Abstract
Multiple myeloma is a B cell malignancy. Recently, it has been demonstrated that bone marrow samples of patients with multiple myeloma display an enhanced angiogenesis. The mechanisms involved seem to be multiple and complex. We here demonstrate that the murine 5T multiple myeloma models are able to induce angiogenesis in vitro by using a rat aortic ring assay and in vivo by determining the microvessel density. The rat aortic rings cultured in 5T multiple myeloma conditioned medium exhibit a higher number of longer and more branched microvessels than the rings cultured in control medium. In bone marrow samples from 5T multiple myeloma diseased mice, a statistically significant increase of the microvessel density was observed when compared to bone marrow samples from age-matched controls. The angiogenic phenotype of both 5T multiple myeloma cells could be related, at least in part, to their capacity to produce vascular endothelial growth factor. These data clearly demonstrate that the 5T multiple myeloma models are good models to study angiogenesis in multiple myeloma and will allow to unravel the mechanisms of neovascularisation, as well as to test new putative inhibitors of angiogenesis.
Similar content being viewed by others
Main
Multiple myeloma (MM) is a B cell neoplasm with three major characteristics: the expansion of malignant plasma cells mainly localised in the bone marrow, the presence of a monoclonal serum immunoglobulin and the activation of osteoclasts leading to osteolysis. The disease occurs mainly at older age and is still incurable in spite of the progress in treatment. Recently, it has been demonstrated that angiogenesis or neovascularisation is also involved in MM disease and its extent, as measured by microvessel density (MVD) of the bone marrow, correlated well with the activity of MM (Vacca et al, 1994). The human U266 MM cell line and bone marrow plasma cells from patients with active MM display a strong angiogenic potential (Vacca et al, 1998, 1999; Laroche et al, 2001). Moreover, bone marrow MVD also seems to be a prognostic factor of survival in MM patients (Sezer et al, 2000).
Angiogenesis is a complex process in which new blood vessels develop from pre-existing ones. It is controlled by a balance between pro-angiogenic and anti-angiogenic factors (Talks and Harris, 2000). While this balance is highly regulated in physiological circumstances such as reproduction, development and wound repair, an imbalance is associated with a variety of disorders including cancers (Folkman and Shing, 1992; Compagni and Christofori, 2000). Tumour growth and metastasis are dependent on new blood vessel formation (Kim et al, 1993; Folkman, 1995). The vessels provide nutrients and a route by which tumour cells exit the primary site and enter the blood stream (Zetter, 1998). Among the various angiogenic stimulators, an important factor involved in normal and pathological angiogenesis is vascular endothelial growth factor (VEGF) (Ferrara, 1999b; Carmeliet and Collen, 2000) which is able to stimulate vascular permeability and is an endothelial cell specific mitogen (Connolly et al, 1989). By alternative splicing of a single gene, several VEGF isoforms can be produced (Shima et al, 1996). VEGF is secreted by many tumour cell lines in vitro and its mRNA is upregulated in the vast majority of human tumours (Ferrara, 1999a).
To investigate the importance and the underlying mechanisms of angiogenesis in the development of MM, an in vivo model is required. Therefore we selected the murine 5TMM models. We have used two MM models, which initially originated spontaneously in ageing C57BL/KaLwRij mice (Radl et al, 1979). Tumour cells from diseased mice were isolated from the bone marrow and transplanted into young syngeneic recipients by intravenous injection. By this way, several 5TMM models were developed (Radl et al, 1979) with similar characteristics as the human disease: the disease occurs spontaneously at older age, the MM cells are localised in the bone marrow, tumour load can be assessed by paraproteinaemia, osteolytic lesions develop and the molecular mechanisms are like in humans (Vanderkerken et al, 1996; Radl et al, 1988; Asosingh et al, 2000). As previously reported, the 5T2MM model more closely represents human MM, characterized by a moderate progressive course of the disease and induction of osteolytic lesions, while the 5T33MM model represents an aggressive, rapidly progressive variant (Vanderkerken et al, 1997; Radl et al, 1988). Therefore, the 5T2MM and 5T33MM models are good in vivo murine models of myeloma which mimic different aspects of the human disease (Asosingh et al, 2000).
The aim of this study was to study the ability of 5T2MM and 5T33MM cells to induce angiogenesis in the bone marrow, as do human MM cells. The angiogenic potential of 5T2MM and 5T33MM cells was assessed both in vitro, in the aortic ring assay, and in vivo, by determining the microvessel density in bone marrow samples of control mice and 5T2MM and 5T33MM-diseased mice. The understanding of neovascularization mechanisms in MM may provide further benefits for both diagnosis and therapy.
Materials and methods
Mice
C57BL/KaLwRij mice were purchased from Harlan CPB (Horst, The Netherlands). They were housed and treated following the conditions approved by the Ethical Committee for Animal Experiments, VUB (License no. LA1230281). The animal ethics meet the standards required by the UKCCCR Guidelines (UKCCCR, 1998).
5TMM models
The in vivo growing 5T2MM and 5T33MM originated initially spontaneous in ageing C57BL/KaLwRij mice and have since been propagated into young syngeneic recipients by intravenous transfer of the diseased bone marrow (Radl et al, 1979). The tumour take was followed up by protein electrophoresis of the serum samples (Vanderkerken et al, 1997). Mice were killed when a serum paraprotein concentration of 10 mg ml−1 was reached. The MM cells in the bone marrow were isolated and purified as previously described (Vanderkerken et al, 1999). Bone marrow was flushed from the hind legs. The isolated cells were suspended in RPMI-1640 medium (Gibco, Life Technologies, Gent, Belgium) supplemented with penicillin/streptomycin, glutamine, MEM and sodium pyruvate (Gibco). After washing, mononuclear cells were isolated by Lympholyte M (Cedarlane, Hornby, Canada) gradient centrifugation at 1000 g for 20 min. Subsequently, the cells were further purified on a Percoll (Pharmacia, Uppsala, Sweden) 60% (for 5T2) and 70% (for 5T8); gradient centrifugation at 450 g for 25 min. Cells were stained with anti-idiotype antibodies and purity was assessed by flow cytometry (Vanderkerken et al, 1997). The 5TMM cells (1 × 106 cells ml−1) were cultured in serum-free MCDB131 medium (an optimised medium for microvascular endothelial cell growth) or were immediately snap-frozen for RNA isolation. After 48 h, conditioned media were collected.
Assessment of angiogenesis by rat aortic ring assay
Angiogenesis was studied by culturing aortic explants in three-dimensional matrix gels according to the procedure of Nicosia and Ottinetti (1990). Thoracic aortas removed from 8- to 12-week-old male Fisher-344 rats were immediately transferred to a culture dish containing cold serum-free Minimum Essential Medium (MEM, Life Technologies). The periaortic fibroadipose tissue was carefully removed with fine microdissecting forceps and iridectomy scissors paying special attention not to damage the aortic wall. Aortic rings (1 mm-long) (approximately 20 per aorta) were sectioned and extensively rinsed in five consecutive washes of MEM medium. Ring-shaped explants of rat aorta were then embedded in gels of rat tail interstitial collagen (1.5 mg ml−1) as previously described by Montesano et al (1983). The final collagen solution was obtained by mixing 7.5 volumes of collagen (2 mg ml−1) (Collagen R, Serva, Heidelberg, Germany) with one volume of 10 × MEM, 1.5 volumes of NaHCO3 (15.6 mg ml−1) and approximately 0.1 volume NaOH (1 M) to adjust the pH to 7.4. The collagen gel cultures were first prepared in cylindrical agarose wells (Nicosia and Ottinetti, 1990) and kept in triplicates at 37°C in 100 mm diameter Petri dishes (bacteriological polystyrene, Falcon, Becton Dickinson, Lincoln Park, NJ, USA). Each dish contained either 30 ml of MCDB131 (Life Technologies) supplemented with 25 mM NaHCO3, 1% glutamine, 100 U ml−1 penicillin and 100 μg ml−1 streptomycin or MCDB131 medium conditioned by 5T2MM or 5T33MM cells. As positive control human recombinant VEGF165 (20 ng ml−1) was added to the medium. The cultures were kept at 37°C in a humidified environment for 1 week and examined by phase contrast microscopy with an Olympus microscope at the appropriate magnification. Image analysis was performed on a WorkStation Sun SPARC30, using the software ‘Visilog5.0’ from Noesis. We used an improved computer-assisted image analysis (Blacher et al, 2001) which allows automatic measurements of the geometrical and morphological parameters. After generation of binary image, the following automatic measurements were performed: the quantification of microvessels according to the vascular complexity, i.e. the determination of the microvessel distribution given by the total number of intersections of microvessels (Ni) in function to the distance to the aortic ring. The Ni value at the first step of the grid gives the number of microvessels (Nv). The grid step at which the last Ni≠0 is measured gives the maximal microvessel length (Lmax). The sum of additional intersections detected at each step of the grid gives the total number of branching (Nb). Statistical parameters have been calculated to describe the distribution of Ni. The mean distribution (D) and the standard deviation correspond to the distance from the explant at which Ni reaches its mean value. The mode (d) corresponds to the distance from the explant at which most cells migrated. The skewness (sk) describes the symmetry of the distribution and the kurtosis (k) describes how flat or peaked the distribution is.
Assessment of angiogenesis by bone marrow microvessel density
Long bones, vertebrae and ribs of age-matched control, 5T2MM and 5T33MM diseased C57BL/KaLwRij mice were fixed in zinc fixative (0.1 M Tris buffer, pH 7.4, 3 mM calcium acetate, 0.027 M zinc acetate and 0.037 M zinc chloride) for 24 h and decalcified in FE10 (0.27 M EDTA, 0.3 M NaOH, 4% formalin) for 3 days. The material was embedded in paraffin and 5 μm longitudinal sections throughout the whole bones were made with a rotation microtome (Microm HM335, Germany). One slide was stained with haematoxylin and eosin, the consecutive slide and two additional slides with a 50 μm distance from each other were immunostained for CD31 in order to visualise the blood vessels and sinusoids in the bone marrow.
For immunostaining, the endogenous peroxydase was quenched by incubating the slides with a 50 ml methanol, 0.5 ml H2O2 (30%) solution for 30 min. Trypsinisation (Trypsin 109827, Boehringer Mannheim, IN, USA) for 20 min, at 37°C was used for antigen retrieval. The slides were preincubated with normal goat serum for 30 min. The primary antibody (PECAM-1, Becton Dickinson PharMingen, LA, USA) was incubated overnight at 4°C at a dilution of 1 out of 100. As secondary antibody, a biotin-conjugated goat anti-rat specific polyclonal immunoglobulin (554014, Becton Dickinson PharMingen, LA, USA) was used at a dilution of 1 out of 100. The TSA (Tyramide Signal Amplification) (NEN Life Science Products, Boston, MA, USA) was used to enhance the signal intensity. Chromogenic visualisation was accomplished through the use of a streptavidin-horseradish peroxydase conjugate, followed by diaminobenzidin.
The haematoxylin and eosin sections of the bone marrow were screened for the presence of myeloma cells, with a light microscope (Axiolab, CarlZeiss, Germany). For quantification of angiogenesis, areas with the highest density of blood vessels (hot spot) were selected on sections immunostained for CD31. The number of blood vessels was counted per 0.22 mm2 using a microscope eyepiece graticule (× 10 ocular and × 20 objective). The microvessel density was independently evaluated by two observers.
Detection of mRNA for VEGF
Total RNA from 5 × 106 purified 5T2MM and 5T33MM cells was isolated using the SV total RNA isolation system (Promega Corporation, Madison WI, USA) according to the manufacturer's instructions. The concentration and purity of RNA was determined by spectrophotometric measurement (Gene Quant II, Pharmacia Biotech, Cambridge, UK).
One to 5 μg of total RNA was converted into cDNA by the superscript first-strand synthesis system (Gibco, Life Technologies) using random hexamers as primers. The cDNA was used as template for PCR in a 25 μl reaction containing: PCR buffer, 2 mM magnesium chloride, 0.2 mM dNTP, 0.5 μM of each primer and 0.625 U Taq polymerase. The sense and antisense primers used for the amplification of the known splice variants of mouse VEGF are 5′-CCTGGTGGACATCTTCCAGGAGTA-3′ and 5′-CTCACCGCCTCGGCTTGTCACA-3′, respectively. Forty PCR cycles were performed preceded by a denaturation step at 95°C for 2 min followed by 20 s at 94°C, 20 s at 66°C and 30 s at 72°C. The predicted size of the amplified products was 479 bp for VEGF188, 407 bp for VEGF164 and 275 bp for VEGF120 (Hajitou et al, 2001). PCR products were analysed by electrophoresis on a 1.5% low-melting agarose gel. The PCR samples were cloned into the pCRII vector (Invitrogen, San Diego, CA, USA) and sequenced with vector-specific primers using the Sequenase version 2.0 DNA sequencing kit (USB Corporation, Cleveland, OH, USA).
Detection of VEGF protein
The secretion of VEGF by the 5TMM cell lines in serum-free conditioned media (48 h) was quantified by an enzyme-linked immunosorbent assay (ELISA, R&Dsystems, Wiesbaden, Germany) according to the manufacturer's instructions. Each sample was performed in duplicate and two independent experiments were done.
Statistical analysis
For statistical analysis the unpaired student's t-test was used. P-values<0.05 were considered as significant.
Results
5TMM cells induce angiogenesis in vitro
The angiogenic potential of the 5T2MM and 5T33MM cells was tested in the in vitro rat aortic ring assay. Aortic rings were cultured in the presence of serum-free MCDB131 medium or MCDB131 medium conditioned by 5T2MM or 5T33MM cells. As positive control human recombinant VEGF165 was added to the medium. After 1 week, the rings cultured in the presence of 5T2MM (Figure 1C) or 5T33MM (Figure 1D) conditioned media (Figure 1A) exhibit more capillaries than those cultured in control medium. Interestingly, in the presence of 5T33MM conditioned medium, the total number of microvessels (Nv=38) was two-fold higher than in control conditions (Nv=20). The addition of 5T2MM conditioned medium induced a less pronounced, but still statistically significant effect on microvessel outgrowth (Nv=24). In the presence of both media conditioned by myeloma cells, the maximal microvessel length was 20% enhanced (Lmax=1.0 mm vs 0.85 mm in control conditions). The vascular network complexity was more precisely studied by statistical analysis according to the methodology described by Blacher et al (2001) (Figure 2). The broadness of the histogram obtained with both conditioned media indicates that microvessel networks were more branched than in control conditions. Indeed, the Ni distribution of 5T2MM and 5T33MM condition had a peak (k ≅ 0) nearby the aortic ring and decreased less steeply (sk=0.65 for 5T2MM and 0.72 for 5T33MM) than in control conditions (sk=1.10). This indicates that myeloma conditioned media enhanced the vascular network complexity which are then characterized by more branched vessels (Figure 2A).
Photomicrographs of rat aortic rings cultured in collagen gels in the presence of serum-free medium (A), serum-free medium supplemented with 20 ng ml−1 rVEGF165 (B), 5T2MM conditioned medium (C), 5T33MM conditioned medium (D). Photomicrographs from one experiment, representative of there, are shown.
Determination of the number of microvessel intersections (Ni) in function of the distance to the aortic ring. Histograms have been constructed for control (A), VEGF (B), 5T2MM (C), 5T33MM (D). Statistical parameters are indicated for each histogram. D: distance from the explant at which Ni reaches its mean value; d: distance from the explant at which most cells migrated; sk>0 indicates that most cells migrate at distances lower than D; k: sharpness of the cell distribution around d.
Assessment of microvessel density in the bone marrow
Using an eye-piece graticule the area with the highest density of blood vessels was selected, discarding the area near the growth plate since this area, as distinctly observed in the non-invaded marrow, has physiologically a higher blood vessel density. When compared to normal bone marrow, the sinusoids were more compressed (probably a mechanical effect) in the tumour-invaded marrows. When dilated sinusoids were observed in invaded marrows, they were massively filled with tumour cells. Since the lumen of the sinusoids belongs to the extramedullary compartment this finding confirms the presence of circulating tumour cells. As previously observed (Vanderkerken et al, 1997), the 5T33MM cells were more blastic (centrally located nuclei, distinctive nucleoli) compared to 5T2MM cells in which the plasmacytic differentiation was more readily observed (eccentric location of the nucleus, clumped chromatin).
In both 5T2MM and 5T33MM invaded bone marrows, we observed a statistically significant increase in blood vessel density as compared to non-invaded controls (Figures 3 and 4). The hot spots coincided with sites of massive invasion.
Expression and secretion of VEGF by the 5T2MM and 5T33MM cells
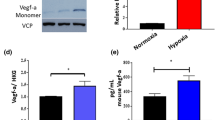
Since VEGF is one of the most important pro-angiogenic factors, we investigated whether 5TMM cells secrete VEGF. The expression of VEGF transcripts was determined by RT–PCR (Figure 5). The positive control (murine kidney) expresses the three VEGF isoforms. The 5T2MM and 5T33MM cells express VEGF transcripts corresponding to the two lower molecular weight isoforms, VEGF164 and VEGF120. This was confirmed by sequencing the two transcripts (Claffey et al, 1992; Shima et al, 1996). The secretion of VEGF by both cell lines was further assessed by ELISA. The concentration of VEGF in culture supernatants of the 5T2MM and 5T33MM cells ranged between 100 and 160 pg ml−1.
Analysis of VEGF expression in the 5TMM cells by RT–PCR. cDNA from purified myeloma cells were subjected to specific VEGF amplification. The amplification products were detected as a band of 275 bp for VEGF120, 407 bp for VEGF164 and 479 bp for VEGF188. Murine kidney was used as positive control for the three isoforms of VEGF. This experiment is representative for two experiments.
Discussion
Recently, it was postulated that neovascularization may play an important role in haematological malignancies (Bertolini et al, 2000; Mangi and Newland, 2000). It was suggested that, like in solid tumours, the progression of MM takes place in two phases. Nonactive MM and monoclonal gammopathies of undetermined significance (MGUS) might represent the prevascular phase, whereas active MM might correspond to the vascular phase (Vacca et al, 1994). Angiogenesis is a complex multistep process characterized first by the stimulation of endothelial cells by angiogenic factors, and then by the production of proteolytic enzymes, such as matrix metalloproteinases (MMPs) and urokinase plasminogen activator (uPA). These enzymes are involved in the degradation of the basement membrane and the extracellular matrix, and promote the migration and proliferation of endothelial cells to form a new blood vessel (Moses, 1997; Zetter, 1998). In human MM, VEGF and bFGF are secreted by tumour cells and the concentrations of these angiogenic factors are higher in more advanced disease (Vacca et al, 1998; Bellamy et al, 1999; Dankbar et al, 2000; Di Raimondo et al, 2000; Sezer et al, 2000; Van Riet et al, 2000). MM cells also secrete MMPs (Barillé et al, 1997, 1999; Vanderkerken et al, 2000) and uPA (Hjertner et al, 2000) and their conditioned medium is able to stimulate the proliferation and migration of endothelial cells (Vacca et al, 1998, 1999). These findings suggest that the MM cells can induce angiogenesis. The ability of medium conditioned by plasma cells from MM patients to stimulate neovascularisation in the chorioallantoic membrane assay supports this hypothesis (Vacca et al, 1998, 1999). These recent insights disclose new possible therapeutic targets. Interestingly, the anti-angiogenic drug, thalidomide, has been reported to be effective in patients with refractory MM (D'Amato et al, 1994; Singhal et al, 1999). The development of drugs acting on different levels of angiogenesis can thus be considered as a promising pathway. This research inevitably requires good in vitro and in vivo experimental models. In the present study, we demonstrate that the 5T2MM and 5T33MM murine multiple myeloma models are interesting in vivo models of angiogenesis. Both cell lines are able to induce in vitro and in vivo angiogenesis. This could be, at least in part, related to their capacity to produce VEGF as demonstrated by RT–PCR and ELISA. These data indicate that MM cells have, at least, the potential to induce the first step of neovascularisation, namely the stimulation of endothelial cells.
The ability of both 5T2MM and 5T33MM cells to induce in vitro angiogenesis was measured in the rat aortic ring assay. The microvessels branched, anastomosed, developed lumina and formed complex networks as initially described by Nicosia and Ottinetti (1990). Compared to control condition, a more complex microvessel outgrowth occurred when the rings were cultured in the presence of media conditioned by 5T2MM or 5T33MM cells. However, the network of blood vessels was less complex than the one induced by recombinant VEGF, as assessed by our original method of vessel quantification based on computer-assisted image analysis (Blacher et al, 2001). In the rat aortic ring assay a dose-dependent effect of VEGF on microvascular network complexity was evidenced by Blacher et al (2001). The much lower VEGF concentration in 5T2MM and 5T33MM conditioned media than the concentration of recombinant VEGF added to the medium of the positive control can explain the difference in network complexity. The MVD in the bone marrow from diseased and control animals was compared in order to investigate the in vivo angiogenesis. To achieve a reliable counting of the bone marrow blood vessels several factors had to be fulfilled: representative samples, optimal fixation and decalcification, thin sections and a sensitive immunohistochemical staining of the blood vessels. Whole bones were collected at different sites of the skeleton; fixation and decalcification methods were optimised. Immunostaining for the CD31 antigen of the blood vessels in the BM of 5T2MM and 5T33MM diseased mice revealed a significantly higher MVD in the diseased animals compared to the control animals. The higher angiogenic potential of the 5T33MM model can be due to the more aggressive growth of the 5T33MM cells compared to the 5T2MM cells (Vanderkerken et al, 1997).
Based on their in vivo and in vitro angiogenic potential and their capacity to produce VEGF, we conclude that the two 5TMM models are suitable for the study of multiple myeloma neovascularization. These models can now be used to unravel the multistep process of neovascularization and to test new angiogenic inhibitors.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Asosingh K, Radl J, Van Riet I, Van Camp B, Vanderkerken K (2000) The 5TMM series: a useful in vivo mouse model of human multiple myeloma. Hematol J 1: 351–356
Barillé S, Akhoundi C, Collette M, Mellerin MP, Rapp MJ, Harousseau JL, Bataille R, Amiot M (1997) Metalloproteinases in multiple myeloma: production of matrix metalloproteinase-9 (MMP-9), activation of proMMP-2, and induction of MMP-1 by myeloma cells. Blood 90: 1649–1655
Barillé S, Bataille R, Rapp MJ, Harousseau JL, Amiot M (1999) Production of metalloproteinase-7 (matrilysin) by human myeloma cells and its potential involvement in metalloproteinase-2 activation. J Immunol 163: 5723–5728
Bellamy WT, Richter L, Frutiger Y, Grogan TM (1999) Expression of vascular endothelial growth factor and its receptors in hematopoietic malignancies. Cancer Res 59: 728–733
Bertolini F, Mancuso P, Gobbi A, Pruneri G (2000) The thin red line: angiogenesis in normal and malignant hematopoiesis. Exp Hematol 28: 993–1000
Blacher S, Devy L, Burbridge MF, Roland G, Tucker G, Noël A, Foidart JM (2001) Improved quantification of angiogenesis in the rat aortic ring assay. Angiogenesis 4: 133–142,
Carmeliet P, Collen D (2000) Molecular basis of angiogenesis. Role of VEGF and VE-cadherin. Ann NY Acad Sci 902: 249–262
Claffey KP, Wilkison WO, Spiegelman BM (1992) Vascular endothelial growth factor: regulation by cell differentiation and activated second messenger pathways. J Biol Chem 267: 16317–16322
Compagni A, Christofori G (2000) Recent advances in research on multistage tumorigenesis. Br J Cancer 83: 1–5
Connolly DT, Heuvelman DM, Nelson R, Olander JV, Eppley BL, Delfino JJ, Siegel NR, Leimgruber RM, Feder J (1989) Tumor vascular permeability factor stimulates endothelial cell growth and angiogenesis. J Clin Invest 84: 1470–1478
D'Amato RJ, Loughnan MS, Flynn E, Folkman J (1994) Thalidomide is an inhibitor of angiogenesis. Proc Natl Acad Sci USA 91: 4082–4085
Dankbar B, Padro T, Leo R, Feldmann B, Kropff M, Mesters RM, Serve H, Berdel WE, Kienast J (2000) Vascular endothelial growth factor and interleukin-6 in paracrine tumor- stromal cell interactions in multiple myeloma. Blood 95: 2630–2636
Di Raimondo F, Azzaro MP, Palumbo G, Bagnato S, Giustolisi G, Floridia P, Sortino G, Giustolisi R (2000) Angiogenic factors in multiple myeloma: higher levels in bone marrow than in peripheral blood. Haematologica 85: 800–805
Ferrara N (1999a) Molecular and biological properties of vascular endothelial growth factor. J Mol Med 77: 527–543
Ferrara N (1999b) Role of vascular endothelial growth factor in the regulation of angiogenesis. Kidney Int 56: 794–814
Folkman J (1995) Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1: 27–31
Folkman J, Shing Y (1992) Angiogenesis. J Biol Chem 267: 10931–10934
Hajitou A, Sounni NE, Devy L, Grignet-Debrus C, Lewalle JM, Li H, Deroanne CF, Lu H, Colige A, Nusgens BV, Frankenne F, Maron A, Yeh P, Perricaudet M, Chang Y, Soria C, Calberg-Bacq CM, Foidart JM, Noel A (2001) Down-regulation of vascular endothelial growth factor by tissue inhibitor of metalloproteinase-2: effect on in vivo mammary tumor growth and angiogenesis. Cancer Res 61: 3450–3457
Hjertner O, Qvigstad G, Hjorth-Hansen H, Seidel C, Woodliff J, Epstein J, Waage A, Sundan A, Borset M (2000) Expression of urokinase plasminogen activator and the urokinase plasminogen activator receptor in myeloma cells. Br J Haematol 109: 815–822
Kim KJ, Li B, Winer J, Armanini M, Gillett N, Phillips HS, Ferrara N (1993) Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 362: 841–844
Laroche M, Brousset P, Ludot I, Mazieres B, Thiechart M, Attal M (2001) Increased vascularization in myeloma. Eur J Haematol 66: 89–93
Mangi MH, Newland AC (2000) Angiogenesis and angiogenic mediators in haematological malignancies. Br J Haematol 111: 43–51
Montesano R, Orci L, Vassalli P (1983) In vitro rapid organization of endothelial cells into capillary-like networks is promoted by collagen matrices. J Cell Biol 97: 1648–1652
Moses MA (1997) The regulation of neovascularization of matrix metalloproteinases and their inhibitors. Stem Cells 15: 180–189
Nicosia RF, Ottinetti A (1990) Growth of microvessels in serum-free matrix culture of rat aorta. A quantitative assay of angiogenesis in vitro. Lab Invest 63: 115–122
Radl J, De Glopper ED, Schuit HR, Zurcher C (1979) Idiopathic paraproteinemia. II. Transplantation of the paraprotein- producing clone from old to young C57BL/KaLwRij mice. J Immunol 122: 609–613
Radl J, Croese JW, Zurcher C, Van den Enden-Vieveen MH, de Leeuw AM (1988) Animal model of human disease. Multiple myeloma. Am J Pathol 132: 593–597
Sezer O, Niemoller K, Eucker J, Jakob C, Kaufmann O, Zavrski I, Dietel M, Possinger K (2000) Bone marrow microvessel density is a prognostic factor for survival in patients with multiple myeloma. Ann Hematol 79: 574–577
Shima DT, Kuroki M, Deutsch U, Ng YS, Adamis AP, D'Amore PA (1996) The mouse gene for vascular endothelial growth factor. Genomic structure, definition of the transcriptional unit, and characterization of transcriptional and post-transcriptional regulatory sequences. J Biol Chem 271: 3877–3883
Singhal S, Mehta J, Desikan R, Ayers D, Roberson P, Eddlemon P, Munshi N, Anaissie E, Wilson C, Dhodapkar M, Zeddis J, Barlogie B (1999) Antitumor activity of thalidomide in refractory multiple myeloma. N Engl J Med 341: 1565–1571
Talks KL, Harris AL (2000) Current status of antiangiogenic factors. Br J Haematol 109: 477–489
UKCCCR (1998) United Kingdom Co-ordinating Committee on Cancer Research (UKCCCR) Guidelines for the welfare of animals in experimental neoplasia. 2nd edn Br J Cancer 77: 1–10
Vacca A, Ribatti D, Roncali L, Ranieri G, Serio G, Silvestris F, Dammacco F (1994) Bone marrow angiogenesis and progression in multiple myeloma. Br J Haematol 87: 503–508
Vacca A, Ribatti D, Iurlaro M, Albini A, Minischetti M, Bussolino F, Pellegrino A, Ria R, Rusnati M, Presta M, Vincenti V, Persico MG, Dammacco F (1998) Human lymphoblastoid cells produce extracellular matrix-degrading enzymes and induce endothelial cell proliferation, migration, morphogenesis, and angiogenesis. Int J Clin Lab Res 28: 55–68
Vacca A, Ribatti D, Presta M, Minischetti M, Iurlaro M, Ria R, Albini A, Bussolino F, Dammacco F (1999) Bone marrow neovascularization, plasma cell angiogenic potential, and matrix metalloproteinase-2 secretion parallel progression of human multiple myeloma. Blood 93: 3064–3073
Vanderkerken K, Goes E, De Raeve H, Radl J, Van Camp B (1996) Follow-up of bone lesions in an experimental multiple myeloma mouse model: description of an in vivo technique using radiography dedicated for mammography. Br J Cancer 73: 1463–1465
Vanderkerken K, De Raeve H, Goes E, Van Meirvenne S, Radl J, Van Riet I, Thielemans K, Van Camp B (1997) Organ involvement and phenotypic adhesion profile of 5T2 and 5T33 myeloma cells in the C57BL/KaLwRij mouse. Br J Cancer 76: 451–460
Vanderkerken K, Asosingh K, Braet F, Van Riet I, Van Camp B (1999) Insulin-like growth factor-1 acts as a chemoattractant factor for 5T2 multiple myeloma cells. Blood 93: 235–241
Vanderkerken K, Van Valckenborgh E, Bakkus M, Munaut C, Noël A, Asosingh K, Van Riet I, Foidart JM, St-Pierre Y, Van Camp B (2000) Bone marrow microenvironmental-induced upregulation of MMP-9 activity in murine multiple myeloma cells. Clin Exp Metast 9: 766
Van Riet I, Hellebaut L, Castronovo V, Vande Broek I, Arras N, Vanderkerken K, Van Camp B (2000) Expression of the angiogenesis-inducing molecules VEGF and bFGF in multiple myeloma and its regulation by paracrine interactions between tumor cells and stromal bone marrow cells. Blood 96: 361a
Zetter BR (1998) Angiogenesis and tumor metastasis. Annu Rev Med 49: 407–424
Acknowledgements
We thank Angelo Willems for his excellent technical assistance in supporting the mouse model, Guy Roland for the rat aortic ring assay, Frank Rylant for the preparation of the bone marrow sections and Günther Vrolix and Liliane Moeneclaye for the immunostaining. The work was financially supported by the Onderzoeksraad-Vrije Universiteit Brussel (OZR-VUB), Fonds voor Wetenschappelijk Onderzoek-Vlaanderen, Kom op tegen Kanker, ASLK, VIVA and VIS. Karin Vanderkerken is a postdoctoral fellow of the ‘Fonds voor Wetenschappelijk Onderzoek-Vlaanderen’ (FWO-Vl). Agnès Noël is a senior researcher associate, Carine Munaut is a research associate and Laetitia Devy is a postdoctoral researcher, all from the National Fund for Scientific Research (Belgium).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Van Valckenborgh, E., De Raeve, H., Devy, L. et al. Murine 5T multiple myeloma cells induce angiogenesis in vitro and in vivo. Br J Cancer 86, 796–802 (2002). https://doi.org/10.1038/sj.bjc.6600137
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6600137
Keywords
This article is cited by
-
Assessment of bone vascularization and its role in bone remodeling
BoneKEy Reports (2015)
-
The effects of JNJ-26481585, a novel hydroxamate-based histone deacetylase inhibitor, on the development of multiple myeloma in the 5T2MM and 5T33MM murine models
Leukemia (2009)
-
Antitumour and antiangiogenic effects of Aplidin® in the 5TMM syngeneic models of multiple myeloma
British Journal of Cancer (2008)
-
Importance of the bone marrow microenvironment in inducing the angiogenic response in multiple myeloma
Oncogene (2006)
-
Endothelial cell-driven regulation of CD9 or motility-related protein-1 expression in multiple myeloma cells within the murine 5T33MM model and myeloma patients
Leukemia (2006)