Abstract
Natural selection can lead to rapid changes in organisms, which can in turn influence ecosystem processes1. A key factor in the functioning of lake ecosystems is the rate at which primary producers are eaten, and major consumers, such as the zooplankton Daphnia2, can be subject to strong selection pressures when phytoplankton assemblages change. Lake Constance in central Europe experienced a period of eutrophication (the biological effects of an input of plant nutrients) during the 1960s–70s3, which caused an increase4 in the abundance of nutritionally poor or even toxic5 cyanobacteria. By hatching long-dormant eggs6 of Daphnia galeata found in lake sediments, we show that the mean resistance of Daphnia genotypes to dietary cyanobacteria increased significantly during this eutrophication. This rapid evolution of resistance has implications for the ways that ecosystems respond to nutrient enrichment through the impact of grazers on primary production.
Similar content being viewed by others
Main
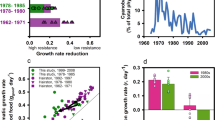
Genetically distinct3 Daphnia galeata clones were collected as diapausing eggs from sediments of known age3 obtained from a core taken from Lake Constance in 1997. Parthenogenetic lines were maintained for each clone for at least 40 generations to minimize maternal effects. We tested 32 clones from three sediment ages for their resistance to dietary cyanobacteria: 12 clones from 1962–64 and 1969–71 (before and just after the appearance of cyanobacteria; Fig. 1), 10 clones from 1978–80 (peak eutrophication), and 10 clones from 1992–94 and 1995–97 (when the period of eutrophication had passed).
Resistance to cyanobacteria was measured as the effect of diet on the somatic juvenile growth rate (gj) of Daphnia7: gj=[ln(Wm)−ln(Wn)]/t, where Wm and Wn are the masses of mature and neonate Daphnia, respectively, and t is the development period. For each clone, gj was measured for animals fed two different diets (supplied at 1 mg carbon per litre): one diet (gj, poor) contained a mixture of a toxic cyanobacterium (Microcystis aeruginosa, 20% by carbon content) and a high-quality algal resource (Scenedesmus obliquus, 80% by carbon content); the other diet (gj, good) contained only Scenedesmus. Microcystis was originally isolated from Lake Constance in 1972, is toxic to Daphnia pulicaria8 and contains high concentrations of microcystin-LR hepatotoxin, whereas Scenedesmus promotes growth and reproduction in Daphnia6. Microcystis and Scenedesmus were fed to Daphnia as single cells of similar size (4.2 µm).
For each clone, gj, good and gj, poor were measured separately by using three replicate flow-through vessels (5–17 individuals each) with food levels in excess of growth-limiting concentrations9. Because there was variation among clones in gj, good (likelihood ratio test, χ2=120.1, d.f.=1, P<0.0001), resistance to dietary cyanobacteria was standardized as the growth-rate reduction, R, which is the fractional reduction in gj on poor food relative to that on good food: R=(gj, good−gj, poor)/gj, good.
The Daphnia population evolved an increased ability to cope with a diet containing cyanobacteria (Fig. 2). Genotypes from both 1978–80 and the 1990s exhibit lower growth-rate reduction than those from 1962–64 and 1969–71 (ANOVA planned contrasts, F=10.3, d.f.=1, 29, P=0.003, and F=11.9, d.f.=1, 29, P=0.002, respectively). The rapid response observed during 1969–80 is attributable entirely to a reduction in genetic variance (likelihood ratio test, χ2=8.24, d.f.=1, P=0.002). There was a broad range of Daphnia genotypes of different growth-rate reductions in the lake during 1962–64 and 1969–71, but the genotypes that were most heavily affected by dietary cyanobacteria were virtually eliminated within ten years of continued summertime exposure to cyanobacteria.
The mean resistance of Daphnia to cyanobacteria was unchanged between 1978–80 and the 1990s (ANOVA planned contrast, F=0.06, d.f.=1, 29, P=0.82). However, the variance of growth-rate reductions was greater in the 1990s than in 1978–80 (likelihood ratio test, χ2=4.72, d.f.=1, P=0.015), perhaps as a result of the water column being reinvaded by animals hatching from the dormant egg bank6.
These short-term evolutionary changes may significantly affect the course of ecosystem change. Greater abundance of cyanobacteria during eutrophication is typically considered to be a response to increased nutrient inputs10. However, rapid adaptive evolution in grazing zooplankton populations may be an important feedback mechanism that is critical to understanding the net effect of eutrophication on primary producers in lakes.
References
Thompson, J. N. Trends Ecol. Evol. 13, 329–332 (1998).
Carpenter, S. R. et al. in Comparative Analyses of Ecosystems: Patterns, Mechanisms, and Theories (eds Cole, J., Lovett, G. & Findlay, S.) 67–96 (Springer, New York, 1991).
Weider, L. J. et al. Proc. R. Soc. Lond. B 264, 1613–1618 (1997).
Kümmerlin, R. Arch. Hydrobiol. Spec. Issue Adv. Limnol. 53, 109–117 (1998).
Lampert, W. Mem. Ist. Ital. Idrobiol. 45, 143–192 (1987).
Hairston, N. G. Jr Limnol. Oceanogr. 41, 1087–1092 (1996).
Lampert, W. Int. Ver. Theor. Angew. Limnol. Verh. 21, 1436–1440 (1981).
Lampert, W. & Trubetskova, I. Funct. Ecol. 10, 631–635 (1996).
Lampert, W., Schmitt, R. D. & Muck, P. Bull. Mar. Sci. 43, 620–640 (1988).
Smith, V. H. in Successes, Limitations, and Frontiers in Ecosystem Science (eds Pace, M. L. & Groffman, P. M.) 7–49 (Springer, New York, 1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hairston, N., Lampert, W., Cáceres, C. et al. Rapid evolution revealed by dormant eggs. Nature 401, 446 (1999). https://doi.org/10.1038/46731
Issue Date:
DOI: https://doi.org/10.1038/46731
This article is cited by
-
Levels of nutrient enrichment determine the emergence of zooplankton from resting egg banks
Hydrobiologia (2024)
-
Daphnia as a versatile model system in ecology and evolution
EvoDevo (2022)
-
Path probability selection in nature and path integral
Scientific Reports (2022)
-
Local adaptation mediates direct and indirect effects of multiple stressors on consumer fitness
Oecologia (2022)
-
Eco-bioengineering tools in ecohydrological assessment of eutrophic water bodies
Ecotoxicology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.