Abstract
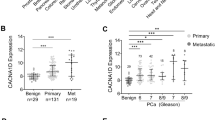
We previously identified high levels of Nav1.7 voltage-gated sodium channel α-subunit (VGSCα) mRNA and protein in human prostate cancer cells and tissues. Here, we investigated auxillary β-subunit (VGSCβs) expression. In vitro, the combined expression of all four VGSCβs was significantly (∼4.5-fold) higher in strongly compared to weakly metastatic cells. This was mainly due to increased β1-expression, which was under androgenic control. In vivo, β1–β4 mRNAs were detectable and their expression in CaP vs non-CaP tissues generally reflected the in vitro levels in relation to metastatic potential. The possible role(s) of VGSCβs (VGSCα-associated and VGSCα-independent) in prostate cancer are discussed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Grimes JA, Fraser SP, Stephens GJ, Downing JE, Laniado ME, Foster CS et al. Differential expression of voltage-activated Na+ currents in two prostatic tumour cell lines: contribution to invasiveness in vitro. FEBS Letts 1995; 369: 290–294.
Laniado ME, Lalani EN, Fraser SP, Grimes JA, Bhangal G, Djamgoz MB et al. Expression and functional analysis of voltage-activated Na+ channels in human prostate cancer cell lines and their contribution to invasiveness in vitro. Am J Path 1997; 150: 1213–1221.
Djamgoz MB, Mycielska M, Madeja Z, Fraser SP, Korohoda W . Directional movement of rat prostate cancer cells in direct-current electric field: involvement of voltage gated Na+ channel activity. J Cell Sci 2001; 114: 2697–2705.
Fraser SP, Salvador V, Manning EA, Mizal J, Altun S, Raza M et al. Contribution of functional voltage-gated Na+ channel expression to cell behaviors involved in the metastatic cascade in rat prostate cancer: I. Lateral motility. J Cell Physiol 2003; 195: 479–487.
Diss JK, Archer SN, Hirano J, Fraser SP, Djamgoz MB . Expression profiles of voltage-gated Na+ channel α-subunit genes in rat and human prostate cancer cell lines. Prostate 2001; 48: 165–178.
Diss JK, Stewart D, Pani F, Foster CS, Walker MM, Patel A et al. A potential novel marker for human prostate cancer: voltage-gated sodium channel expression in vivo. Prostate Cancer Prostate Dis 2005; 8: 266–273.
Isom LL, De Jongh KS, Patton DE, Reber BF, Offord J, Charbonneau H et al. Primary structure and functional expression of the beta 1 subunit of the rat brain sodium channel. Science 1992; 256: 839–842.
Isom LL, Ragsdale DS, De Jongh KS, Westenbroek RE, Reber BF, Scheuer T et al. Structure and function of the beta 2 subunit of brain sodium channels, a transmembrane glycoprotein with a CAM motif. Cell 1995; 83: 433–442.
Morgan K, Stevens EB, Shah B, Cox PJ, Dixon AK, Lee K et al. beta 3: an additional auxiliary subunit of the voltage-sensitive sodium channel that modulates channel gating with distinct kinetics. Proc Nat Acad Sci USA 2000; 97: 2308–2313.
Yu FH, Westenbroek RE, Silos-Santiago I, McCormick KA, Lawson D, Ge P et al. Sodium channel beta4, a new disulfide-linked auxiliary subunit with similarity to beta2. J Neurosci 2003; 23: 7577–7585.
Oh Y, Waxman SG . Beta 1 subunit mRNA of the rat brain Na+ channel is expressed in glial cells. Proc Natl Acad Sci USA 1994; 91: 9985–9989.
Kazen-Gillespie KA, Ragsdale DS, D'Andrea MR, Mattei LN, Rogers KE, Isom LL . Cloning, localization, and functional expression of sodium channel beta1A subunits. J Biol Chem 2000; 275: 1079–1088.
Qin N, D'Andrea MR, Lubin ML, Shafaee N, Codd EE, Correa AM . Molecular cloning and functional expression of the human sodium channel beta1B subunit, a novel splicing variant of the beta1 subunit. Eur J Biochem 2003; 270: 4762–4770.
Fahmi AI, Patel M, Stevens EB, Fowden AL, John JE, Lee K et al. The sodium channel beta-subunit SCN3b modulates the kinetics of SCN5a and is expressed heterogeneously in sheep heart. J Physiol 2001; 537: 693–700.
Qu Y, Curtis R, Lawson D, Gilbride K, Ge P, Di Stefano PS et al. Differential modulation of sodium channel gating and persistent sodium currents by the beta1, beta2, and beta3 subunits. Mol Cell Neurosci 2001; 18: 570–580.
Meadows LS, Chen YS, Powell AJ, Clare JJ, Ragsdale DS . Functional modulation of human brain Nav1.3 sodium channels, expressed in mammalian cells, by auxiliary beta 1, beta 2 and beta 3 subunits. Neuroscience 2002; 114: 745–753.
Vijayaragavan K, Powell AJ, Kinghorn IJ, Chahine M . Role of auxiliary beta1-, beta2-, and beta3-subunits and their interaction with Na(v)1.8 voltage-gated sodium channel. Biochem Biophys Res Commun 2004; 319: 531–540.
Chen C, Bharucha V, Chen Y, Westenbroek RE, Brown A, Malhotra JD et al. Reduced sodium channel density, altered voltage dependence of inactivation, and increased susceptibility to seizures in mice lacking sodium channel beta 2-subunits. Proc Nat Acad Sci USA 2002; 99: 17072–17077.
Malhotra JD, Koopmann MC, Kazen-Gillespie KA, Fetman N, Hortsch M, Isom LL . Structural requirements for interaction of sodium channel beta 1 subunits with ankyrin. J Biol Chem 2002; 277: 26681–26688.
Xiao ZC, Ragsdale DS, Malhotra JD, Mattei LN, Braun PE, Schachner M et al. Tenascin-R is a functional modulator of sodium channel beta subunits. J Biol Chem 1999; 274: 26511–26517.
Malhotra JD, Kazen-Gillespie K, Hortsch M, Isom LL . Sodium channel beta subunits mediate homophilic cell adhesion and recruit ankyrin to points of cell-cell contact. J Biol Chem 2000; 275: 11383–11388.
Davis TH, Chen C, Isom LL . Sodium channel beta 1 subunits promote neurite outgrowth in cerebellar granule neurons. J Biol Chem 2004; 279: 51424–51432.
Kazarinova-Noyes K, Malhotra JD, McEwan DP, Mattei LN, Berglund EO, Ranscht B et al. Contactin associates with Na+ channels and increases their functional expression. J Neurosci 2001; 21: 7517–7525.
Ratcliffe CF, Westenbroek RE, Curtis R, Catterall WA . Sodium channel beta1 and beta3 subunits associate with neurofascin through their extracellular immunoglobulin-like domain. J Cell Biol 2001; 154: 427–434.
Srinivasan J, Schachner M, Catterall WA . Interaction of voltage-gated sodium channels with the extracellular matrix molecules tenascin-C and tenascin-R. Proc Nat Acad Sci USA 1998; 95: 15753–15757.
Okegawa T, Li Y, Pong RC, Hsieh JT . Cell adhesion proteins as tumor suppressors. J Urol 2002; 167: 1836–1843.
Johnson D, Montpetit ML, Stocker PJ, Bennett ES . The sialic acid component of the beta1 subunit modulates voltage-gated sodium channel function. J Biol Chem 2004; 279: 44303–44310.
Chevrier P, Vijayaragavan K, Chahine M . Differential modulation of Nav1.7 and Nav1.8 peripheral nerve sodium channels by the local anesthetic lidocaine. Br J Pharmacol 2004; 142: 576–584.
Berthon P, Dimitrov T, Stower M, Cussenot O, Maitland NJ . A microdissection approach to detect molecular markers during progression of prostate cancer. Br J Cancer 1995; 72: 946–951.
Moriyama N, Yamaguchi T, Takeuchi T, Sakamoto E, Ueki T, Tsujimoto G et al. Semi-quantitative evaluation of α1A-adrenoceptor subtype mRNA in human hypertrophied and non-hypertrophied prostates: regional comparison. Life Sci 1999; 64: 201–210.
Hudson DL, Guy AT, Fry P, O'Hare MJ, Watt FM, Masters JR . Epithelial cell differentiation pathways in the human prostate: identification of intermediate phenotypes by keratin expression. J Hist Cytochem 2001; 49: 271–278.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001; 25: 402–408.
De Kok JB, Verhaegh GW, Roelofs RW, Hessels D, Kiemeney LA, Aalders TW et al. DD3(PCA3), a very sensitive and specific marker to detect prostate tumors. Cancer Res 2002; 62: 2695–2698.
DePrimo SE, Diehn M, Nelson JB, Reiter RE, Matese J, Fero M et al. Transcriptional programs activated by exposure of human prostate cancer cells to androgen. Genome Biol 2002; 3: RESEARCH0032.1–0032.12.
Corcoran NM, Costello AJ . Interleukin-6: minor player or starring role in the development of hormone-refractory prostate cancer? BJU Int 2003; 91: 545–553.
Jo T, Nagata T, Iida H, Imuta H, Iwasawa K, Ma J et al. Voltage-gated sodium channel expressed in cultured human smooth muscle cells: involvement of SCN9A. FEBS Lett 2004; 567: 339–343.
Gosling M, Harley SL, Turner RJ, Carey N, Powell JT . Human saphenous vein endothelial cells express a tetrodotoxin-resistant, voltage-gated sodium current. J Biol Chem 1998; 273: 21084–21090.
Estacion M . Characterization of ion channels seen in subconfluent human dermal fibroblasts. J Physiol 1991; 436: 579–601.
Fraser SP, Diss JK, Lloyd LJ, Pani F, Chioni AM, George AJ et al. T-lymphocyte invasiveness: control by voltage-gated Na+ channel activity. FEBS Lett 2004; 569: 191–194.
Hinson AW, Gu XQ, Dib-Hajj S, Black JA, Waxman SG . Schwann cells modulate sodium channel expression in spinal sensory neurons in vitro. Glia 1997; 21: 339–349.
Shcherbatko A, Ono F, Mandel G, Brehm P . Voltage-dependent sodium channel function is regulated through membrane mechanics. Biophys J 1999; 77: 1945–1959.
Vijayaragavan K, O'Leary ME, Chahine M . Gating properties of Na(v)1.7 and Na(v)1.8 peripheral nerve sodium channels. J Neurosci 2001; 21: 7909–7918.
Bennett ES, Smith BA, Harper JM . Voltage-gated Na+ channels confer invasive properties to human prostate cancer cells. Pflugers Arch 2004; 447: 908–914.
Lenkowski PW, Shah BS, Dinn AE, Lee K, Patel MK . Lidocaine block of neonatal Nav1.3 is differentially modulated by co-expression of beta1 and beta3 subunits. Eur J Pharmacol 2003; 467: 23–30.
Adachi K, Toyota M, Sasaki Y, Yamashita T, Ishida S, Ohe-Toyota M et al. Identification of SCN3B as a novel p53-inducible proapoptotic gene. Oncogene 2004; 23: 7791–7798.
Van Bokhoven A, Varella-Garcia M, Korch C, Johannes WU, Smith EE, Miller HL et al. Molecular characterization of human prostate carcinoma cell lines. Prostate 2003; 57: 205–225.
Culig Z, Klocker H, Bartsch G, Hobisch A . Androgen receptors in prostate cancer. Endocr Relat Cancer 2002; 9: 155–170.
Wada A . Roles of voltage-dependent sodium channels in neuronal development, pain, and neurodegeneration. J Pharmacol Sci 2006; 102: 253–268.
Acknowledgements
This study was supported by the Pro Cancer Research Fund (PCRF). We thank Dr Rory Curtis (Elixir Pharmaceuticals) for his comments on the manuscript and Petros Andrikopoulos for his assistance with the real-time PCR experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Diss, J., Fraser, S., Walker, M. et al. β-Subunits of voltage-gated sodium channels in human prostate cancer: quantitative in vitro and in vivo analyses of mRNA expression. Prostate Cancer Prostatic Dis 11, 325–333 (2008). https://doi.org/10.1038/sj.pcan.4501012
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.pcan.4501012
Keywords
This article is cited by
-
Contribution of voltage-gated sodium channel β-subunits to cervical cancer cells metastatic behavior
Cancer Cell International (2019)
-
The role of REST and HDAC2 in epigenetic dysregulation of Nav1.5 and nNav1.5 expression in breast cancer
Cancer Cell International (2017)
-
Analgesic-antitumor peptide inhibits the migration and invasion of HepG2 cells by an upregulated VGSC β1 subunit
Tumor Biology (2016)
-
Therapeutic potential for phenytoin: targeting Nav1.5 sodium channels to reduce migration and invasion in metastatic breast cancer
Breast Cancer Research and Treatment (2012)