Summary
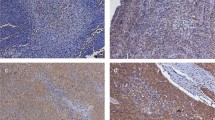
The syndecans are a family of cell-surface heparan sulphate proteoglycans that regulate cell behaviour by binding extracellular matrix molecules such as growth factors. The syndecan family has four members, of which syndecan-1 is the most studied and best characterized. We have studied the prognostic significance of syndecan-1 expression in squamous cell carcinoma (SCC) of the head and neck treated with surgery and post-operative radiotherapy. Paraffin-embedded tissue samples taken from 175 patients with primary SCC, followed up from 2 to 15 years after surgery, were studied for expression of syndecan-1 by immunohistochemistry. A low number (≤50%, the median value) of syndecan-1-positive tumour cells was associated with low histological grade of differentiation (P < 0.0001), a large primary tumour size (T1–2 vs T3–4, P = 0.02), positive nodal status (N0 vs N1–3, P = 0.0006), and high clinical stage (stage I or II vs III or IV, P < 0.0001). Low syndecan-1 expression was also associated with unfavourable overall survival in a univariate analysis (P = 0.001). In a multivariate survival analysis, the clinical stage and syndecan-1 expression were the only independent prognostic factors. We conclude that syndecan-1 is a novel prognostic factor in SCC of the head and neck treated with surgery and post-operative radiotherapy.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Benasso, M., Bonelli, L., Numico, G., Corvo, R., Sanguineti, G., Rosso, R., Vitale, V. & Merlano, M. (1997). Treatment with cisplatin and fluorouracil alternating with radiation favourably affects prognosis of inoperable squamous cell carcinoma of the head and neck: results of a multivariate analysis on 273 patients. Ann Oncol 8: 773–779.
Bernfield, M., Kokenyesi, R., Kato, M., Hinkes, M. T., Spring, J., Gallo, R. L. & Lose, E. (1992). Biology of syndecans. Annu Rev Cell Biol 8: 365–393.
Bundgaard, T., Sørensen, F. B., Gaihede, M., Søgaard, H. & Overgaard, J. (1992). Stereometric, histopathologic, flow cytometric and clinical parameters in the prognostic evaluation of 74 patients with intraoral squamous cell carcinomas. Cancer 70: 1–13.
Cook, D. M., Hinkes, M. T., Bernfield, M. & Rausher, FLIII (1997). Transcriptional activation of the syndecan-1 promoter by the Wilms’ tumor protein WT1. Oncogene 13: 1789–1799.
Elenius, K., Salmivirta, M., Inki, P., Mali, M. & Jalkanen, M. (1990). Binding of human syndecan to extracellular matrix proteins. J Biol Chem 29: 17837–17843.
Elenius, K., Vainio, S., Laato, M., Salmivirta, M., Thesleff, I. & Jalkanen, M. (1991). Induced expression of syndecan in healing wounds. J Cell Biol 114: 585–595.
Elenius, K., Määttä, A., Salmivirta, M. & Jalkanen, M. (1992). Growth factors induce 3T3 cells to express bFGF-binding syndecan. J Biol Chem 25: 6435–6441.
El-Sayed, S. & Nelson, N. (1996). Adjuvant and adjunctive chemotherapy in the management of squamous cell carcinoma of the head and neck region. A meta-analysis of prospective and randomized trials. J Clin Oncol 14: 838–847.
Hayashi, K., Hayashi, M., Jalkanen, M., Firestone, J. H., Trelstad, R. L. & Bernfield, M. (1987). Immunocytochemistry of cell surface heparan sulfate proteoglycan in mouse tissue. A light and electron microscopic study. J Histochem Cytochem 35: 1079–1088.
Horiot, J. C., Le Fur, R., N’guyen, T., Chenal, C., Schraub, S., Alfonsi, S., Gardani, G., VanDenBogaert, W., Danzak, S., Bolla, M., VanGlabbeke, M. & DePauw, M. (1992). Hyperfractionation versus conventional fractionation in oropharyngeal carcinoma: final analysis of a randomized trial of the EORTC cooperative group of radiotherapy. Radiother Oncol 25: 231–241.
Inki, P., Stenbäck, F., Talve, L. & Jalkanen, M. (1991). Immunohistochemical localization of syndecan in mouse skin tumors induced by UV irradiation. Am J Pathol 139: 1333–1340.
Inki, P., Kujari, H. & Jalkanen, M. (1992). Syndecan in carcinomas produced from transformed epithelial cells in nude mice. Lab Invest 66: 314–323.
Inki, P., Larjava, H., Haapasalmi, K., Miettinen, H., Grenman, R. & Jalkanen, M. (1994). Expression of syndecan-1 is induced by differentiation and suppressed by malignant transformation of human keratinocytes. Eur J Cell Biol 63: 43–51.
Inki, P., Joensuu, H., Grenman, R., Klemi, P. & Jalkanen, M. (1994). Association between syndecan-1 expression and clinical outcome in squamous cell carcinoma of the head and neck. Br J Cancer 70: 319–323.
Jalkanen, M., Elenius, K. & Rapraeger, A. (1993). Syndecan: regulator of cell morphology and growth factor action at the cell–matrix interface. Trends Glycosci Glycotechn 5: 107–120.
Koda, J., Rapraeger, A. & Bernfield, M. (1985). Heparan sulfate proteoglycans from mouse mammary epithelial cells. Cell surface proteoglycan as a receptor for interstitial collagens. J Biol Chem 260: 8157–8162.
Kojima, T., Scworak, N. W. & Rosenberg, R. D. (1992). Molecular cloning and expression of two distinct cDNAs encoding heparan sulfate proteoglycan core proteins from a rat endothelial cell line. J Biol Chem 267: 4870–4877.
Leppä, S., Mali, M., Miettinen, H. & Jalkanen, M. (1992). Syndecan expression regulates cell morphology and growth of mouse mammary epithelial tumor cells. Proc Natl Acad Sci USA 89: 932–936.
Liotta, L. A., Rao, C. N. & Wewer, U. M. (1986). Biochemical interactions of tumor cells with the basement membrane. Annu Rev Biochem 55: 1037–1051.
Liu, K., Kasper, M., Bierhaus, A., Langer, S., Peterson, I., Muller, M. & Trott, K. R. (1997). Differential expression of CD44 and CD44v10 proteins and syndecan in normal and irradiated mouse epidermis. Histochem Clee Biol 107: 159–167.
Pera, E., Moreno, A. & Galino, L. (1986). Prognostic factors in laryngeal carcinoma. Cancer 58: 928–934.
Pulkkinen, J. O., Penttinen, M., Jalkanen, M., Klemi, P. & Grenman, R. (1997). Syndecan-1: a new prognostic marker in laryngeal cancer. Acta Otolaryngol (Stockh) 117: 312–315.
Rapraeger, A. & Bernfield, M. (1983). Heparan sulfate proteoglycans from mouse mammary epithelial cells. A putative membrane proteoglycan associates quantitatively with lipid vesicles. J Biol Chem 258: 3632–3636.
Ruoslahti, E. (1989). Proteoglycans in cell regulation. J Biol Chem 264: 13369–13372.
Salmivirta, M., Elenius, K., Vainio, S., Hofer, U., Chiquet-Erismann, R., Thesleff, I. & Jalkanen, M. (1991). Syndecan from embryonic tooth mesenchyme binds tenascin. J Biol Chem 266: 7733–7739.
Salmivirta, M., Rauvala, H., Elenius, K. & Jalkanen, M. (1992). Neurite growth-promoting protein (amphoterin, p30) binds syndecan. Exp Cell Res 200: 444–451.
Salmivirta, M., Mali, M., Heino, J., Hermonen, J. & Jalkanen, M. (1994). A novel laminin-binding form of syndecan-1 (cell surface proteoglycan) produced by syndecan-1 cDNA-transfected NIH-3T3 cells. Exp Cell Res 215: 180–188.
Saunders, S. & Bernfield, M. (1988). Cell surface proteoglycan binds mouse mammary epithelial cells to fibronectin and behaves as a receptor on interstitial matrix. J Cell Biol 106: 423–430.
Shanmugaratnam, K. & Sobin, L. H. (1978). Histological Typing of Upper Respiratory Tract Tumours, pp. 14–33, WHO: Geneva
Sun, X., Mosher, D. F. & Rapraeger, A. (1989). Heparan sulfate-mediated binding of epithelial cell surface proteoglycan to thrombospondin. J Biol Chem 264: 2885–2889.
Thesleff, I., Jalkanen, M., Vainio, S. & Bernfield, M. (1988). Cell surface proteoglycan expression correlates with epithelial–mesenchymal interaction during tooth morphogenesis. Dev Biol 129: 565–572.
Vainio, S., Lehtonen, E., Jalkanen, M., Bernfield, M. & Saxen, L. (1989). Epithelial–mesenchymal interactions regulate the stage-specific expression of a cell surface proteoglycan, in the developing kidney. Dev Biol 134: 382–391.
Weinstein, R. S., Merk, F. B. & Alroy, J. (1976). The structure and function of intercellular junctions in cancer. Adv Cancer Res 23: 25–33.
Wiernik, G., Millard, P. R. & Haybittle, J. L. (1991). The predictive value of histological classification into degrees of differentiation of squamous cell carcinoma of the larynx and hypopharynx compared with the survival of patients. Histopathology 19: 411–417.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Anttonen, A., Kajanti, M., Heikkilä, P. et al. Syndecan-1 expression has prognostic significance in head and neck carcinoma. Br J Cancer 79, 558–564 (1999). https://doi.org/10.1038/sj.bjc.6690088
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690088
Keywords
This article is cited by
-
Biomarkers in Head and Neck Cancer an Update
Indian Journal of Otolaryngology and Head & Neck Surgery (2019)
-
Syndecan-1 induction in lung microenvironment supports the establishment of breast tumor metastases
Breast Cancer Research (2018)
-
Proteoglycan-based diversification of disease outcome in head and neck cancer patients identifies NG2/CSPG4 and syndecan-2 as unique relapse and overall survival predicting factors
BMC Cancer (2015)
-
Clinical implications in the shift of syndecan-1 expression from the cell membrane to the cytoplasm in bladder cancer
BMC Cancer (2014)
-
Prognostic significance of syndecan-1 expression in squamous cell carcinoma of the tonsil
International Journal of Clinical Oncology (2014)