Abstract
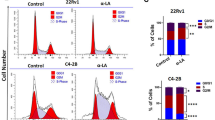
Zoledronic acid (ZA) has been shown to inhibit prostate tumor growth in vitro and have beneficial effects in patients with advanced prostate cancer (CaP). The aim of this study was to determine whether ZA exhibits direct anti-tumor effects on CaP cells in vivo. To distinguish the effects of inhibition of osteolysis and direct anti-tumor activity of ZA in vivo, we compared the results of treatment with ZA and osteoprotegerin (Fc-OPG), which inhibits osteolysis, but without significant direct anti-tumor effects. In vitro Fc-OPG had no significant effects on C4-2 proliferation, whereas ZA decreased proliferation. However, both agents decreased tumor growth in bone. Moreover, both increased bone volume and prevented the overall decreases in BMD associated with growth of C4-2 cells in bone. Our study provides novel and significant observations that the in vivo effects of ZA are consistent with indirect effects mediated by osteoclasts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Clarke NW, McClure J, George NJR . Morphometric evidence for bone resorption and replacement in prostate cancer. Br J Urol 1991; 68: 74–80.
Nemeth JA et al. Matrix metalloproteinase activity, bone matrix turnover, and tumor cell proliferation in prostate cancer bone metastasis. J Natl Cancer Inst 2002; 94: 17–25.
Zhang J et al. Osteoprotegerin inhibits prostate cancer-induced osteoclastogenesis and prevents prostate tumor growth in the bone. J Clin Invest 2001; 107: 1235–1244.
Corey E et al. Zoledronic acid exhibits inhibitory effects on osteoblastic and osteolytic metastases of prostate cancer. Clin Cancer Res 2003; 9: 295–306.
Lee MV et al. Bisphosphonate treatment inhibits the growth of prostate cancer cells. Cancer Res 2001; 61: 2602–2608.
Lee YP et al. Use of zoledronate to treat osteoblastic versus osteolytic lesions in a severe-combined-immunodeficient mouse model. Cancer Res 2002; 62: 5564–5570.
Diel IJ et al. Reduction in new metastases in breast cancer with adjuvant clodronate treatment. N Engl J Med 1998; 339: 357–363.
Hiraga T et al. Zoledronic acid inhibits visceral metastases in the 4T1/luc mouse breast cancer model. Clin Cancer Res 2004; 10: 4559–4567.
Major P et al. Zoledronic acid is superior to pamidronate in the treatment of hypercalcemia of malignancy: a pooled analysis of two randomized, controlled clinical trials. J Clin Oncol 2001; 19: 558–567.
Fleisch H . Zoledronic acid: an evolving role in the treatment of cancer patients with bone disease. Semin Oncol 2001; 28: 45–47.
van der Pluijm G et al. Bisphosphonates inhibit the adhesion of breast cancer cells to bone matrices in vitro. J Clin Invest 1996; 98: 698–705.
Peyruchaud O et al. Early detection of bone metastases in a murine model using fluorescent human breast cancer cells: application to the use of the bisphosphonate zoledronic acid in the treatment of osteolytic lesions. J Bone Miner Res 2001; 16: 2027–2034.
Mundy GR, Yoneda T, Hiraga T . Preclinical studies with zoledronic acid and other bisphosphonates: impact on the bone microenvironment. Semin Oncol 2001; 28: 35–44.
Simonet WS et al. Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 1997; 89: 309–319.
Bucay N et al. Osteoprotegerin-deficient mice develop early onset osteoporosis and arterial calcification. Genes Dev 1998; 12: 1260–1268.
Mizuno A et al. Severe osteoporosis in mice lacking osteoclastogenesis inhibitory factor/osteoprotegerin. Biochem Biophys Res Commun 1998; 247: 610–615.
Hofbauer LC, Heufelder AE . Role of receptor activator of nuclear factor-kappaB ligand and osteoprotegerin in bone cell biology. J Mol Med 2001; 79: 243–253.
Morony S et al. Osteoprotegerin inhibits osteolysis and decreases skeletal tumor burden in syngeneic and nude mouse models of experimental bone metastasis. Cancer Res 2001; 61: 4432–4436.
Yonou H et al. Osteoprotegerin/osteoclastogenesis inhibitory factor decreases human prostate cancer burden in human adult bone implanted into nonobese diabetic/severe combined immunodeficient mice. Cancer Res 2003; 63: 2096–2102.
Morony S et al. A chimeric form of osteoprotegerin inhibits hypercalcemia and bone resorption induced by IL-1beta, TNF-alpha, PTH, PTHrP, and 1, 25(OH)2D3. J Bone Miner Res 1999; 14: 1478–1485.
Corey E et al. Establishment and characterization of osseous prostate cancer models: intra-tibial injection of human prostate cancer cells. Prostate 2002; 52: 20–33.
Pfitzenmaier J et al. Characterization of C4-2 prostate cancer bone metastases and their response to castration. J Bone Miner Res 2003; 18: 1882–1888.
Sato M et al. Bisphosphonate action. Alendronate localization in rat bone and effects on osteoclast ultrastructure. J Clin Invest 1991; 88: 2095–2105.
Pan B et al. The nitrogen-containing bisphosphonate, zoledronic acid, influences RANKL expression in human osteoblast-like cells by activating TNF-alpha converting enzyme (TACE). J Bone Miner Res 2004; 19: 147–154.
Viereck V et al. Bisphosphonates pamidronate and zoledronic acid stimulate osteoprotegerin production by primary human osteoblasts. Biochem Biophys Res Commun 2002; 291: 680–686.
Kiefer JA et al. The effect of osteoprotegerin administration on the intra-tibial growth of the osteoblastic LuCaP 23.1 prostate cancer xenograft. Clin Exp Metastasis 2004; 21: 381–387.
Acknowledgements
This research was supported by US Army Medical Research Material Command Prostate Cancer Research Program DAMD17-00-1-0529, National Institute of Health PO1CA85859-01A, and the Richard M Lucas Foundation. ZA was kindly provided by Pharma Novartis AG, Basel, Switzerland. Fc-OPG was kindly provided by Amgen, Inc, Thousand Oaks, CA, and PSA assay materials by Abbott Laboratories, Abbott Park, IL. We thank Dr M Corey for useful comments and editorial help, and Mr A Odman for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Quinn, J., Brown, L., Zhang, J. et al. Comparison of Fc-osteoprotegerin and zoledronic acid activities suggests that zoledronic acid inhibits prostate cancer in bone by indirect mechanisms. Prostate Cancer Prostatic Dis 8, 253–259 (2005). https://doi.org/10.1038/sj.pcan.4500815
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.pcan.4500815
Keywords
This article is cited by
-
The association between RANKL and Osteoprotegerin gene polymorphisms with breast cancer
Molecular and Cellular Biochemistry (2015)
-
Expression of osteoprotegerin from a replicating adenovirus inhibits the progression of prostate cancer bone metastases in a murine model
Laboratory Investigation (2013)
-
Bench to bedside: elucidation of the OPG–RANK–RANKL pathway and the development of denosumab
Nature Reviews Drug Discovery (2012)
-
Host-derived RANKL is responsible for osteolysis in a C4-2 human prostate cancer xenograft model of experimental bone metastases
BMC Cancer (2007)
-
Effect of zoledronic acid on the doxycycline-induced decrease in tumour burden in a bone metastasis model of human breast cancer
British Journal of Cancer (2007)