Summary
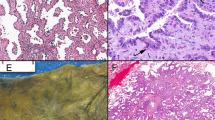
A high level of apoptotic activity and an independence of apoptosis from the expression of p53 and bcl-2 have been observed in non-small-cell lung carcinoma. We examined 44 samples of normal, metaplastic and premalignant (i.e. mild, moderate and severe dysplasias and carcinoma in situ) bronchial epithelia to evaluate whether differences in the apoptotic activity could already be seen in the stages preceding squamous cell carcinoma of the lung (SQCLC). Apoptotic cells and bodies were visualized by 3′ end labelling. The expression of p53 and members of the bcl-2 gene family, such as bcl-2, bax and mcl-1, were determined immunohistochemically with specific antibodies. The relative number of apoptotic cells and bodies [apoptotic index (AI%)] was already increased threefold as the normal bronchial epithelium changed to squamous metaplasia, and the AIs of the dysplastic lesions were about four times higher than those of the normal epithelium. Apoptosis was significantly associated with cell proliferation, as determined by proliferating cell nuclear antigen (PCNA) immunohistochemistry. However, the extent of apoptosis did not correlate with the expression of p53, bcl-2, bax and mcl-1. We conclude that, in the metaplasia–dysplasia–carcinoma sequence in the lung, the elevation of the AI% is an early event associated with cell proliferation activity, but is independent of the expression of p53, bcl-2, mcl-1 and bax.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Aihara, M., Scardino, P. T., Truong, L. D., Wheeler, T. M., Goad, J. R., Yang, G. & Thompson, T. C. (1995). The frequency of apoptosis correlates with the prognosis of Gleason grade 3 adenocarcinoma of the prostate. Cancer 75: 522–529.
Arai, T. & Kino, I. (1995). Role of apoptosis in modulation of the growth of human colorectal tubular and villous adenomas. J Pathol 176: 37–44.
Auerbach, O., Gere, B., Forman, J., Petrick, T., Smolin, H., Muehsam, G., Kassouny, D. & Stout, A. (1957). Changes in the bronchial epithelium in relation to smoking and cancer of the lung. N Engl J Med 256: 97–104.
Auerbach, O., Stout, A., Hammond, E. & Garfinkel, L. (1962). Changes in bronchial epithelium in relation to sex, age, residence, smoking and pneumonia. N Engl J Med 267: 111–125.
Auerbach, O., Hammond, E. & Garfinkel, L. (1979). Changes in bronchial epithelium in relation to cigarette smoking, 1955–1960 vs. 1970–1977. N Engl J Med 300: 381–386.
Bakhshi, A., Jensen, J. P., Goldman, P., Wright, J. J., Wesley McBride, O. Epstein, A. L. & Korsmeyer, S. J. (1985). Cloning of the chromosomal breakpoint of t(14:18) human lymphomas: clustering around JHon chromosome 14 and near a transcriptional unit on chromosome 18. Cell 41: 899–906.
Bardeesy, N., Beckwith, J. B. & Pelletier, J. (1995). Clonal expansion and attenuated apoptosis in Wilms’ tumors are associated with p53 gene mutations. Cancer Res 55: 215–219.
Baretton, G. B., Diebold, J., Christoforis, G., Vogt, M., Müller, C., Dopfer, K., Schneiderbanger, K., Schmidt, M. & Löhrs, U. (1996). Apoptosis and immunohistochemical bcl-2 expression in colorectal adenomas and carcinomas. Aspects of carcinogenesis and prognostic significance. Cancer 77: 255–264.
Bennett, W. P., Colby, T. V., Travis, W. D., Borkowski, A., Jones, R. T., Lane, D. P., Metcalf, R. A., Samet, J. M., Takeshima, Y., Gu, J. R., Vähäkangas, K. V., Soini, Y., Pääkkö, P., Welsh, J. A., Trump, B. F. & Harris, C. C. (1993). p53 protein accumulates frequently in early bronchial neoplasia. Cancer Res 53: 4817–4822.
Birchall, M. A., Winterford, C. M., Allan, D. J. & Harmon, B. V. (1995). Apoptosis in normal epithelium, premalignant and malignant lesions of the oropharynx and oral cavity: a preliminary study. Eur J Cancer Oral Oncol 31B: 380–383.
Bodrug, S. E., Aimé-Sempé, C., Sato, T., Krajewski, S., Hanada, M. & Reed, J. C. (1995). Biochemical and functional comparisons of Mcl-1 and Bcl-2 proteins: evidence for a novel mechanism of regulating Bcl-2 family protein function. Cell Death Differ 2: 173–182.
Eerola, A-K, Törmänen, U., Rainio, P., Vähäkangas, K., Sormunen, R., Bloigu, R., Lehto, V-P & Pääkkö, P. (1997). Apoptosis in operated small cell lung carcinoma is inversely related to tumour necrosis and p53 immunoreactivity. J Pathol 181: 172–177.
Greenblatt, M. S., Bennett, W. P., Hollstein, M. & Harris, C. C. (1994). Mutations in the p53 tumor suppressor gene: clues to cancer etiology and molecular pathogenesis. Cancer Res 54: 4855–4878.
Hellquist, H. B., Olejnicka, B., Jadner, M., Andersson, T. & Sederholm, C. (1997). Fas receptor is expressed in human lung squamous cell carcinomas, whereas bcl-2 and apoptosis are not pronounced: a preliminary report. Br J Cancer 76: 175–179.
Hirano, T., Franzén, B., Kato, H., Ebihara, Y. & Auer, G. (1994). Genesis of squamous cell lung carcinoma. Sequential changes of proliferation, DNA ploidy and p53 expression. Am J Pathol 144: 296–302.
Hockenbery, D. M., Zutter, M., Hickey, W., Nahm, M. & Korsmeyer, S. J. (1991). Bcl-2 protein is topographically restricted in tissues characterized by apoptotic cell death. Proc Natl Acad Sci USA 88: 6961–6965.
Ishida, M., Gomyo, Y., Tatebe, S., Ohfuji, S. & Ito, H. (1996). Apoptosis in human gastric mucosa, chronic gastritis, dysplasia and carcinoma: analysis by terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labelling. Virchows Arch 428: 229–235.
Kerr, J. F. R., Wyllie, A. H. & Currie, A. R. (1972). Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26: 239–257.
King, E. D., Matteson, J., Jacobs, S. C. & Kyprianou, N. (1996). Incidence of apoptosis, cell proliferation and bcl-2 expression in transitional cell carcinoma of the bladder: association with tumor progression. J Urol 155: 316–320.
Lipponen, P. K. & Aaltomaa, S. (1994). Apoptosis in bladder cancer as related to standard prognostic factors and prognosis. J Pathol 173: 333–339.
Lipponen, P., Aaltomaa, S., Kosma, V-M & Syrjänen, K. (1994). Apoptosis in breast cancer as related to histopathological characteristics and prognosis. Eur J Cancer 14: 2068–2073.
Lowe, S. W., Bodis, S., McClatchey, A., Remington, L., Ruley, H. E., Fisher, D. E., Housman, D. E. & Jacks, T. (1994). p53 status and the efficacy of cancer therapy in vivo. Science 266: 807–810.
Koshida, Y., Saegusa, M. & Okayasu, I. (1997). Apoptosis, cell proliferation and expression of bcl-2 and bax in gastric carcinomas: immunohistochemical and clinicopathological study. Br J Cancer 75: 367–373.
Kroemer, G. (1997). The proto-oncogene Bcl-2 and its role in regulating apoptosis. Nature Med 6: 614–620.
Lu, Q-L Poulsom, R., Wong, L. & Hanby, A. M. (1993). bcl-2 expression in adult and embryonic non-haematopoietic tissues. J Pathol 169: 431–437.
Minn, A. J., Vélez, P., Schendel, S. L., Liang, H., Muchmore, S. W., Fesik, S. W., Fill, M. & Thompson, G. B. (1997). Bcl-xLforms an ion channel in synthetic lipid membranes. Nature 385: 353–357.
Muzio, M., Chinnaiyan, A. M., Kischkel, F. C., O’Rourke, K., Schevchenko, A., Ni, J., Scaffidi, C., Bretz, J. D., Zhang, M., Gentz, R., Mann, M., Krammer, P. H., Peter, M. E. & Dixit, V. M. (1996). FLICE, a novel FADD-homologous ICE/CED-3-like protease, is required to the CD95 (Fas/APO-1) death-inducing signaling complex. Cell 85: 817–827.
Nagata, S. (1997). Apoptosis by death factor. Cell 88: 355–365.
Nuorva, K., Soini, Y., Kamel, D., Autio-Harmainen, H., Risteli, L., Risteli, J., Vähäkangas, K. & Pääkkö, P. (1993). Concurrent p53 expression in bronchial dysplasias and squamous cell lung carcinomas. Am J Pathol 142: 725–732.
Oltvai, Z., Milliman, C. & Korsmeyer, S. J. (1993). Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 74: 609–619.
Reed, J. C. (1994). Bcl-2 and the regulation of programmed cell death. J Cell Biol 124: 1–6.
Saegusa, M., Kamata, Y., Isono, M. & Okayasu, I. (1996). bcl-2 expression is correlated with a low apoptotic index and is associated with progesterone receptor immunoreactivity in endometrial carcinomas. J Pathol 180: 275–282.
Soini, Y., Kamel, D., Nuorva, K., Lane, D. P., Vähäkangas, K. & Pääkkö, P. (1992). Low p53 protein expression in salivary gland tumours compared with lung carcinomas. Virchows Arch (Pathol Anat) 421: 415–420.
Soini, Y., Virkajärvi, N., Lehto, V-P & Pääkkö, P. (1996). Hepatocellular carcinomas with a high proliferation index and a low degree of apoptosis and necrosis are associated with a shortened survival. Br J Cancer 73: 1025–1030.
Sozzi, G., Miozzo, M., Donghi, R., Pilotti, S., Cariani, C. T., Pastorino, U., Della Porta, G. & Pierotti, M. A. (1992). Deletions of 17p and p53 mutations in preneoplastic lesions of the lung. Cancer Res 52: 6079–6082.
Staunton, M. J. & Gaffney, E. F. (1995). Tumor type is a determinant of susceptibility to apoptosis. Am J Clin Pathol 103: 300–307.
Sundaresan, V., Ganly, P., Hasleton, P., Rudd, R., Sinha, G., Bleehen, N. M. & Rabbits, P. (1992). p53 and chromosome 3 abnormalities, characteristic of malignant lung tumours, are detectable in preinvasive lesions of the bronchus. Oncogene 7: 1989–1997.
Törmänen, U., Eerola, A-K, Rainio, P., Vähäkangas, K., Soini, Y., Sormunen, R., Bloigu, R., Lehto, V-P & Pääkkö, P. (1995). Enhanced apoptosis predicts shortened survival in non-small cell lung carcinoma. Cancer Res 55: 5595–5602.
Tsujimoto, Y., Finger, L. R., Yunis, J., Nowell, P. C. & Croce, C. M. (1984). Cloning of the chromosome breakpoint of neoplastic B cells with the t(14:18) chromosome translocation. Science 226: 1097–1099.
Vähäkangas, K. H., Samet, J. M., Metcalf, R. A., Welsh, J. A., Bennett, W. P., Lane, D. P. & Harris, C. C. (1992). Mutations of p53 and ras genes in radon-associated lung cancer from uranium miners. Lancet 339: 576–580.
Walker, C., Robertson, L. J., Myskow, M. W., Pendleton, N. & Dixon, G. R. (1994). p53 expression in normal and dysplastic bronchial epithelium and in lung carcinomas. Br J Cancer 70: 297–303.
Walker, C., Robertson, L., Myskow, M. & Dixon, G. (1995). Expression of the bcl-2 protein in normal and dysplastic bronchial epithelium and in lung carcinomas. Br J Cancer 72: 164–169.
World Health Organization (1981). Histological Typing of Lung Tumours. International Classification of Tumours. No 1, World Health Organization: Geneva
Yang, J., Xuesong, L., Bhalla, K., Caryn Naekyung, K., Ibrado, A. M., Cai, J., Tsung-IP Jones, D. P. & Wang, X. (1997). Prevention of apoptosis by bcl-2: release of cytochrome c from mitochondria blocked. Science 275: 1129–1132.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Törmänen, U., Nuorva, K., Soini, Y. et al. Apoptotic activity is increased in parallel with the metaplasia–dysplasia–carcinoma sequence of the bronchial epithelium. Br J Cancer 79, 996–1002 (1999). https://doi.org/10.1038/sj.bjc.6690159
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690159
Keywords
This article is cited by
-
The role and prognostic value of apoptosis in colorectal carcinoma
BMC Clinical Pathology (2013)
-
MCM2 - a promising marker for premalignant lesions of the lung: a cohort study
BMC Cancer (2001)
-
In vivo cell kinetics in breast carcinogenesis
Breast Cancer Research (2001)
-
Expression of Bax and Apoptosis-Related Proteins in Human Esophageal Squamous Cell Carcinoma Including Dysplasia
Modern Pathology (2001)