Abstract
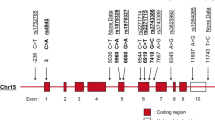
Systemic lupus erythematosus (SLE) is an autoimmune disorder characterized by production of autoantibodies against intracellular antigens and tissue injury. Defective apoptosis of activated immune cells leads to the development of autoantibodies in SLE. FasL initiated apoptosis is central for peripheral tolerance. Fas deficiencies in humans and mice predispose toward systemic autoimmunity. SLE is conferred by many genes. The genetic effects may be concentrated by familial clustering or by stratifying of subphenotypes. We have tested polymorphisms and haplotypes in FAS and FASL for association to SLE or subphenotypes in 126 multiplex American SLE pedigrees and found association of the FAS codon214 ACC/T as well as the FAS−670G>A′-codon214 ACC/T′ haplotype to thrombocytopenia in SLE. Furthermore we have functionally characterized the FAS/FASL promoter polymorphisms associated with SLE in other populations and demonstrate that the activity depends on the allelic variants as well as on the haplotype. The presence of FAS−670G, which affects STAT1 binding, leads to the highest activity. FASL−844C activity is modified by the cis acting −478A and, hence, the haplotype and not the individual variant, determines the promoter activity. We conclude that the FAS/FASL promoter haplotypes are functional and that polymorphisms in FAS may contribute to thrombocytopenia in SLE.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Harley JB, Moser KL, Gaffney PM, Behrens TW . The genetics of human systemic lupus erythematosus. Curr Opin Immunol 1998; 10: 690–696.
Deapen D, Escalante A, Weinrib L et al. A revised estimate of twin concordance in systemic lupus erythematosus. Arthritis Rheum 1992; 35: 311–318.
Kelly JA, Moser KL, Harley JB . The genetics of systemic lupus erythematosus: putting the pieces together. Genes Immun 2002; 3 (Suppl 1): S71–S85.
Salmon JE, Millard S, Schachter LA et al. Fc gamma RIIA alleles are heritable risk factors for lupus nephritis in African Americans. J Clin Invest 1996; 97: 1348–1354.
Arnett FC . The genetics of human lupus. In: Wallace DJ, Hahn BH (eds). Dubois' Lupus Erythematosus. Williams & Wilkins: Baltimore, 1997, pp 77–117.
Moser KL, Neas BR, Salmon JE et al. Genome scan of human systemic lupus erythematosus: evidence for linkage on chromosome 1q in African-American pedigrees. Proc Natl Acad Sci USA 1998; 95: 14869–14874.
Gaffney PM, Kearns GM, Shark KB et al. A genome-wide search for susceptibility genes in human systemic lupus erythematosus sib-pair families. Proc Natl Acad Sci USA 1998; 95: 14875–14879.
Lindqvist AKB, Steinsson K, Johanneson B et al. A susceptibility locus for human systemic lupus erythematosus (hSLE1) on chromosome 2q. J Autoimmun 2000; 14: 169–178.
Gaffney PM, Ortmann WA, Selby SA et al. Genome screening in human systemic lupus erythematosus: results from a second Minnesota cohort and combined analyses of 187 sib-pair families. Am J Hum Genet 2000; 66: 547–556.
Shai R, Quismorio FP, Li L et al. Genome-wide screen for systemic lupus erythematosus susceptibility genes in multiplex families. Hum Mol Genet 1999; 8: 639–644.
Gray-Mcguire C, Moser KL, Gaffney PM et al. Genome scan of human systemic lupus erythematosus by regression modeling: evidence of linkage and epistasis at 4p16–15.2. Am J Hum Genet 2000; 67: 1460–1469.
Koskenmies S, Lahermo P, Julkunen H, Ollikainen V, Kere J, Widen E . Linkage mapping of systemic lupus erythematosus (SLE) in Finnish families multiply affected by SLE. J Med Genet 2004; 41: e2–e5.
Tsao BP, Grossman JA, Riemekasten G et al. Familiality and co-occurrence of clinical features of systemic lupus erythematosus. Arthritis Rheum 2002; 46: 2678–2685.
Rao S, Olson JM, Moser KL et al. Linkage analysis of human systemic lupus erythematosus-related traits: a principal component approach. Arthritis Rheum 2001; 44: 2807–2818.
Olson JM, Song Y, Dudek DM et al. A genome screen of systemic lupus erythematosus using affected-relative-pair linkage analysis with covariates demonstrates genetic heterogeneity. Genes Immun 2002; 3 (Suppl 1): S5–S12.
Drenkard C, Villa AR, Alarcon-Segovia D, Perez-Vazquez ME . Influence of the antiphospholipid syndrome in the survival of patients with systemic lupus erythematosus. J Rheumatol 1994; 21: 1067–1072.
Mok CC, Lee KW, Ho CT, Lau CS, Wong RW . A prospective study of survival and prognostic indicators of systemic lupus erythematosus in a southern Chinese population. Rheumatology (Oxford) 2000; 39: 399–406.
Reveille JD, Bartolucci A, Alarcon GS . Prognosis in systemic lupus erythematosus. Negative impact of increasing age at onset, black race, and thrombocytopenia, as well as causes of death. Arthritis Rheum 1990; 33: 37–48.
Ward MM, Pyun E, Studenski S . Mortality risks associated with specific clinical manifestations of systemic lupus erythematosus. Arch Intern Med 1996; 156: 1337–1344.
Scofield RH, Bruner GR, Kelly JA et al. Thrombocytopenia identifies a severe familial phenotype of systemic lupus erythematosus and reveals genetic linkages at 1q22 and 11p13. Blood 2003; 101: 992.
Adachi M, Watanabe-Fukunaga R, Nagata S . Aberrant transcription caused by the insertion of an early transposable element in an intron of the Fas antigen gene of lpr mice. Proc Natl Acad Sci USA 1993; 90: 1756–1760.
Watanabe-Fukunaga R, Brannan CI, Copeland NG, Jenkins NA, Nagata S . Lymphoproliferation disorder in mice explained by defects in Fas antigen that mediates apoptosis. Nature 1992; 356: 314–317.
Takahashi T, Tanaka M, Brannan CI et al. Generalized lymphoproliferative disease in mice, caused by a point mutation in the Fas ligand. Cell 1994; 76: 969–976.
Nagata S, Golstein P . The Fas death factor. Science 1995; 267: 1449–1456.
Giese T, Davidson WF . Chronic treatment of C3H-lpr/lpr and C3H-gld/gld mice with anti-CD8 monoclonal antibody prevents the accumulation of double negative T cells but not autoantibody production. J immunol 1994; 152: 2000.
Budihardjo I, Oliver H, Lutter M, Luo X, Wang X . Biochemical pathways of caspase activation during apoptosis. Annu Rev Cell Dev Biol 1999; 15: 269–290.
Cohen GM . Caspases: the executioners of apoptosis. Biochem J 1997; 326 (Part 1): 1–16.
Fukuyama H, Adachi M, Suematsu S et al. Requirement of Fas expression in B cells for tolerance induction. Eur J Immunol 2002; 32: 223–230.
Inaba M, Kurasawa K, Mamura M, Kumano K, Saito Y, Iwamoto I . Primed T cells are more resistant to Fas-mediated activation- induced cell death than naive T cells. J Immunol 1999; 163: 1315–1320.
Nolsoe RL, Kristiansen OP, Sangthongpitag K et al. Complete molecular scanning of the human Fas gene: mutational analysis and linkage studies in families with type I diabetes mellitus. The Danish Study Group of Diabetes in Childhood and The Danish IDDM Epidemiology and Genetics Group. Diabetologia 2000; 43: 800.
Nolsoe RL, Kristiansen OP, Larsen ZM, Johannesen J, Pociot F, Mandrup-Poulsen T . Complete mutation scan of the human Fas ligand gene: linkage studies in Type I diabetes mellitus families. Diabetologia 2002; 45: 134.
Sunden SL, Alward WL, Nichols BE et al. Fine mapping of the autosomal dominant juvenile open angle glaucoma (GLC1A) region and evaluation of candidate genes. Genome Res 1996; 6: 862.
Reichlin M, Mattioli M . Correlation of a precipitin reaction to an RNAprotein antigen and a low prevalence of nephritis in patients with systemic lupus erythematosus. N Engl J Med 1972; 286: 908.
Kanemitsu S, Ihara K, Saifddin A et al. A functional polymorphism in Fas (CD95/APO-1) gene promoter associated with systemic lupus erythematosus. J Rheumatol 2002; 29: 1183–1188.
Rudert F, Lindridge E, Lasham A, Wang Y, Grandison P, Watson J . Silencer and enhancer regions in the human CD95 (Fas/APO-1) gene with sequence similarity to the granulocyte–macrophage colony-stimulating factor promoter: binding of single strand-specific silencer factors and AP-1 and NF-AT-like enhancer factors. DNA Cell Biol 1998; 17: 991.
Lee YH, Kim YR, Ji JD, Sohn J, Song GG . Fas promoter −670 polymorphism is associated with development of anti-RNP antibodies in systemic lupus erythematosus. J Rheumatol 2001; 28: 2008–2011.
Huang QR, Danis V, Lassere M, Edmonds J, Manolios N . Evaluation of a new Apo-1/Fas promoter polymorphism in rheumatoid arthritis and systemic lupus erythematosus patients. Rheumatol 1999; 38: 645–651.
Wu J, Metz C, Xu X et al. A novel polymorphic CAAT/enhancer-binding protein beta element in the FasL gene promoter alters Fas ligand expression: a candidate background gene in African American systemic lupus erythematosus patients. J Immunol 2003; 170: 132–138.
Cartegni L, Chew SL, Krainer AR . Listening to silence and understanding nonsense: exonic mutations that affect splicing. Nat Rev Genet 2002; 3: 285–298.
Rust S, Funke H, Assmann G . Mutagenically separated PCR (MS-PCR): a highly specific one step procedure for easy mutation detection. Nucleic Acids Res 1993; 21: 3623–3629.
Spielman RS, McGinnis RE, Ewens WJ . Transmission test for linkage disequilibrium: the insulin gene region and insulin-dependent diabetes mellitus (IDDM). Am J Hum Genet 1993; 52: 506–516.
Martin ER, Monks SA, Warren LL, Kaplan NL . A test for linkage and association in general pedigrees: the Pedigree Disequilibrium Test. Am J Hum Genet 2000; 67: 146–154.
Kong A, Cox NJ . Allele-sharing models: LOD scores and accurate linkage tests. Am J Hum Genet 1997; 61: 1179–1188.
Kaplan NL, Hill WG, Weir BS . Likelihood methods for locating disease genes in nonequilibrium populations. Am J Hum Genet 1995; 56: 18–32.
Lewontin RC . On measures of gametic disequilibrium. Genetics 1988; 120: 849–852.
Holtz-Heppelmann CJ, Algeciras A, Badley AD, Paya CV . Transcriptional regulation of the human FasL promoter-enhancer region. J Biol Chem 1998; 273: 4416.
Acknowledgements
We thank Marja Deckert for expert technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nolsøe, R., Kelly, J., Pociot, F. et al. Functional promoter haplotypes of the human FAS gene are associated with the phenotype of SLE characterized by thrombocytopenia. Genes Immun 6, 699–706 (2005). https://doi.org/10.1038/sj.gene.6364259
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gene.6364259
Keywords
This article is cited by
-
Fas/FasL gene polymorphism in patients with Hashimoto’s thyroiditis in Turkish population
Journal of Endocrinological Investigation (2017)
-
Functional polymorphisms in cell death pathway genes FAS and FAS ligand and risk of alopecia areata
Archives of Dermatological Research (2013)
-
Functional Polymorphisms of the FAS Gene Associated with Risk of Vitiligo in Chinese Populations: A Case–Control Analysis
Journal of Investigative Dermatology (2008)
-
Fas/Fas ligand gene polymorphism in patients with papillary thyroid cancer in the Turkish population
Journal of Endocrinological Investigation (2007)
-
Association of a microsatellite in FASL to type II diabetes and of the FAS-670G>A genotype to insulin resistance
Genes & Immunity (2006)