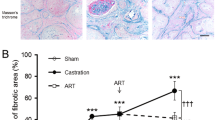
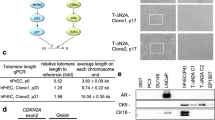
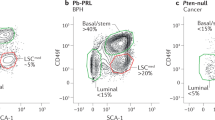
Abstract
We established explant primary cultures in order to study the growth and hormone responsiveness, and the differentiation process of prostatic epithelial cells. Cell outgrowth was achieved from explant tissue by using a new DU145-cell-conditioned medium and special plastic coverslips. To define the present model, proliferation assays were tested by [3H]thymidine assay and planimetric analysis. Cells were analyzed using immunocytochemistry, light, phase contrast and electron microscopy, polymerase chain reaction, telomerase ELISA and immunoassay (PSA). Morphology and electron microscopy revealed typical epithelial differentiation. Immunocytochemistry showed the content of basal and secretory epithelial cells, endocrine paracrine cells and a high level of proliferation. With increasing culture time, mature epithelial differentiation (PSA) increases and the initial increase of α-smooth muscle actin (α-SMA) decreases again. After further passaging, α-SMA expression is no longer detected and PSA expression decreases. Furthermore, epithelial cells showed both androgen responsiveness and androgen receptor expression. These findings show the presence of epithelial cells in a process of differentiation with endocrine paracrine cells and a high level of proliferation. This model may maintain the cellular and functional properties more closely related to the human prostate and may provide a valuable tool for studying stem cells and differentiation characteristics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout











Similar content being viewed by others
References
Peehl DM, Wong ST, Bazinet M, Stamey TA . In vitro studies of human prostatic epithelial cells: attempts to identify distinguishing features of malignant cells. Growth Factors 1989; 1: 237–250.
Merchant DJ . Model systems for the study of prostatic cancer. Oncology 1977; 34: 100–101.
Horoszewicz JS, Leong SS, Kawinski E, Karr JP, Rosenthal H, Chu TM, Mirand EA, Murphy GP . LNCaP model of human prostatic carcinoma. Cancer Res 1983; 43: 1809–1818.
Kaighn ME, Narayan KS, Ohnuki Y, Lechner JF, Jones LW . Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Invest Urol 1979; 17: 16–23.
Stone KR, Mickey DD, Wunderli H, Mickey GH, Paulson DF . Isolation of a human prostatic carcinoma cell line (DU145). Int J Cancer 1978; 21: 274–281.
Iizumi T, Yazaki T, Kanoh S, Kondo I, Koiso K . Establishment of a new prostatic carcinoma cell line (TSU-Pr1). J Urol 1987; 137: 1304–1306.
McKeehan WL, Adams PS, Fast D . Different hormonal requirements for androgen-independent growth of normal and tumor epithelial cells from rat prostate. In Vitro Cell DevBiol 1987; 23: 147–152.
Berthon P, Cussenot O, Hopwood L, LeDuc A, Maitland NJ . Functional expression of SV40 in normal human prostate epithelium and fibroblast cells differentiation pattern of nontumorigenic cell lines. Int J Oncol 1995; 6: 333–343.
Peehl DM . Serial culture of adult human prostatic epithelial cells. J Tissue Culture Methods 1985; 9: 53–60.
Grant ES, Batchelor KW, Habib FK . Androgen independence of primary epithelial cultures of the prostate is associated with a down-regulation of androgen receptor gene expression. Prostate 1996; 29: 339–349.
Chaproniere DM, McKeehan WL . Serial culture of single adult human prostatic epithelial cells in serum-free medium containing low calcium and a new growth factor from bovine brain. Cancer Res 1986; 46: 819–824.
Peehl DM, Leung K, Wong ST . Keratin expression, a measure of phenotypic modulation of human prostate epithelial cells by growth inhibitory factors. Cell Tissue Res 1994; 277: 11–18.
Peehl DM, Wong ST, Stamey TA . Clonal growth characteristics of adult human prostate epithelial cells. In Vitro Cell Dev Biol 1988; 24: 530–536.
Cunha GR, Hayward SW, Dahiya R, Foster BA . Smooth muscle–epithelial interactions in normal and neoplastic prostatic development. Acta Anatomica 1996; 155: 63–72.
Hayward SW, Cunha GR, Dahiya R . Normal development and carcinogenesis of the prostate: a unifying hypothesis. Ann NY Acad Sci 1996; 784: 50–62.
Chung LW, Gleave ME, Hsieh JT, Hong SJ, Zhau HE . Reciprocal mesenchymal–epithelial interaction affecting prostate tumor growth and hormonal responsiveness. Cancer Surv 1991; 11: 91–121.
Chung LW, Chang SM, Bell C, Zhau HE, Ro JY, Eschenbach AC . Co-inoculation of tumorigenetic rat prostate mesenchymal cells with non-tumorigenic epithelial cells results in the development of carcinosarcoma in syngeneic and athymic animals. Int J Cancer 1989; 43: 1179–1187.
Olumi AF, Grossfeld GD, Hayward SW, Carroll PR, Tlsty TD, Cunha GR . Carcinoma associated fibroblasts direct tumor progression of initiated human prostate epithelium. Cancer Res 1999; 59: 5002–5011.
Evans GS, Chandler JA . Cell proliferation studies in rat prostate I. The proliferative role of basal and secretory epithelial cells during normal growth. Prostate 1987; 10: 163–178.
Aumuller G . Morphologic and endocrine aspects of prostatic function. Prostate 1983; 4: 195–214.
Bonkhoff H, Remberger K . Differentiation pathways and histogenetic aspects of normal and abnormal prostatic growth: a stem cell model. Prostate 1996; 28: 98–106.
Heatfield BM, Sanefuji H, Trump BF . Studies on carcinogenesis of human prostate. III. Long term explant culture of normal prostate and benign prostatic hyperplasia: transmission and scanning electron microscopy. J Natl Cancer Inst 1982; 69: 757–766.
Bonkhoff H, Stein U, Remberger K . The proliferative function of basal cells in the normal and hyperplastic human prostate. Prostate 1994; 24: 114–118.
Nevalainen MT, Harkonen PL, Valve EM, Ping W, Nurmi M, Martikainen PM . Hormone regulation of human prostate in organ culture. Cancer Res 1993; 53: 5199–5207.
Ma Y, Fujiyama C, Masaki Z, Sugihara H . Reconstruction of prostatic acinus-like structure from ventral and dorsolateral prostatic epithelial cells of the rat in three-dimensional collagen gel matrix culture. J Urol 1997; 157: 1025–1031.
Martikainen PM, Harkonen PL, Vanhala T, Makela SI, Viljanen M, Suominen J . Multihormonal control of synthesis and secretion of prostatein in cultured rat ventral prostate. Endocrinology 1987; 121: 604–611.
Chromczynski P, Sacchi N . Single step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal Biochem 1987; 162: 156–159.
Taplin ME, Bubley GJ, Shuster TD, Frantz ME, Spooner AE, Ogata GK, Keer HN, Balk SP . Mutation of the androgen-receptor gene in metastatic androgen-independent prostate cancer. N Engl J Med 1995; 332: 1393–1398.
Svitkina TM, Veskhorsky AB, Bonisy CG . Improved procedures for electron microscopic visualization of the cytoskeleton of cultured cells. J Struct Biol 1995; 115: 290–303.
Peehl DM, Wong ST, Terris MK, Stamey TA . Culture of prostatic epithelial cells from ultrasound-guided needle biopsies. Prostate 1991; 19: 141–147.
Merchant DJ . Primary explant culture of human prostate tissue: a model for the study of prostate physiology and pathology. Prostate 1990; 16: 103–126.
Brothman AR, Lesho LJ, Somers KD, Schellhammer PF, Ladaga LE, Merchant DJ . Cytogenetic analysis of four primary prostatic cultures. Cancer Genet Cytogenet 1989; 37: 241–248.
Sherwood ER, Van Dongen JL, Wood CG, Liao S, Kozlowski JM, Lee C . Epidermal growth factor receptor activation in androgen-independent but not androgen-stimulated growth of human prostatic carcinoma cells. Br J Cancer 1998; 77: 855–861.
Geldof AA, De Kleijn MA, Rao BR, Newling DW . Nerve growth factor stimulates in vitro invasive capacity of DU 145 human prostatic cancer cells. J Cancer Res Clin Oncol 1997; 123: 107–112.
Okamoto M, Lee C, Oyasu R . Interleukin-6 as a paracrine and autocrine growth factor in human prostatic carcinoma cells in vitro. Cancer Res 1997; 57: 141–146.
Rokhlin OW, Griebling TL, Karassina NV, Raines MA, Cohen MB . Human prostate carcinoma cell lines secrete GM-CSF and express GM-CSF-receptor on their cell surface. Anticancer Res 1996; 16: 557–563.
Tillotson JK, Rose DP . Endogenous secretion of epidermal growth factor peptides stimulates growth of DU 145 prostate cancer cells. Cancer Lett 1991; 60: 109–112.
Krill D, Shuman M, Thompson MT, Becich MJ, Strom SC . A simple method for the isolation and culture of epithelial and stromal cells from benign and neoplastic prostates. Urology 1997; 49: 981–988.
Peehl DM, Wong ST, Rubin JS . KGF and EGF differentially regulate the phenotype of prostatic epithelial cells. Growth Regul 1996; 6: 22–31.
McKeehan WL, Adams PS, Rosser MP . Direct mitogenic effects of insulin, epidermal growth factor, glucocorticoid, cholera toxin, unknown pituitary factors and possibly prolactin, but not androgen, on normal rat prostate epthelial cells in serum-free, primary cell culture. Cancer Res 1984; 44: 1998–2010.
Planz B, Kirley SD, Wang QF, Tabatabaei S, Aretz HT, McDougal WS . Characterization of a stromal cell model of the human benign and malignant prostate from explant culture. J Urol 1999; 161: 1329–1336.
Cunha GR, Sugimura Y, Forster B, Rubin JS, Aaronson SA . The role of growth factors in the development and growth of the prostate and seminal vesicle. Biomed Pharmacother 1994; 48 (Suppl 1): 9s–17s.
Nemeth JA, Lee C . Prostatic ductal system in rats: regional variation in stromal organization. Prostate 1996; 28: 124–128.
Ronnov-Jessen L, van Deurs B, Cells JE, Petersen OW . Smooth muscle differentiation in cultured human breast gland stromal cells. Lab Invest 1990; 63: 532–543.
Sherwood ER, Berg LA, Mitchell NJ, McNeal JE, Kozlowski JM, Lee C . Differential cytokeratin expression in normal, hyperplastic and malignant epithelial cells form human prostate. J Urol 1990; 43: 167–171.
Hayward SW, Del Buono R, Deshpande N, Hall PA . A functional model of adult human prostate epithelium. The role of androgens and stroma in architectual organisation and the maintenance of differentiated secretory function. J Cell Sci 1992; 102: 361–372.
Brawer MK, Peehl DM, Stamey TA, Bostwick DG . Keratin immunoreactivity in the benign and neoplastic human prostate. Cancer Res 1985; 45: 3663–3667.
Kooistra A, Elissen NM, Konig JJ, Vermey M, van der Kwast TH, Romijn JC, Schroder FH . Immunocytochemical characterization of explant cultures of human prostatic stromal cells. Prostate 1995; 27: 42–49.
Ben-Ze’ev A . Differential control of cytokeratins and vimentin synthesis by cell–cell contact and cell spreading in cultured epithelial cells. J Cell Biol 1984; 99: 1424–1433.
di Sant’Agnese PA . Neuroendocrine differentiation in carcinoma of the prostate. Diagnostic, prognostic, and therapeutic implications. Cancer 1992; 70: 254–268.
Hall PA, Watt FM . Stem cells: the generation and maintenance of cell diversity. Development 1989; 106: 619–633.
Planz B, Aretz T, Wang QF, Tabatabaei S, Kirley SD, Lin C-W, McDougal WS . Immunolocalization of the keratinocyte growth factor in benign and neoplastic human prostate and its relation to androgen receptor. Prostate 1999; 41: 233–242.
Puy L, MacLusky NJ, Becker L, Karsan N, Trachtenberg J, Brown TJ . Immunocytochemical detection of androgen receptor in human temporal cortex: characterization and application of polyclonal androgen receptor antibodies in frozen and paraffin-embedded tissues. J Steroid Biochem Mol Biol 1995; 55: 197–209.
Sar M, Lubahn DB, French FS, Wilson EM . Immunohistochemical localization of the androgen receptor in rat and human tissues. Endocrinology 1990; 127: 3180–3186.
Bonkhoff H, Wernert N, Dhom G, Remberger K . Relation of endocrine-paracrine cells to cell proliferation in normal, hyperplastic and neoplastic human prostate. Prostate 1991; 19: 91–98.
Acknowledgements
We thank Dr GR Dickersin for helpful comments in the electron microscopic evaluation and P Sherwood for technical support in electron microscopy. This work was supported by Urology Department Funds, Massachusetts General Hospital, Boston, MA; National Cooperative Program for Infertility Research Center at the Massachusetts General Hospital (HD29164); and The German Research Foundation (DFG PL-2).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Planz, B., Tabatabaei, S., Kirley, S. et al. Studies on the differentiation pathway and growth characteristics of epithelial culture cells of the human prostate. Prostate Cancer Prostatic Dis 7, 73–83 (2004). https://doi.org/10.1038/sj.pcan.4500704
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.pcan.4500704
Keywords
This article is cited by
-
Progenitor cells are responsible for formation primary epithelial cultures in the prostate epithelial model
International Urology and Nephrology (2007)
-
Down regulation of PSA by C/EBPα is associated with loss of AR expression and inhibition of PSA promoter activity in the LNCaP cell Line
BMC Cancer (2006)