T, Tyler B, Domb AJ, Brat DJ and Brem H (1996) Interstitial delivery of carboplatin via biodegradable polymers is effective against experimental glioma in the rat. Cancer Chem Pharmacol 39: 90–96
Reza MS and Whateley TL (1998) Iodo-2′-deoxyuridine (IUdR) and125IUdR loaded biodegradable microspheres for controlled delivery to the brain. J Microencapsulation 15: 789–801
Reza S and Whateley TL (1999) Biodegradable thin films for sustained delivery to the brain. Drug Delivery, (in press)
Sahu SK, Wen PYC, Foulon CF, Nagel JS, Black PMCL, Adelstein SJ and Kassis AI (1997) Intrathecal 5-[125I]iodo-2′-deoxyuridine in a rat model of leptomeningeal metastases. J Nucl Med 38: 386–390
Sipos EP, Tyler B, Piantadosi S, Burger PC and Brem H (1997) Optimizing interstitial delivery of BCNU from controlled release polymers for the treatment of brain tumors. Cancer Chem Pharmacol 39: 383–389
Vaidyanathan G, Larsen RH and Zalutsky MR (1996) (5-[211At]astato-2′-deoxyuridine, an α-particle-emitting endoradiotherapeutic agent undergoing DNA incorporation. Cancer Res 56: 1204–1209
Vertosick FT, Selker RG, Grossman SJ and Joyse JM (1994) Correlation of thallium-201 single photon emission computed tomography and survival after treatment failure in patients with malignant glioma. Neurosurgery 34: 396–401
Walicka MA, Vaidyanathan G, Zalutsky MR, Adelstein SJ and Kassis AI (1998) Survival and DNA damage in chinese hamster V79 cells exposed to alpha particles emitted by DNA-incorporated astatine-211. Radiat Res 150: 263–268
Whateley TL, Rampling R, Robertson L, Crossan IM, Fallon PA, Plumb JA and Kerr DJ (1995) Biodegradable systems for sustained delivery of drugs to brain-tumors. J Cell Biochem S19A: p. 178
Williams JA, Dillehay LE, Tabassi K, Sipos E, Fahlman C and Brem H (1997) Implantable biodegradable polymers for IUdR radiosensitisation of experimental human malignant glioma. J Neurooncol 32: 181–192
Zhu J, Zhang L, Hanisch UK, Felgner PL and Reszka RA (1996) Continuous intracerebral gene delivery system for in vivo liposome-mediated gene therapy. Gene Ther 3: 472–476
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Mairs, R., Wideman, C., Angerson, W. et al. Comparison of different methods of intracerebral administration of radioiododeoxyuridine for glioma therapy using a rat model. Br J Cancer 82, 74–80 (2000). https://doi.org/10.1054/bjoc.1999.0879
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.1999.0879
Keywords
This article is cited by
-
Brachytherapy for brain tumors
Journal of Neuro-Oncology (2005)