Summary
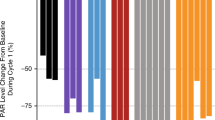
We conducted a phase I and pharmacokinetic study of the topoisomerase II catalytic inhibitor fostriecin. Fostriecin was administered intravenously over 60 min on days 1–5 at 4-week intervals. Dose was escalated from 2 mg m–2day–1to 20 mg m–2day–1in 20 patients. Drug pharmacokinetics was analysed with high performance liquid chromatography with UV-detection. Plasma collected during drug administration was tested in vitro for growth inhibition of a teniposide-resistant small-cell lung cancer (SCLC) cell line. The predominant toxicities were elevated liver transaminases (maximum common toxicity criteria (CTC) grade 4) and serum creatinine (maximum CTC grade 2). These showed only a limited increase with increasing doses, often recovered during drug administration and were fully reversible. Duration of elevated alanine–amino transferase (ALT) was dose-limiting in one patient at 20 mg m–2. Other frequent toxicities were grade 1–2 nausea/vomiting, fever and mild fatigue. Mean fostriecin plasma half-life was 0.36 h (initial; 95% CI, 0–0.76 h) and 1.51 h (terminal; 95% CI, 0.41–2.61 h). A metabolite, most probably dephosphorylated fostriecin, was detected in plasma and urine. No tumour responses were observed, but the plasma concentrations reached in the patients were insufficient to induce significant growth inhibition in vitro. The maximum tolerated dose (MTD) has not been reached, because drug supply was stopped at the 20 mg m–2dose level. However, further escalation seems possible and is warranted to achieve potentially effective drug levels. Fostriecin has a short plasma half-life and longer duration of infusion should be considered.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Beck, W. T. & Danks, M. K. (1991). Mechanisms of resistance to drugs that inhibit DNA topoisomerases. Sem Cancer Biol 2: 235–244.
Boritzki, T. J., Wolfard, T. S., Besserer, J. A., Jackson, R. C. & Fry, D. W. (1988). Inhibition of type II topoisomerase by fostriecin. Biochem Pharmacol 37: 4063–4068.
Bouma, P. & Uges, D. R. A. (1980). Preparation of highly efficient columns for high-performance liquid chromatography. In: The Serum Concentration of Drugs, International Congress Series 501, Merkus FWHM (ed), pp 278–280, Excerpta Medica: Amsterdam
Clinical Brochure Fostriecin, (1991). Bethesda, MD, USA National Cancer Institute
Cummings, J. & Smyth, J. F. (1993). DNA topoisomerase I and II as targets for rational design of new anticancer drugs. Ann Oncol 4: 533–543.
De Jong, S., Zijlstra, J. G., Mulder, N. H. & De Vries, E. G. E. (1991). Lack of cross-resistance to fostriecin in a human small-cell lung carcinoma cell line showing topoisomerase II-related drug resistance. Cancer Chemother Pharmacol 28: 461–464.
De Jong, R. S., De Vries, E. G. E., Meijer, S., De Jong, P. E. & Mulder, N. H. (1998). Renal toxicity of the anticancer drug fostriecin. Cancer Chemother Pharmacol 42: 160–164.
Froehlich-Ammon, S. J. & Osheroff, N. (1995). Topoisomerase poisons: harnessing the dark side of enzyme mechanism. J Biol Chem 270: 21429–21432.
Fry, D. W., Besserer, J. A. & Boritzki, T. J. (1984). Transport of the anti-tumour antibiotic CI-920 into L1210 leukemia cells by the reduced folate carrier system. Cancer Res 44: 3366–3370.
Guo, X. W., Thng, J. P. H., Swank, R. A., Anderson, H. J., Tudan, C., Bradbury, E. M. & Roberge, M. (1995). Chromosome condensation induced by fostriecin does not require p34(cdc2) kinase activity and histone H1 hyperphosphorylation, but is associated with enhanced histone H2A and H3 phosphorylation. EMBO J 14: 976–985.
Leopold, W. R., Shillis, J. L., Mertus, A. E., Nelson, J. M., Roberts, B. J. & Jackson, R. C. (1984). Anticancer activity of the structurally novel antibiotic CI-920 and its analogues. Cancer Res 44: 1928–1932.
Osheroff, N., Zechiedrich, E. L. & Gale, K. C. (1991). Catalytic function of DNA topoisomerase II. BioEssays 13: 269–275.
Pillon, L., Moore, M. J. & Thiessen, J. J. (1994). Determination of fostriecin pharmacokinetics in plasma using high-pressure liquid chromatography assay. Ther Drug Monit 16: 186–190.
Proost, J. H. & Meijer, D. K. F. (1992). MW/PHARM, an integrated software package for drug dosage regimen calculation and therapeutic drug monitoring. Comput Biol Med 22: 155–160.
Roberge, M., Tudan, C., Hung, S. M., Harder, K. W., Jirik, F. R. & Anderson, H. (1994). Anti-tumour drug fostriecin inhibits the mitotic entry checkpoint and protein phosphatases 1 and 2A. Cancer Res 54: 6115–6121.
Scheithauer, W., Von Hoff, D. D., Clark, G. M., Shillis, J. L. & Elslager, E. F. (1986). In vitro activity of the novel antitumour antibiotic fostriecin (CI-920) in a human tumour cloning assay. Eur J Cancer Clin Oncol 22: 921–926.
Schilsky, R. L., Ramirez, J., Wilson, K., Vokes, E., Kobayashi, K., Berezin, F., Wulff, W. & Ratain, M. J. (1994). Phase I clinical and pharmacological study of fostriecin in patients with advanced cancer. Proc Am Soc Clin Oncol 13: 321
Susick, R. L., Hawkins, K. L. & Pegg, D. G. (1990). Preclinical toxicological evaluation of fostriecin, a novel anticancer antibiotic, in rats. Fundam Appl Toxicol 15: 258–269.
Timmer-Bosscha, H., Hospers, G. A. P., Meijer, C., Mulder, N. H., Muskiet, F. A. J., Martini, I. A., Uges, D. R. A. & De Vries, E. G. E. (1989). Influence of docosahexaenoic acid on cisplatin resistance in a human small cell lung carcinoma cell line. J Natl Cancer Inst 81: 1069–1075.
Withoff, S., De Vries, E. G. E., Keith, W. N., Nienhuis, E. F., Van der Graaf, W. T. A., Uges, D. R. A. & Mulder, N. H. (1996). Differential expression of DNA Topoisomerase IIα and β in P-gp and MRP negative VM26, m-AMSA and mitoxantrone resistant sublines of the human SCLC cell line GLC4 . Br J Cancer 74: 1869–1876.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Jong, R., Mulder, N., Uges, D. et al. Phase I and pharmacokinetic study of the topoisomerase II catalytic inhibitor fostriecin. Br J Cancer 79, 882–887 (1999). https://doi.org/10.1038/sj.bjc.6690141
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690141
Keywords
This article is cited by
-
Anti-proliferative effects of simocyclinone D8 (SD8), a novel catalytic inhibitor of topoisomerase II
Investigational New Drugs (2010)
-
Genetic manipulation of the biosynthetic process leading to phoslactomycins, potent protein phosphatase 2A inhibitors
Journal of Industrial Microbiology & Biotechnology (2006)
-
A pH-Stability Study of Phoslactomycin B and Analysis of the Acid and Base Degradation Products
The Journal of Antibiotics (2005)