Abstract
Multiparity decreases the risk of breast cancer in white women, whereas it is a risk factor in black women <50 years. Early-onset breast cancer (<50 years) has been associated with high insulin-like growth factor-1 (IGF-1) levels. Absence of the common IGF1 19 cytosine-adenine (CA)-repeat allele (IGF1-19/-19) inverts the effect of several non-genetic factors on breast cancer risk but the interaction between IGF1-19/-19 and multiparity on breast cancer risk is unknown. As IGF1-19/-19, multiparity and early-onset breast cancer are more common in black than in white women, we aimed to study whether multiparity combined with IGF1-19/-19 increases the risk of early-onset breast cancer. Four hundred and three breast cancer patients diagnosed in Lund, Sweden, at age 25–99 years were genotyped for the IGF1 CA-repeat length using fragment analysis. Overall, 12.9% carried the IGF1-19/-19 genotype. There was a highly significant interaction between multiparity and IGF1-19/-19 on age at breast cancer diagnosis (P=0.007). Among IGF1-19/-19 patients, multiparity was associated with a 9.2 year earlier age at diagnosis compared with uniparity or nulliparity (P=0.006). Multiparity combined with IGF1-19/-19 was associated with an early age at breast cancer diagnosis. If confirmed, IGF1-19/-19 may help identify a subgroup of women for earlier breast cancer screening.
Similar content being viewed by others
Main
Breast cancer is the most common malignancy in women worldwide (Parkin et al, 2001, 2002). Approximately 10% of women living in Sweden will develop the disease during their lifetime. The median age at breast cancer diagnosis in women in Sweden is 60 years; approximately 4% of women are 40 years or younger at diagnosis (data from the Swedish Cancer Registry). The prognosis is often poorer in younger than in older women (Sidoni et al, 2003) in terms of overall survival and relapse (Han et al, 2004), partly because young women do not benefit from breast cancer screening and early-onset breast cancers are usually detected clinically at a more advanced stage. In Lund, Sweden, mammography screening is initiated at age 45 years and is performed every 18 months until the age of 74 years. As of today, no tests are routinely performed to identify the subgroup of women who do not belong to high-risk breast cancer families (eg BRCA1 families) but who would nevertheless benefit from breast cancer screening at an earlier age owing to an increased risk for early-onset breast cancer.
Breast cancer risk is determined by genetic factors and non-genetic factors, for example, reproductive factors (Beral and Reeves, 1993; Kelsey et al, 1993). An increased age at first full-term pregnancy and low parity have been associated with an increased risk of developing breast cancer in the developed world (Kelsey et al, 1993). In the short term, a first full-term pregnancy increases the risk of breast cancer (Beral and Reeves, 1993). Each subsequent live-birth reportedly decreases the risk of breast cancer by 30% and after three full-term pregnancies, the short-term risk of developing breast cancer is similar to the risk of nulliparous women (McCredie et al, 1998). In the long-term, a full-term pregnancy confers a decreased risk of developing breast cancer (McCredie et al, 1998) and each subsequent full-term pregnancy magnifies this protective effect.
In the developing world, a higher proportion of the diagnosed breast cancers are early-onset breast cancers Parkin et al (2002) compared to the developed world. This fact is unexpected, as multiple pregnancies and young age at first full-term pregnancy are much more common in the developing world than in the developed world. Therefore, the higher proportion of early-onset breast cancer may be owing to either genetic or non-genetic factors aside from reproductive factors. That genetic factors play an important role is suggested by studies, which show that early-onset breast cancer is more common in African-American women than in white women in the US (Amend et al, 2006; Ries et al, 2006) and that multiparity is associated with an increased breast cancer risk before age 45 years in young African-American women (Palmer et al, 2003), but not in young white women (Hall et al, 2005).
Insulin-like growth factor-1 (IGF-1) levels are higher in young black woman than in young white women (Jernström et al, 2001a) and a higher level of circulating IGF-1 is a known risk factor for early-onset breast cancer (Peyrat et al, 1993; Hankinson et al, 1998; Renehan et al, 2004; Sugumar et al, 2004; Fletcher et al, 2005; Schernhammer et al, 2005). It has been estimated that between 38 and 77% of the individual variation in IGF-1 levels is because of genetic factors (Harrela et al, 1996; Verhaeghe et al, 1996). Circulating IGF-1 levels may be modified (Rosen et al, 1998) by a cytosine-adenine (CA) repeat in the proximity of the promoter, 1 kb upstream from the transcription start site of the IGF1 gene (Rotwein et al, 1986). The 19-repeat allele is the most common CA repeat length among white women and only 6–13% of white women have no 19-repeat allele (IGF1-19/-19 genotype) (Jernström et al, 2001a, 2001b, 2005; Vaessen et al, 2001; DeLellis et al, 2003), whereas more than 32% of black women and men have the IGF1-19/-19 genotype (Jernström et al, 2001a; Schildkraut et al, 2005). The IGF1-19/-19 genotype is thus approximately three times more common in black women than in white women. Furthermore, the IGF1-19/-19 genotype is present in 16% of Indian-Pakistani women and in over 33% of other Asian women (Jernström et al, 2001a; Wen et al, 2005).
Breast cancer risk appears to be influenced by the interaction between the IGF1 genotype and non-genetic factors. Oral contraceptive (OC) use in combination with the IGF-19/-19 genotype increases the risk of early-onset breast cancer (Jernström et al, 2005; Cleveland et al, 2006). In women who never had used OCs, the IGF1-19/-19 genotype was associated with a decreased risk of early-onset breast cancer as compared with women who have at least one copy of the 19-repeat allele (IGF1+19) (Cleveland et al, 2006). Oral contraceptive use decreased IGF-1 levels in women with the IGF1+19 genotype but increased IGF-1 levels in white, black, Asian and Indian-Pakistani women with the IGF-19/-19 genotype (Jernström et al, 2001a). The effects of hormone replacement therapy (HRT), alcohol intake, body mass index (BMI) and smoking on breast cancer risk were all likewise modified by the presence or absence of the IGF-19/-19 genotype (Cleveland et al, 2006). The interaction between multiparity and the IGF1-19/-19 genotype on breast cancer risk was not addressed in the study by Cleveland et al, 2006.
The protective effect of increasing parity on breast cancer risk in the developed world is thought to be mediated in part by decreasing circulating IGF-1 levels (Holmes et al, 2002). As multiparity is protective against breast cancer in the developed world but not in the developing world, as the IGF1-19/-19 genotype is more common among black, Indian-Pakistani and other Asian women than among white women, and as the IGF1-19/-19 genotype inverts the effect of several non-genetic factors on breast cancer risk, we aimed to investigate whether the effect of multiparity on age at breast cancer diagnosis is influenced by the IGF1-19/-19 genotype.
Materials and methods
All women with a primary breast cancer were invited to take part in an ongoing study of breast cancer at the Lund University Hospital, Sweden, regardless of ethnic background, age and stage. Patients who had recently been diagnosed and treated for another type of cancer were not eligible to participate. Four hundred and three patients were included between October 2002 and July 2006. The study was approved by the Ethics Committee of the Lund University. All patients provided a written informed consent.
During the pre-operative visit to the Department of Surgery of the Lund University Hospital, a research nurse collected blood samples (EDTA plasma and serum) and recorded the time and date when the blood samples were drawn. The blood was centrifuged and separated. Serum, EDTA plasma and blood cells were stored at −70°C. All samples were labelled with serial codes to enable blinded analyses.
Body weight, height, waist and hip circumferences and breast volumes were measured. The breast volume was measured with plastic cups used by plastic surgeons doing breast reductions. They come in the following 11 sizes: 200, 275, 350, 500, 650, 800, 950, 1150, 1325, 1500 and 2000 ml (Ringberg et al, 2006).
All patients filled out a baseline questionnaire including questions on the use of exogenous hormones and concomitant medications, smoking, alcohol intake, reproductive history and family history of cancer.
Patients who had not experienced a menstrual period during the last year were defined as postmenopausal. However, postmenopausal patients who used HRT may have had HRT induced bleeding and may therefore have been misclassified as premenopausal. Patients who had had their uterus removed before menopause but not their ovaries may also have been misclassified as postmenopausal. We therefore classified patients according to age (< 50 years or ⩾50 years of age) instead of reported menopausal status. We chose the cutoff at age 50 years as others have shown that high IGF-1 levels influence the risk of breast cancer in women under the age of 50 years (Hankinson et al, 1998). We also chose to use a cutoff age of 45 years as Lund's screening program for breast cancer starts at this age.
Additional baseline information including type of surgery, sentinel node biopsy, axillary node dissection, tumour size, histological type and grade, axillary node involvement, signs of distant metastases, oestrogen receptor and progesterone receptor status was obtained from each patient's chart and pathology report. Oestrogen receptor status and progesterone receptor status were reported as either positive or negative. HER-2/neu status was not routinely analysed.
Genetic analysis
Genomic DNA was extracted from 300 μl of peripheral blood using Wizard, Genomic DNA Purification Kit (Promega, Madison, WI, USA). The CA repeat in the proximity of the IGF1 promoter ranged in size from 11 to 23 repeats. Polymerase chain reaction (PCR) primers 5′-GCTAGCCAGCTGGTGTTATT and 5′-GTTTCTTACCACTCTGGGAGAAGGGTA were used, where the forward primer was fluorescently labelled with FAM (MWG-Biotech AG, Ebersberg, Germany). Polymerase chain reaction was performed in 15 μl reactions using 25 ng of DNA, 0.4 μ M of each primer, 0.1 μ M of each deoxynucleotide (Amersham Biosciences, Buckinghamshire, UK), 5% DMSO (Sigma, St Louis, MO, USA), 2.5 mM MgCl2 (Applied Biosystems, Foster City, CA, USA), 1 × PCR Gold Buffer (Applied Biosystems, Foster City, CA, USA) and 1 U AmpliTaq Gold (Applied Biosystems, Foster City, CA, USA).
The PCR product was analysed in an ABI3100 Prism Genetic Analyzer (Applied Biosystems, Foster City, CA, USA) and the results were evaluated using Genescan software. The number of repeats was determined by sequencing samples of varying sizes (Big Dye, Terminator Cycle Sequencing, Applied Biosystems, Foster City, CA, USA) and using them as standards in the fragment analysis. The results from the ABI3100 Prism Genetic Analyzer were manually evaluated and each sample was read in duplicate. The first 360 samples were analysed using this method.
For quality control, the first 352 samples were run in duplicate in separate PCR and fragment analyses. Thereafter, every fourth sample was run in duplicate. The concordance rate was 98.3%. The discordant samples were re-checked, and we found that the genotypes were identical in the separate PCR and fragment analyses, but that the wrong peaks had been manually selected during one of the readings.
In April 2006, the system was upgraded to an ABI Prism 3130xl Genetic Analyzer (Applied Biosystems, Foster City, CA, USA). Forty-three samples were run on the upgraded system. The repeat lengths were manually evaluated, as stated above, as well as automatically evaluated by the GeneMapper Software v.4.0. The concordance rate was 100% between the manual and automatic readings. The GeneMapper software could not determine repeat sizes below 17 repeats, but these alleles are rare (0.2%).
Statistical analysis
Data analysis was performed with the statistical software SPSS 13.0. for Windows (SPSS Inc. Chicago, IL, USA). For univariate analyses, χ2 square analysis was used for dichotomous variables and Student's t-test was used for continuous variables. Age at diagnosis was used as a continuous variable. Age at diagnosis was also categorised as age < 50 years (yes/no) and age < 45 years (yes/no). Multiparity was classified as having two or more children (yes/no). The IGF1-19/-19 genotype was classified as having no IGF1 19-repeat allele (yes/no). Multivariate linear regression was used to examine the relationship between the IGF1-19/-19 genotype and multiparity on age at breast cancer diagnosis. An interaction variable was computed between these two factors. Adjustments were carried out for other factors such as OC use, HRT use, alcohol intake, BMI and current smoking. The study was based on an a priori hypothesis, namely, that the IGF1 genotype in combination with multiparity, is associated with early-onset breast cancer. Therefore no adjustment for multiple testing is required for the P-value for interaction, neither in the univariate nor in the multivariate analyses. All P-values are two-tailed and presented without Bonferroni correction for multiple testing.
Results
Age at diagnosis and parity
The baseline characteristics for all patients (n=403) are shown in Table 1. Age at breast cancer diagnosis ranged from 25 to 99 years, with a mean age of 58.5 years. We classified patients diagnosed < 50 years of age as younger patients (n=96) and patients diagnosed ⩾50 years of age as older patients (n=307). One hundred and twenty-eight patients were nulliparous or uniparous whereas 275 patients were multiparous, that is, had two or more children. Parity did not differ between the younger and the older patients (1.8 children vs 1.9 children; P=0.49), but age at first full-term pregnancy was higher in the younger than in the older patients (26.6 vs 24.6 years; P=0.001, significant also after Bonferroni correction). The only factor included in Table 1 that was associated with the IGF1-19/-19 genotype was age at breast cancer diagnosis. Patients with the IGF1-19/-19 genotype had a 3.5-year earlier age at diagnosis compared with patients with the IGF1+19 genotype, (P=0.04).
IGF1-19/-19 genotype frequency and age at diagnosis
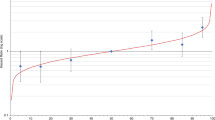
The allele frequency distribution of the IGF1 CA-repeat length in all patients is described in Table 2. Overall, 12.9% carried the IGF1-19/-19 genotype. The distribution was not equal in all age groups. The IGF1-19/-19 genotype was most common (over 20%) in patients diagnosed before age 45 years and in patients diagnosed between the ages of 55 and 59 years, and thus displayed a biphasic pattern, (Figure 1). Age did not differ between patients who were heterozygous and patients who were homozygous for the 19-repeat allele (59.0 vs 58.8 years; P=0.86). We therefore combined these two groups and classified their genotypes as IGF1+19.
Frequency of the IGF1-19/-19 genotype in the 403 patients according to age at diagnosis. The total number of women in each age category is indicated. The IGF1-19/-19 genotype was most common (over 20 %) in patients diagnosed before age 45 years and in patients diagnosed between the ages of 55 and 59 years and thus displayed a biphasic distribution.
The frequency of IGF1-19/-19 genotype in relation to parity and age at diagnosis
Younger patients with the IGF1-19/-19 genotype had more children than younger patients with the IGF1+19 genotype (2.4 children vs 1.7 children; P=0.01). In fact, only one young patient with the IGF1-19/-19 genotype had less than two children, that is, one child. Parity did not differ by IGF1-19/-19 genotype among the older patients (1.9 children vs 1.9 children; P=0.85).
Among the younger multiparous patients, 19.7% carried the IGF1-19/-19 genotype compared with 11.6% among all other patients, odds ratio (OR) 1.7 (95% confidence interval (CI) 0.96–3.0; P=0.07). As mammography is initiated at age 45 years in Lund, Sweden, we reanalysed our results using 45 years as an age cutoff. With this age cutoff, the IGF1-19/-19 genotype was more than twice as common in multiparous patients diagnosed before age 45 than in other patients (27.3% vs 11.6%), OR 2.3 (95% CI 1.3–4.4; P=0.01).
The effect of IGF1-19/-19 genotype and parity on age at diagnosis
In multiparous patients with the IGF1-19/-19 genotype, the mean age at breast cancer diagnosis was 9.2 years earlier compared with uniparous or nulliparous patients (P=0.006). Age at breast cancer diagnosis was not significantly influenced by multiparity in women with the IGF1+19 genotype (P=0.33). The interaction between multiparity and the IGF1-19/-19 genotype on age at breast cancer diagnosis, which was our a priori hypothesis, was highly significant (P=0.007) (Table 3), and it remained significant after adjustment for ever OC use, ever HRT use, alcohol intake, BMI and current smoking (Pinteraction=0.03).
Nine patients had twins. Five of these nine patients had only one full-term pregnancy. The results remained essentially unchanged when these patients were reclassified as uniparous instead of multiparous. We then analysed the effect of recognised pregnancies (whether full-term or not). The interaction between two or more recognised pregnancies and the IGF1-19/-19 genotype on the age at breast cancer diagnosis was still significant (P=0.02). The one uniparous patient with the IGF1-19/-19 genotype whose breast cancer was diagnosed before age 50 years had in fact had several recognised pregnancies; no patient with the IGF1-19/-19 genotype with less than two recognised pregnancies was diagnosed before age 57 years.
IGF1 genotype and mode of cancer detection, tumour characteristics and family history of breast cancer
Among the 322 patients aged 45–74 years, there was no difference in mode of detection in patients with or without the IGF1-19/-19 genotype (P=0.82). IGF1 genotype was associated with neither of the following tumour characteristics: tumour size, nodal involvement, grade, oestrogen receptor status or progesterone receptor status in either age group (all P values ⩾0.16) nor with a history of breast cancer in a first and/or second degree relative (P=0.70).
Discussion
The main finding of this study was that there was a highly significant interaction between multiparity and the IGF1-19/-19 genotype on age at breast cancer diagnosis. Multiparous patients with the IGF1-19/-19 genotype had a 9.2-year earlier age at breast cancer diagnosis compared with nulliparous or uniparous patients with the same genotype. Among our study population, no patient with the IGF1-19/-19 genotype and less than two recognised pregnancies (whether full-term or not) was diagnosed before age 57 years. Among women with the IGF1+19 genotype, multiparity had no significant effect on age at diagnosis. The interaction between the IGF1-19/-19 genotype and multiparity on the age at breast cancer diagnosis has to our knowledge not been reported previously.
We also found that the IGF1-19/-19 genotype was more than twice as common among the youngest patients (diagnosed <45 years of age) who were multiparous compared with all other patients. It was noteworthy that only one of the young breast cancer patients with the IGF1-19/-19 genotype had less than two children. This observation strengthens the hypothesis that it is the combination of the IGF1-19/-19 genotype and multiparity that is deleterious and suggests that the IGF1-19/-19 genotype may be associated with a substantial number of the early-onset breast cancers in the developing world where both this genotype and multiparity are common. Conversely, in China, where it is uncommon to have more than one child, the IGF1-19/-19 genotype was associated with a decreased risk of early onset breast cancer (Wen et al, 2005). Their result is in line with our finding that patients with the IGF1-19/-19 genotype and fewer than two pregnancies had a later age at diagnosis.
As suggested by Holmes et al (2002) parity may exert its protective effect by lowering IGF-1 levels post-partum. Such results were not replicated in a previous study of young white women from high-risk breast cancer families (Jernström et al, 2005). In that study, the IGF-1 levels of parous and nulliparous women were similar. Furthermore, we found that the frequency of the IGF1-19/-19 genotype was significantly higher among known BRCA1 carriers compared with other high-risk women and we have reported previously that each pregnancy up to three confers an increased risk for breast cancer before age 40 years among BRCA1 and BRCA2 carriers (Jernström et al, 1999). We therefore hypothesise that the IGF-1 levels do not decrease post-partum in women with the IGF1-19/-19 genotype. Each pregnancy may thus confer an increased risk of early-onset breast cancer.
In our study, patients with the IGF1-19/-19 genotype who had two or more children had a lower age at diagnosis. In the study by Cleveland et al (2006), the IGF1-19/-19 genotype modified the breast cancer risk after exposure to hormonal factors such as OCs and women with the IGF1-19/-19 genotype who had ever used OCs had an increased breast cancer risk whereas this genotype was protective in women without OC exposure. However, our results remained significant after adjustment for OC use, HRT use as well as alcohol intake, BMI and current smoking.
The frequency of the IGF1-19/-19 genotype was 12.9% in our patient population and is consistent with the frequency among white women that has been reported to vary between 6 and 13% (Vaessen et al, 2001; Jernström et al, 2001b, 2005; DeLellis et al, 2003). In this study, we observed a biphasic variation in the frequency of the IGF1-19/-19 genotype in relation to age at diagnosis. This observation needs confirmation in other studies, as it has not been previously reported. Among multiparous patients diagnosed before age 50 years, the frequency of the IGF1-19/-19 genotype was almost twice as common as in the other patients. As mammography screening is initiated at the age of 45 years in Lund, Sweden, we then re-analysed our results. The frequency of the IGF1-19/-19 genotype in the multiparous patients (< 45 years) was then more than twice as common as in the other patients. As examining the frequency of the IGF1-19/-19 genotype in relation to different age cutoffs was not part of our a priori hypothesis, these results have to be regarded as hypothesis generating. However, they suggest that multiparous patients with the IGF1-19/-19 may not only have an earlier age at breast cancer diagnosis but also an increased risk of developing the malignancy.
This study was based on a series of breast cancer patients diagnosed with a primary breast cancer at Lund University Hospital, Sweden. All breast cancers were verified by the Department of Pathology, Lund University Hospital. The baseline information was filled out by all patients at the pre-surgical visit, which minimises bias owing to survival differences between patients.
It has been suggested that contradictory findings regarding IGF1 CA-repeat lengths and breast cancer risk may be owing to differences in the methods used to determine the repeat lengths, sequencing or fragment analysis (Tran et al, 2004). The repeat numbers obtained with fragment analysis have differed when analysing the results on an ABI Prism 3100 Genetic Analyzer using the internal standard supplied by the manufacturer as compared to the results received when using a ladder that has been verified by direct sequencing (Rodriguez et al, 2006). It is therefore important to emphasise that all the genetic analyses in this study were performed with the same method, that is, fragment analysis on an ABI Prism 3100 or a 3130xl Genetic Analyzer, using control samples with lengths verified by direct sequencing. To evaluate the reproducibility of this method, 353 results were re-run and the concordance rate was 98.3%. When re-examining the discordant samples, we found that the errors were of human nature in misinterpreting the technically sound genotype results. This will not be an issue in the future when the samples will also be automatically evaluated by the GeneMapper software.
The IGF1 genotype was not associated with TNM staging, grade or hormone receptor status in either age group, nor was it associated with mode of detection among the patients whose cancer was diagnosed between the ages of 45 and 74 years. As early-onset breast cancer in itself is associated with a poorer prognosis (Sidoni et al, 2003), it may be advantageous to initiate breast cancer screening among multiparous women with the IGF1-19/-19 genotype at an earlier age. Alternative screening methods may be needed in this age group as mammography shows poor sensitivity in young women (Houssami et al, 2003). Conversely, among patients with fewer than two children, the IGF1-19/-19 genotype was associated with a later age of breast cancer onset, and these women may initiate screening at a later age. If confirmed, our data suggest that IGF1 genotyping should be considered in all women with multiple pregnancies to determine the optimal start age for breast cancer screening.
Conclusion
Our findings suggest that the combination of IGF1-19/-19 genotype and multiparity is associated with an early age at breast cancer diagnosis. These findings may explain why the proportion of early-onset breast cancers is higher in the developing than in the developed world. Further studies are warranted to confirm our findings. If confirmed, the IGF1-19/-19 genotype may help identify a subgroup of patients for whom early breast cancer screening is warranted as well as a subgroup of patients for whom the onset of regular screening can safely be postponed.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Amend K, Hicks D, Ambrosone CB (2006) Breast cancer in african-american women: differences in tumor biology from European-american women. Cancer Res 66: 8327–8330
Beral V, Reeves G (1993) Childbearing, oral contraceptive use, and breast cancer. Lancet 341: 1102
Cleveland RJ, Gammon MD, Edmiston SN, Teitelbaum SL, Britton JA, Terry MB, Eng SM, Neugut AI, Santella RM, Conway K (2006) IGF1 CA repeat polymorphisms, lifestyle factors and breast cancer risk in the Long Island Breast Cancer Study Project. Carcinogenesis 27: 758–765
DeLellis K, Ingles S, Kolonel L, McKean-Cowdin R, Henderson B, Stanczyk F, Probst-Hensch NM (2003) IGF1 genotype, mean plasma level and breast cancer risk in the Hawaii/Los Angeles multiethnic cohort. Br J Cancer 88: 277–282
Fletcher O, Gibson L, Johnson N, Altmann DR, Holly JM, Ashworth A, Peto J, Silva Idos S (2005) Polymorphisms and circulating levels in the insulin-like growth factor system and risk of breast cancer: a systematic review. Cancer Epidemiol Biomarkers Prev 14: 2–19
Hall IJ, Moorman PG, Millikan RC, Newman B (2005) Comparative analysis of breast cancer risk factors among African-American women and White women. Am J Epidemiol 161: 40–51
Han W, Kim SW, Park IA, Kang D, Kim SW, Youn YK, Oh SK, Choe KJ, Noh DY (2004) Young age: an independent risk factor for disease-free survival in women with operable breast cancer. BMC Cancer 4: 82
Hankinson SE, Willett WC, Colditz GA, Hunter DJ, Michaud DS, Deroo B, Rosner B, Speizer FE, Pollak M (1998) Circulating concentrations of insulin-like growth factor-I and risk of breast cancer. Lancet 351: 1393–1396
Harrela M, Koistinen H, Kaprio J, Lehtovirta M, Tuomilehto J, Eriksson J, Toivanen L, Koskenvuo M, Leinonen P, Koistinen R, Seppala M (1996) Genetic and environmental components of interindividual variation in circulating levels of IGF-I, IGF-II, IGFBP-1, and IGFBP-3. J Clin Invest 98: 2612–2615
Holmes MD, Pollak MN, Hankinson SE (2002) Lifestyle correlates of plasma insulin-like growth factor I and insulin-like growth factor binding protein 3 concentrations. Cancer Epidemiol Biomarkers Prev 11: 862–867
Houssami N, Irwig L, Simpson JM, McKessar M, Blome S, Noakes J (2003) Sydney Breast Imaging Accuracy Study: Comparative sensitivity and specificity of mammography and sonography in young women with symptoms. AJR Am J Roentgenol 180: 935–940
Jernström H, Chu W, Vesprini D, Tao Y, Majeed N, Deal C, Pollak M, Narod SA (2001a) Genetic factors related to racial variation in plasma levels of insulin-like growth factor-1: implications for premenopausal breast cancer risk. Mol Genet Metab 72: 144–154
Jernström H, Deal C, Wilkin F, Chu W, Tao Y, Majeed N, Hudson T, Narod SA, Pollak M (2001b) Genetic and nongenetic factors associated with variation of plasma levels of insulin-like growth factor-I and insulin-like growth factor-binding protein-3 in healthy premenopausal women. Cancer Epidemiol Biomarkers Prev 10: 377–384
Jernström H, Lerman C, Ghadirian P, Lynch HT, Weber B, Garber J, Daly M, Olopade OI, Foulkes WD, Warner E, Brunet JS, Narod SA (1999) Pregnancy and risk of early breast cancer in carriers of BRCA1 and BRCA2. Lancet 354: 1846–1850
Jernström H, Sandberg T, Bågeman E, Borg Å, Olsson H (2005) Insulin-like growth factor-1 (IGF1) genotype predicts breast volume after pregnancy and hormonal contraception and is associated with circulating IGF-1 levels: implications for risk of early-onset breast cancer in young women from hereditary breast cancer families. Br J Cancer 92: 857–866
Kelsey JL, Gammon MD, John EM (1993) Reproductive factors and breast cancer. Epidemiol Rev 15: 36–47
McCredie MR, Dite GS, Giles GG, Hopper JL (1998) Breast cancer in Australian women under the age of 40. Cancer Causes Control 9: 189–198
Palmer JR, Wise LA, Horton NJ, Adams-Campbell LL, Rosenberg L (2003) Dual effect of parity on breast cancer risk in African-American women. J Natl Cancer Inst 95: 478–483
Parkin DM, Bray FI, Devesa SS (2001) Cancer burden in the year 2000. The global picture. Eur J Cancer 37 Suppl 8: S4–S66
Parkin DM, Whelan SL, Ferlay J, Teppo L, Thomas DB (eds) (2002) Cancer incidence in five continents. Volume VIII. IARC Sci Publ 155: 1–781
Peyrat JP, Bonneterre J, Hecquet B, Vennin P, Louchez MM, Fournier C, Lefebvre J, Demaille A (1993) Plasma insulin-like growth factor-1 (IGF-1) concentrations in human breast cancer. Eur J Cancer 29A: 492–497
Renehan AG, Zwahlen M, Minder C, O’wyer ST, Shalet SM, Egger M (2004) Insulin-like growth factor (IGF)-I, IGF binding protein-3, and cancer risk: systematic review and meta-regression analysis. Lancet 363: 1346–1353
Ries LAG, Harkins D, Krapcho M, Mariotto A, Miller BA, Feuer EJ, Clegg L, Eisner MP, Horner MJ, Howlander N, Hayat M, Hankey BF, Edwards Bke (2006) SEER Cancer Statistics Review, 1975–2003. Bethesda, MD: National Cancer Insitute
Ringberg A, Bågeman E, Rose C, Ingvar C, Jernström H (2006) Of cup and bra size: reply to a prospective study of breast size and premenopausal breast cancer incidence. Int J Cancer 119: 2242–2243
Rodriguez G, Bilbao C, Ramirez R, Falcon O, Leon L, Chirino R, Falcon Jr O, Diaz BP, Rivero JF, Perucho M, Diaz-Chico BN, Diaz-Chico JC (2006) Alleles with short CAG and GGN repeats in the androgen receptor gene are associated with benign endometrial cancer. Int J Cancer 118: 1420–1425
Rosen CJ, Kurland ES, Vereault D, Adler RA, Rackoff PJ, Craig WY, Witte S, Rogers J, Bilezikian JP (1998) Association between serum insulin growth factor-I (IGF-I) and a simple sequence repeat in IGF-I gene: implications for genetic studies of bone mineral density. J Clin Endocrinol Metab 83: 2286–2290
Rotwein P, Pollock KM, Didier DK, Krivi GG (1986) Organization and sequence of the human insulin-like growth factor I gene. Alternative RNA processing produces two insulin-like growth factor I precursor peptides. J Biol Chem 261: 4828–4832
Schernhammer ES, Holly JM, Pollak MN, Hankinson SE (2005) Circulating levels of insulin-like growth factors, their binding proteins, and breast cancer risk. Cancer Epidemiol Biomarkers Prev 14: 699–704
Schildkraut JM, Demark-Wahnefried W, Wenham RM, Grubber J, Jeffreys AS, Grambow SC, Marks JR, Moorman PG, Hoyo C, Ali S, Walther PJ (2005) IGF1 (CA)19 repeat and IGFBP3 -202 A/C genotypes and the risk of prostate cancer in Black and White men. Cancer Epidemiol Biomarkers Prev 14: 403–408
Sidoni A, Cavaliere A, Bellezza G, Scheibel M, Bucciarelli E (2003) Breast cancer in young women: clinicopathological features and biological specificity. Breast 12: 247–250
Sugumar A, Liu YC, Xia Q, Koh YS, Matsuo K (2004) Insulin-like growth factor (IGF)-I and IGF-binding protein 3 and the risk of premenopausal breast cancer: a meta-analysis of literature. Int J Cancer 111: 293–297
Tran N, Bharaj BS, Diamandis EP, Smith M, Li BD, Yu H (2004) Short tandem repeat polymorphism and cancer risk: influence of laboratory analysis on epidemiologic findings. Cancer Epidemiol Biomarkers Prev 13: 2133–2140
Vaessen N, Heutink P, Janssen JA, Witteman JC, Testers L, Hofman A, Lamberts SW, Oostra BA, Pols HA, van Duijn CM (2001) A polymorphism in the gene for IGF-I: functional properties and risk for type 2 diabetes and myocardial infarction. Diabetes 50: 637–642
Wen W, Gao YT, Shu XO, Yu H, Cai Q, Smith JR, Zheng W (2005) Insulin-like growth factor-I gene polymorphism and breast cancer risk in Chinese women. Int J Cancer 113: 307–311
Verhaeghe J, Loos R, Vlietinck R, Herck EV, van Bree R, Schutter AM (1996) C-peptide, insulin-like growth factors I and II, and insulin-like growth factor binding protein-1 in cord serum of twins: genetic versus environmental regulation. Am J Obstet Gynecol 175: 1180–1188
Acknowledgements
We thank the participants of this study. We also thank our research nurses for data collection; Annette Möller, Karin Henriksson, Anna Weddig and Linda Hansson. Furthermore, we would acknowledge Maj-Britt Hedenblad and Sol-Britt Olsson for data entry and DNA-extraction. We thank Dr Pär-Ola Bendahl, biostatistician, for assistance with the statistics and Dr Eric T Dryver for proofreading the manuscript. This study was supported by grants from The Swedish Cancer Society, The Mrs Berta Kamprad Cancer Foundation, Lund University Hospital Fund, The Crafoord Foundation, The G. Nilsson Foundation, The Swedish Research Council (K2001-27GX-14120-01A), The GA's Donation for Breast Cancer Research, the 1049 Fund at the Lund Oncology Clinic, the Region Skåne ALF, and an unrestricted grant by Novartis. Helena Jernström's position was supported by the Swedish Research Council (K2002-27GP-14104-02B) and the Medical Faculty of Lund University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Bågeman, E., Ingvar, C., Rose, C. et al. Absence of the common Insulin-like growth factor-1 19-repeat allele is associated with early age at breast cancer diagnosis in multiparous women. Br J Cancer 96, 712–717 (2007). https://doi.org/10.1038/sj.bjc.6603632
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6603632