Abstract
Rett syndrome is a largely sporadic, X-linked neurological disorder with a characteristic phenotype, but which exhibits substantial phenotypic variability. This variability has been partly attributed to an effect of X chromosome inactivation (XCI). There have been conflicting reports regarding incidence of skewed X inactivation in Rett syndrome. In rare familial cases of Rett syndrome, favourably skewed X inactivation has been found in phenotypically normal carrier mothers. We have investigated the X inactivation pattern in DNA from blood and buccal cells of sporadic Rett patients (n=96) and their mothers (n=84). The mean degree of skewing in blood was higher in patients (70.7%) than controls (64.9%). Unexpectedly, the mothers of these patients also had a higher mean degree of skewing in blood (70.8%) than controls. In accordance with these findings, the frequency of skewed (XCI ≥80%) X inactivation in blood was also higher in both patients (25%) and mothers (30%) than in controls (11%). To test whether the Rett patients with skewed X inactivation were daughters of skewed mothers, 49 mother–daughter pairs were analysed. Of 14 patients with skewed X inactivation, only three had a mother with skewed X inactivation. Among patients, mildly affected cases were shown to be more skewed than more severely affected cases, and there was a trend towards preferential inactivation of the paternally inherited X chromosome in skewed cases. These findings, particularly the greater degree of X inactivation skewing in Rett syndrome patients, are of potential significance in the analysis of genotype–phenotype correlations in Rett syndrome.
Similar content being viewed by others
Introduction
Rett syndrome (RTT) is an X-linked neurological disorder primarily affecting females.1 The disorder is characterised by grossly normal but subtly compromised development over 6–18 months, followed by developmental regression, loss of speech, loss of purposeful hand skills, development of stereotypical hand movements, reduced brain growth and often severe mental retardation. There is a degree of variability in the phenotype despite the presence of all the cardinal features. RTT is usually caused by mutations in the methyl-CpG-binding protein type 2 gene, MECP2, in chromosome Xq28.2
X chromosome inactivation (XCI) renders females mosaic for two cell populations. Most females have approximately equal amounts of the two cell types. A skewed X inactivation pattern may arise due to chance, to inherited factors or to selective forces acting on dividing cells with particular genotypes at X-linked genes.3, 4, 5, 6 For some X-linked disorders, the X inactivation patterns in female carriers are correlated with the phenotype.7, 8, 9, 10 The phenotypic variation in Rett syndrome may therefore be influenced by a skewed X inactivation pattern.
An increased frequency of skewed X inactivation in patients with Rett syndrome has been reported,11, 12, 13 but with no clear association between X inactivation pattern and clinical phenotype. In a small number of cases, phenotypically normal or mildly affected mothers with MECP2 mutations have classically affected daughters. The mothers in these cases have been shown to have extremely skewed X inactivation and it is assumed that the direction of skewing favours expression from the X chromosome carrying the normal allele.14, 15, 16, 17 In this study, we present an analysis of X chromosome inactivation in patients with sporadic Rett syndrome and their mothers.
Materials and methods
Subjects
The Rett patients and their mothers were collected from two different sources. Set 1 comprised 35 patients and 34 mothers recruited at a Family Weekend arranged by the Rett Syndrome Association UK in October 2000. Set 2 comprised 61 patients and 50 mothers recruited by the Glasgow group employing postal invitation. None of the cases were recruited on the basis of pre-existing knowledge of their mutation status. Set 2 were selected to consist of a mixture of classic and atypical cases on clinical grounds, weighted towards the classic picture. All cases were recorded in the British Isles Survey (BIS) of Rett syndrome database.18 Composite clinical severity scores (on a scale of 0–10) were assigned by AMK, as described.18 Briefly, the composite score is made up of 5 sub-scores, on a 0–2 scale, for feeding difficulties, predominant muscle tone on examination, ability to walk unsupported, presence of epilepsy and scoliosis. For each item the score is 0 if there is no problem, 1 if there is any problem and 2 if the problem is severe. The final score is given out of 10, but may be expressed as a percentage, for example, 1 out of 10 becomes ‘10%’. Blood and buccal cell samples were collected with informed consent from parents or carers. Appropriate ethical approval was obtained. Informative XCI results were obtained in 69 blood and 63 buccal samples from patients and in 67 blood and 49 buccal samples from mothers. Of the patients with blood XCI ratio results and known severity score, 60 had classic Rett syndrome and seven had non-classic Rett syndrome. Patients were of age 2–41 years (median age 15 years, median severity score 7) and mothers of age 28–55 years.
In all, 85% of the patients in Set 1 and 79% of the patients in those members of Set 2 used in this study harboured identified MECP2 mutations.19
Control samples used in statistical comparisons were 49 previously analysed (Knudsen et al, unpublished) healthy females age 19–63 years (for buccal cell XCI data) and 153 normal healthy females (for blood XCI data), of whom 117 were anonymised blood donors (age 19–55 years) and 36 were sampled from the set of individuals used in the buccal control set (age 19–55).
Sample collection and DNA preparation
DNA was isolated from blood by standard methods. Buccal cell samples were collected with buccal swabs (C.E.P Gibco or Epicentre Technologies). Swabs were gently brushed across the inside of the cheek six times on each side, and then air-dried. DNA was extracted using the QiaPREP kit (QIAGEN Inc.).
X inactivation analysis
The X inactivation pattern was determined by PCR analysis of a polymorphic trinucleotide repeat in the first exon of the androgen receptor (AR) gene after digestion with HpaII.20 All samples were analysed in duplicate. All samples were analysed with and without HpaII digestion. A control female with known extreme skewed X inactivation as well as a male control were included in each run. Representative samples from each batch of DNA were analysed by PCR amplification of a segment of the MIC2 gene, which escapes inactivation and is unmethylated on both chromosomes.21 All samples analysed showed complete digestion. Degree of skewing of X inactivation was calculated as the fractional peak height ratio (expressed as %) for the more strongly amplified allele. Degree of skewing thus varies between 50 and 100%, where 50% reflects a random pattern and 100% a completely skewed pattern. Values of 80% were classified as ‘skewed’. Parental origin of the inactivated X chromosome in patients was assessed by comparison of AR repeat allele sizes of patients, mothers, and in some cases fathers also.
Statistical methods
The statistical software SPSS v11 was used for all analyses.
The distributions of degree of skewing were not normally distributed (Kolmogorov–Smirnov test of normality). A log-transformation was done for the distribution of degree of skewing in blood, with subsequent ANOVA test and post hoc Bonferroni correction (P-values multiplied by the number of groups compared; three). The mean degree of skewing was calculated by using the mean of the lognormal distribution (L: μ, σ, m=exp μ): mean=m × exp((1/2)σ2). Log values were backtransformed for presentation in Table 1. The distribution of degree of skewing in buccal cells was not transformed, and comparisons were done using the Kruskal–Wallis test, with subsequent post hoc Mann–Whitney tests and Bonferroni correction (three groups compared). The Mann–Whitney test was used for comparing the age distributions of skewed and non-skewed patients.
The Pearson χ2 test was used for comparing frequencies of skewed X inactivation. All three groups were compared first, and if they were significantly different, subsequent pair wise comparisons were done with Bonferroni correction.
Pearson Correlation was used for comparing degree of skewing in mothers and daughters.
Degree of skewing in three clinical severity groups was compared by ANOVA (degree of skewing in these three patient groups was normally distributed) with post hoc Bonferroni corrections.
For analysis of origin of the preferentially active X, a binomial test with a test probability of 0.5 for either maternal or paternal origin was used.
The threshold for statistical significance was set at 5% (2-tailed). All Bonferroni corrected P-values are noted P*.
Results
Distribution and comparison of X chromosome inactivation patterns
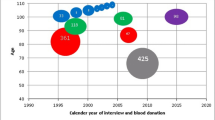
The distribution of degree of skewing in patients, mothers and controls is presented in Figure 1, and mean (or median) degree of skewing and frequency of skewed individuals are shown in Table 1.
The degree of skewing in blood differed between the three groups (P<0.001), and mean degree of skewing was higher in both mothers and patients than in controls (Table 1). In buccal cells, the degree of skewing differed between the groups (P=0.015), and median degree of skewing in mothers was higher than in controls (P*=0.015) (Table 1).
The frequency of skewed X inactivation in blood also differed (P=0.002), with a higher frequency in patients (25%, P*=0.03) and mothers (30%, P*=0.003) than in controls (11%). There was no difference in age between patients with skewed X inactivation (age 3–30 years, median 15.5) and non-skewed X inactivation (age 2–41 years, median 13) (P=0.548). There was no significant difference in frequency of skewed X inactivation in buccal cells (Table 1).
Comparison of X chromosome inactivation pattern in mothers and daughters
To test for a possible familial effect on skewed X inactivation, 49 mother–daughter pairs were analysed. Of 14 patients with skewed X inactivation in blood, only three had mothers with skewed X inactivation. Furthermore, there was no significant correlation in the degree of skewing between mothers and their daughters (Pearson Correlation R=−0.09, P=0.52).
X chromosome inactivation skewing and clinical severity
The relationship between XCI skewing and clinical severity was explored by dividing the patients into three severity groups, mild (BIRS composite severity score 0–3; age at determination of severity score, 2–26 years, median 8.5), moderate (severity score 4–6; age 2–37 years, median 12.0) and severe (severity score 7–10; age 4–37 years, median 12.5), and comparing mean degree of skewing in blood statistically. The mean degree of skewing differed between the three groups (P=0.01). Post hoc tests showed that mild cases had a higher mean degree of skewing (80.6%, n=8) than both moderate (67.0%, n=23) (P*=0.008) and severely affected cases (70.4 %, n=36) (P*=0.048), whereas moderate and severe groups were not significantly different. The relationship between skewing and possession of an MECP2 mutation was explored in similar fashion. There was no significant difference in mean degree of skewing or frequency of skewing between mutation-positive (N=54) and mutation-negative (N=13) patients (data not shown). The proportion of mutation-positive patients did not differ between skewed and non-skewed patients: 14 of 17 patients with skewed X inactivation and 40 of 50 patients with non-skewed X inactivation were mutation-positive (χ2 test, P=0.832). The three mutation-negative patients with skewed X inactivation fell into all three BIRS severity categories, mild, moderate and severe (one patient in each).
There was no difference in frequency of skewed X inactivation between classical Rett patients (13/60) and non-classical Rett patients (2/7) (P=0.649).
Parental origin of the preferentially inactive X chromosome in skewed patients
The parental origin of the preferentially inactive X could be determined in blood in 15 patients with skewed X inactivation. In four patients the maternal X was inactivated, and in 11 the paternal X was inactivated. There was thus a weak trend towards a higher frequency of inactivation of the paternal X (P=0.11). The four patients with preferential inactivation of the maternal X had severity scores of 2, 7, 8, and 10, while nine patients with preferential inactivation of the paternal X had scores of 0, 1, 1, 4, 4, 7, 8, 8, and 9. Composite severity score data were not available for the remaining two patients. Mean severity score was higher (6.8) in the patients with maternal inactivation compared to the patients with paternal inactivation (4.7), but the difference was not statistically significant (Mann Whitney test, P=0.33). Two of the four patients with preferential inactivation of the maternal X had identified mutations, whereas all 11 of those with preferential inactivation of the paternal X did so (test for difference in these proportions almost significant, P=0.057 Fisher's Exact Test).
Discussion
There are conflicting reports regarding the frequency of skewed X inactivation in Rett syndrome patients' blood cells. A few studies, employing small patient sample sizes, have shown random X inactivation in brain tissue.22, 23 This is in contrast with results using Mecp2 knockout mouse models, where an association between skewed X inactivation in brain cells and phenotypic severity was found.24, 25 The difference in reported frequencies of skewed X inactivation may partly be attributed to different definitions of the ‘skewed’ state (ranging from >65 to >85%) and different patient groups.11, 12, 13, 23, 26, 27, 28, 29 Using a cutoff of 80% for skewing, our results indicate a higher frequency of skewed X inactivation in the blood of Rett patients than in controls. X inactivation ratios were less skewed in buccal cells than in blood in the patients and mothers, in agreement with expectation from previous studies showing that in the general population X inactivation in buccal epithelia is less skewed than in blood cells.30 Higher frequencies of skewed X inactivation would thus be more difficult to detect in buccal cells.
Studies of X inactivation and phenotype correlation in Rett syndrome have given discordant results. Most studies have found an association between skewed X inactivation and a milder phenotype,17, 26, 28, 31 whereas one study also found an association between skewed X inactivation and a more severe phenotype.28 Our results support the reported association between a milder phenotype and skewed X inactivation, as we found a higher mean degree of skewing in mild cases than in moderate or severe cases. There is more than one possible explanation for this finding, however. It could, in part, be a reflection of a selection process, where cells that have inactivated the X chromosome harbouring the mutant allele have a proliferative advantage. Alternatively, it is also what would be expected if most mutations are paternal and if the paternal X is more often inactivated.
Taking this reasoning further, it has previously been reported that the paternally inherited X chromosome is preferentially inactivated in skewed Rett syndrome patients,29 and we found a similar tendency. Of 13 cases with skewing and known mutations, 11 had preferentially inactivated the paternal X chromosome in blood lymphocytes. New mutations, particularly the CpG to TpG transitions that constitute many of the commonest MECP2 mutations causing sporadic Rett, more commonly occur in the male germline.32, 33 Assuming that the mutation is of paternal origin in most cases, one would expect the four patients (two of whom have known mutations) with a preferentially inactivated maternal X to be on average more severely affected (due to the mutant allele being more commonly on the active X chromosome in these patients), while the 11 patients with a preferentially inactivated paternal X are therefore expected to be more mildly affected. The limited data set available to us in this respect did show a trend towards this prediction, but we cannot draw any firm conclusions until larger sample sizes are available and until mutation category can be taken into account in the analysis. We also do not at present have definitive data about the parent of origin of the mutation in each patient (and thus the direction of skewing relative to expression of the mutant and normal alleles), which may be expected to have a significant impact on our ability to detect differences in severity score distribution between patient groups with paternally and maternally inactive X chromosomes. More extensive studies addressing these points are ongoing and will be reported in a future publication.
Surprisingly, we found a high frequency of skewed X inactivation in mothers of Rett patients as well as in the patients themselves. Mothers of Rett patients with more than one affected child have been reported to have a skewed X inactivation pattern.15, 16, 17 Of the mothers studied here, 45 had been tested for the MECP2 mutation carried by their daughter with Rett syndrome, all proving not to carry the mutant allele. The remaining 39 mothers in the sample have not been tested, but are unlikely to be carriers, as 99.5% of Rett syndrome cases are sporadic. It is, therefore, very unlikely that skewing of X inactivation in a mother of a Rett patient is related to carrier status in the mother.
The high mean degree and frequency of skewing in the mothers cannot be explained by the similar findings in their affected daughters, since we found no correlation between degree of X inactivation skewing in mothers and that in daughters, and skewing in both mother and patient was only observed in three of 49 mother/daughter pairs. Similarly, simple age effects cannot explain the higher skewing and increased frequency of skewing in mothers. Age has been shown to have an effect on XCI skewing, but is only significant in females over 55 years of age.34, 35 The mothers in our study sample were aged from 28 to 55, and the age-related increase in degree of skewing is not therefore expected in the sample. This is also true of the patients and controls. An alternative explanation that can account for increased skewing in mothers of Rett patients is not easy to posit. We have not specifically compared mothers of patients with mothers in the general population, although we would not expect to find the same increase in degree of skewing in the latter group compared with a cross-sectional control group. The only obvious difference between mothers of Rett patients and mothers of children without the disorder is that the former are selected for having conceived and carried to term a foetus that was later to develop into an individual with Rett syndrome, the majority of whom will be carrying a paternal X chromosome with an MECP2 mutation. Thus, it is possible that skewing in mothers of Rett patients is related to conception and foetal development of this subset of children, although the mechanism of such an effect is obscure.
In conclusion, we have found a higher degree of XCI skewing in Rett patients than in controls, and shown that skewing may relate to clinical severity across the spectrum and not just at the extremes of XCI ratio. Unexpectedly, we also found a higher degree of skewing in mothers of Rett patients both in blood and buccal cells. The high degree of skewing in mothers may indicate that X inactivation influences conception or survival of Rett foetuses. Whether skewed X inactivation in mothers of sporadic Rett patients has clinical significance remains to be determined.
References
Hagberg B : Clinical manifestations and stages of Rett syndrome. Ment Retard Dev Disabil Res Rev 2002; 8: 61–65.
Amir RE, Van dV I, Wan M, Tran CQ, Francke U, Zoghbi HY : Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet 1999; 23: 185–188.
Belmont JW : Genetic control of X inactivation and processes leading to X-inactivation skewing. Am J Hum Genet 1996; 58: 1101–1108.
Naumova AK, Plenge RM, Bird LM et al: Heritability of X chromosome – inactivation phenotype in a large family. Am J Hum Genet 1996; 58: 1111–1119.
Puck JM, Willard HF : X inactivation in females with X-linked disease. N Engl J Med 1998; 338: 325–328.
Migeon BR, Haisley-Royster C : Familial skewed X inactivation and X-linked mutations: unbalanced X inactivation is a powerful means to ascertain X-linked genes that affect cell proliferation. Am J Hum Genet 1998; 62: 1555–1557.
Allen RC, Nachtman RG, Rosenblatt HM, Belmont JW : Application of carrier testing to genetic counseling for X-linked agammaglobulinemia. Am J Hum Genet 1994; 54: 25–35.
Fearon ER, Kohn DB, Winkelstein JA, Vogelstein B, Blaese RM : Carrier detection in the Wiskott Aldrich syndrome. Blood 1988; 72: 1735–1739.
Nyhan WL, Bakay B, Connor JD, Marks JF, Keele DK : Hemizygous expression of glucose-6-phosphate dehydrogenase in erythrocytes of heterozygotes for the Lesch-Nyhan syndrome. Proc Natl Acad Sci USA 1970; 65: 214–218.
Orstavik KH, Orstavik RE, Naumova AK et al: X chromosome inactivation in carriers of Barth syndrome. Am J Hum Genet 1998; 63: 1457–1463.
Camus P, Abbadi N, Perrier MC, Chery M, Gilgenkrantz S : X chromosome inactivation in 30 girls with Rett syndrome: analysis using the probe. Hum Genet 1996; 97: 247–250.
Krepischi AC, Kok F, Otto PG : X chromosome-inactivation patterns in patients with Rett syndrome. Hum Genet 1998; 102: 319–321.
Weaving LS, Williamson SL, Bennetts B et al: Effects of MECP2 mutation type, location and X-inactivation in modulating Rett syndrome phenotype. Am J Med Genet 2003; 118A: 103–114.
Schanen NC, Dahle EJ, Capozzoli F, Holm VA, Zoghbi HY, Francke U : A new Rett syndrome family consistent with X-linked inheritance expands the X chromosome exclusion map. Am J Hum Genet 1997; 61: 634–641.
Sirianni N, Naidu S, Pereira J, Pillotto RF, Hoffman EP : Rett syndrome: confirmation of X-linked dominant inheritance, and localization of the gene to Xq28. Am J Hum Genet 1998; 63: 1552–1558.
Villard L, Levy N, Xiang F et al: Segregation of a totally skewed pattern of X chromosome inactivation in four familial cases of Rett syndrome without MECP2 mutation: implications for the disease. J Med Genet 2001; 38: 435–442.
Wan M, Lee SS, Zhang X et al: Rett syndrome and beyond: recurrent spontaneous and familial MECP2 mutations at CpG hotspots. Am J Hum Genet 1999; 65: 1520–1529.
Kerr AM, Prescott RJ : Predictive value of the early clinical signs in Rett disorder. Brain Dev 2005; 27 (Suppl 1): S20–S24.
Charman T, Neilson TC, Mash V et al: Dimensional phenotypic analysis and functional categorisation of mutations reveal novel genotype-phenotype associations in Rett syndrome. Eur J Hum Genet 2005; 13: 1121–1130.
Pegoraro E, Schimke RN, Arahata K et al: Detection of new paternal dystrophin gene mutations in isolated cases of dystrophinopathy in females. Am J Hum Genet 1994; 54: 989–1003.
Anderson CL, Brown CJ : Variability of X chromosome inactivation: effect on levels of TIMP1 RNA and role of DNA methylation. Hum Genet 2002; 110: 271–278.
Shahbazian MD, Sun Y, Zoghbi HY : Balanced X chromosome inactivation patterns in the Rett syndrome brain. Am J Med Genet 2002; 111: 164–168.
Zoghbi HY, Percy AK, Schultz RJ, Fill C : Patterns of X chromosome inactivation in the Rett syndrome. Brain Dev 1990; 12: 131–135.
Watson CM, Pelka GJ, Radziewic T et al: Reduced proportion of Purkinje cells expressing paternally derived mutant Mecp2308 allele in female mouse cerebellum is not due to a skewed primary pattern of X-chromosome inactivation. Hum Mol Genet 2005; 14: 1851–1861.
Young JI, Zoghbi HY : X-chromosome inactivation patterns are unbalanced and affect the phenotypic outcome in a mouse model of rett syndrome. Am J Hum Genet 2004; 74: 511–520.
Amir RE, Van dV I, Schultz R et al: Influence of mutation type and X chromosome inactivation on Rett syndrome phenotypes. Ann Neurol 2000; 47: 670–679.
Auranen M, Vanhala R, Vosman M et al: MECP2 gene analysis in classical Rett syndrome and in patients with Rett-like features. Neurology 2001; 56: 611–617.
Hoffbuhr K, Devaney JM, LaFleur B et al: MeCP2 mutations in children with and without the phenotype of Rett syndrome. Neurology 2001; 56: 1486–1495.
Nielsen JB, Henriksen KF, Hansen C, Silahtaroglu A, Schwartz M, Tommerup N : MECP2 mutations in Danish patients with Rett syndrome: high frequency of mutations but no consistent correlations with clinical severity or with the X chromosome inactivation pattern. Eur J Hum Genet 2001; 9: 178–184.
Sharp A, Robinson D, Jacobs P : Age- and tissue-specific variation of X chromosome inactivation ratios in normal women. Hum Genet 2000; 107: 343–349.
Bienvenu T, Villard L, de Roux N et al: Spectrum of MECP2 mutations in Rett syndrome. Genet Test 2002; 6: 1–6.
Trappe R, Laccone F, Cobilanschi J et al: MECP2 mutations in sporadic cases of Rett syndrome are almost exclusively of paternal origin. Am J Hum Genet 2001; 68: 1093–1101.
Girard M, Couvert P, Carrie A et al: Parental origin of de novo MECP2 mutations in Rett syndrome. Eur J Hum Genet 2001; 9: 231–236.
Busque L, Mio R, Mattioli J et al: Nonrandom X-inactivation patterns in normal females: lyonization ratios vary with age. Blood 1996; 88: 59–65.
Kristiansen M, Knudsen GP, Bathum L et al: Twin study of genetic and aging effects on X chromosome inactivation. Eur J Hum Genet 2005; 13: 599–606.
Acknowledgements
This work was supported by the Research Council of Norway (Research Council of Norway scholarship held by GPSK), Rett Syndrome Research Foundation, International Rett Syndrome Association, Rett Syndrome Association (UK), Rett Syndrome Association (Scotland), Scottish Hospital Endowments Research Trust (SHERT/Cruden scholarship to MESB), and Institute of Biomedical and Life Sciences Research Committee, University of Glasgow. We are grateful to parents, carers and individuals with Rett syndrome for their participation in this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Knudsen, G., Neilson, T., Pedersen, J. et al. Increased skewing of X chromosome inactivation in Rett syndrome patients and their mothers. Eur J Hum Genet 14, 1189–1194 (2006). https://doi.org/10.1038/sj.ejhg.5201682
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejhg.5201682
Keywords
This article is cited by
-
Rett syndrome in Ireland: a demographic study
Orphanet Journal of Rare Diseases (2024)
-
Wide spectrum of neuronal and network phenotypes in human stem cell-derived excitatory neurons with Rett syndrome-associated MECP2 mutations
Translational Psychiatry (2022)
-
Choline Rescues Behavioural Deficits in a Mouse Model of Rett Syndrome by Modulating Neuronal Plasticity
Molecular Neurobiology (2019)
-
Treating Rett syndrome: from mouse models to human therapies
Mammalian Genome (2019)
-
X Chromosome Inactivation and Autoimmunity
Clinical Reviews in Allergy & Immunology (2010)