Abstract
Data sources
The Medline and Embase databases and hand searches in the journals International Journal of Paediatric Dentistry and European Archives of Paediatric Dentistry.
Study selection
English language cohort and case-control studies.
Data extraction and synthesis
Study selection was carried out independently by two reviewers with data abstraction being conducted by a single reviewer and checked by a second reviewer. Risk of bias was assessed using a modified version of the Newcastle-Ottawa Scale (NOS). Adjusted (aOR) and unadjusted odds ratios (uOR), P-values and 95% confidence intervals (CI) were obtained from the studies. Meta-analysis was not conducted.
Results
Twenty-eight studies were included; 25 reported on MIH, three on hypomineralised second primary molars (HSPM). Nineteen of the studies were of cohort design (six prospective,13 retrospective) and nine were case controls. There was little evidence of an association between the most frequently investigated prenatal factors (smoking, maternal illness, maternal medication, maternal stress) and MIH. Similarly there was little evidence of an association between MIH and perinatal factors such as prematurity, low birth weight, caesarean delivery and birth complications. Early childhood illness, up to three or four years of age, was widely investigated, with six studies reporting a crude association. Associations between antibiotics, anti-asthma medication and breastfeeding were also evaluated. Only three studies looked at HSPM; one study suggested that maternal antibiotic use during pregnancy is unlikely to be associated with HSPM but maternal alcohol intake may be. Another study reported possible associations with a large number of factors, with perinatal factors and neonatal illness being most common, followed by prenatal factors.
Conclusions
Prenatal and perinatal factors are infrequently associated with MIH. However, despite a lack of prospective studies, early childhood illness (in particular fever) appears to be associated with MIH. Further prospective studies that adjust for confounding based on biological principles, as well as genetic and epigenetic studies, are needed because the aetiology is likely to be multifactorial.
Similar content being viewed by others
Commentary
Molar-incisor hypomineralisation (MIH) is an acquired qualitative defect of the enamel of 1–4 first permanent molars with or without maxillary/mandibular permanent incisor involvement.1,2,3 A wide variation in MIH defect prevalence, (2.4-40.2%) has been reported.4 Hypomineralised second primary molars (HSPM) are less common with reported prevalence ranging from 3.6%5 to 4.88%.6 The enamel defects clinically present as demarcated opacities of different colours in the affected teeth, occasionally undergoing post-eruptive breakdown due to soft and porous enamel.7 Occasionally, the term ‘cheese molars’ is used to denote these teeth due to the distinctive clinical appearance.8 These teeth are often of poor long-term prognosis given the inherent structural integrity and increased susceptibility to dental caries,1 however, little research has been carried out at an ultrastructural and biochemical level to confirm this theory.2,4,9 Tentative links between childhood/maternal illness during prenatal, perinatal and postnatal periods and MIH have been suggested, due to the effect hypoxia and pyrexia from these conditions have on ameloblast function,10 however the true aetiology of MIH still remains unclear due to limited prospective evidence available.2,34,7
This systematic review focuses on the aetiology of MIH and HSPM. A robust and thorough search strategy was adopted. Search terms used to search electronic databases Medline and Embase were provided as appendices in addition to the authors' hand searching of the reference lists of selected studies and the grey literature. Appropriately, only observational studies that monitor patients over a period of time, cohort and case-control studies, were included in this review. Only studies reported in English were considered for this review. All exposures were eligible for inclusion in the review. The reviewers report their primary outcome measure as the presence or absence of MIH and/or HSPM for each patient, and therefore included in their review studies that used MIH/HSPM specific criteria or those that reported demarcated opacities in FPM only. Studies that reported on teeth other than FPM, permanent incisors or second primary molars were excluded from this review in addition to those that reported combined demarcated and diffuse opacities as the primary outcome measure.
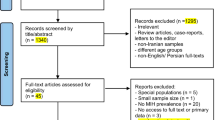
The screening and assessment of the eligibility process was clearly described and displayed in a PRISMA flowchart and was appropriate. Screening was carried out independently by two reviewers with data extraction performed by a single reviewer and checked by a second reviewer. Any disagreements were resolved by consensus with the authors reporting that a total of three amendments were made during this review. A measure of intra-rater and inter-rater reliability was not provided. After 2,254 records were identified (following the removal of duplicates), 28 studies were included, with the authors unable to complete a meta-analysis because too few studies had adjusted for confounding. The level of evidence and risk of bias, assessed using a modified Newcastle-Ottawa scale, were determined for each study. This information was presented in a table which included the country the study was carried out in and number of participants in each study. In total, three case-control and four cohort studies had a high risk of bias. The authors report that the measurement of exposures were mainly obtained from parental reports through interview or questionnaires, introducing a degree of parental recall bias, with only a small number of studies using medical records to obtain information. The authors report a distinct variation between studies in the specificity of criteria used to identify participants affected by MIH and the lack of detail pertaining to how participants were examined, who examined them and whether they were calibrated amongst several studies.
Prenatal exposures, perinatal exposures and early childhood illness factors and their associations with MIH are displayed using a forest plot. Comparing the studies this way may confuse the readers, as a component of any forest plot is the overall effect estimate from a meta-analysis, however, due to high heterogeneity the authors have not carried this out. A simple table may have been more appropriate. Early childhood medication use and breastfeeding are not tabulated due to the small numbers of studies assessing them.
The authors reported little evidence of an association between the most frequently investigated prenatal exposures (maternal smoking, maternal illness and maternal medicine use) and MIH. A crude association between maternal illness and MIH was reported in this review, however, several papers failed to adjust for confounders which reduces the quality of the study findings. The authors did report that one study showed a weak association between maternal stress and MIH (aOR 3.24, 95% CI: 1.33–7.78), however further work is required to demonstrate this association.
Similar to prenatal exposures, little evidence of an association between perinatal exposures (prematurity, low birthweight, cesarean section and birth complications) and MIH was found. Low birthweight and MIH appear weakly associated with only one study, which had adjusted for confounding, reporting higher odds of MIH in their cohort of patients. Following best practice, the authors reported that clarification of the definition of ‘birth mode’ was required to fully assess certain studies, with several authors being contacted due to ambiguous reporting. Similar clarifications were required for the reporting of birth complications, as some studies reported on delivery complications whereas others combined these complications in addition to the neonatal period.
Several retrospective cohort studies observed an association between general health/illness in the first three to four years and MIH, and the one study that conducted multiple logistic regression came to the same conclusion, however, the authors of this review found that variation in the definition of general health/illness made analysis problematic. There is conflicting evidence in the studies included in this review as to a positive association with asthma and MIH.
The authors report a lack of consensus agreement amongst the studies that evaluated an association between antibiotic use, both type and timing, and MIH. The association between breastfeeding and MIH is not apparent.
The authors conclude that the results from the three studies that investigated the aetiology of HSPM are relatively robust due to the prospective nature of one study and the low risk of bias and adjustment for confounders in the others. These studies would suggest that antibiotic use during pregnancy is unlikely to be associated with MIH but maternal alcohol intake may be. Due to the small number of studies, including two on the same specific patient population, the conclusions should be interpreted with caution, particularly maternal alcohol intake, as this potentially could be more of a confounder than a risk factor.
Systematic reviews of observational studies are often challenging due to the variation in observational study design. This review seems to follow that pattern with the authors of this review discussing three major problems with the MIH studies included in this review: lack of adjustment for confounders; retrospective nature of most of the studies; lack of detail and consistency in exposures investigated limits comparisons between studies. Due to these differences, a meta-analysis was not able to be undertaken and a pooled result was not obtainable. The authors highlighted that omitting non-English papers could have resulted in important findings not being assessed but I wonder whether it would have made a significant difference. Despite the limitations noted in this review, the authors confirm there appears to be growing evidence for an association between early childhood illness and MIH. Further prospective studies, adjusting for confounders, are required to investigate this further.
Practice point
-
Early childhood and maternal illness, during pre-, peri- and postnatal periods are essential pieces of information to elicit from a history when MIH/HSPM is suspected.
-
MIH can pose a real problem to the clinician and a specialist paediatric dental opinion is often recommended.
References
Balmer R, Toumba J, Godson J, Duggal M . The prevalence of molar incisor hypomineralisation in Northern England and its relationship to socioeconomic status and water fluoridation. Int J Paediatr Dent 2012; 22:250–257.
Willmott NS, Bryan RAE, Duggal MS . Molar-incisor-hypomineralisation: a literature review. Eur Arch Paediatr Dent 2008; 9: 172–179.
Weerheijm KL, Mejàre I . Molar incisor hypomineralization: a questionnaire inventory of its occurrence in member countries of the European Academy of Paediatric Dentistry (EAPD). Int J Paediatr Dent 2003; 13: 411–416.
Lygidakis NA, Wong F, Jälevik B, Vierrou AM, Alaluusua S, Espelid I . Best Clinical Practice Guidance for clinicians dealing with children presenting with Molar-Incisor-Hypomineralisation (MIH): An EAPD Policy Document. Eur Arch Paediatr Dent 2010; 11: 75–81.
Elfrink ME, Schuller AA, Weerheijm KL, Veerkamp JS . Hypomineralized second primary molars: prevalence data in Dutch 5-year-olds. Caries Res 2008; 42:282–285.
Mittal R, Chandak S, Chandwani M, Singh P, Pimpale J . Assessment of association between molar incisor hypomineralization and hypomineralized second primary molar. J Int Soc Prev Community Dent 2016; 6: 34–39.
Lygidakis NA, Dimou G, Marinou D . Molar-incisor-hypomineralisation (MIH). A retrospective clinical study in Greek children. II. Possible medical aetiological factors. Eur Arch Paediatr Dent 2008; 9: 207–217.
van Amerongen WE, Kreulen CM . Cheese molars - a pilot study of the etiology of hypocalcifcations in first permanent molars. ASDC J Dent Child 1995; 62: 266–269.
Waidyasekera K, Nikaido T, Weerasinghe D, et al. Why does fluorosed dentine show a higher susceptibility for caries: an ultra-morphological explanation. J Med Dent Sci. 2010; 57: 17–23.
Jälevik B, Norén JG, Klingberg G, Barregård L . Etiologic factors influencing the prevalence of demarcated opacities in permanent first molars in a group of Swedish children. Eur J Oral Sci 2001; 109: 230–234.
Author information
Authors and Affiliations
Additional information
Address for correspondence: Mihiri Silva, Plastic Surgery Research, Murdoch Children's Research Institute, Royal Children's Hospital, 70 Flemington Rd, Parkville 3052, Vic., Australia. E-mail: mihiri.silva@mcri.edu.au
Silva MJ, Scurrah KJ, Craig JM, Manton DJ, Kilpatrick N. Etiology of molar incisor hypomineralization - A systematic review. Community Dent Oral Epidemiol 2016; 44: doi: 10.1111/cdoe.12229. [Epub ahead of print] Review. PubMed PMID: 27121068
Rights and permissions
About this article
Cite this article
Taylor, G. Molar incisor hypomineralisation. Evid Based Dent 18, 15–16 (2017). https://doi.org/10.1038/sj.ebd.6401219
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ebd.6401219
This article is cited by
-
Dietary and metabolic effects on the oral status of patients with phenylketonuria: a nation-based cross-sectional study
Clinical Oral Investigations (2023)
-
Diagnostik und Therapie der Molaren-Inzisiven-Hypomineralisation – Volkskrankheit Kreidezähne
wissen kompakt (2022)
-
Saliva proteomic patterns in patients with molar incisor hypomineralization
Scientific Reports (2020)
-
Prevalence and presentation patterns of enamel hypomineralisation (MIH and HSPM) among paediatric hospital dental patients in Toronto, Canada: a cross-sectional study
European Archives of Paediatric Dentistry (2020)