Abstract
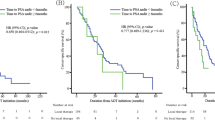
We conducted a study in order to characterize changes after withdrawal of androgen ablation (AA) for prostate cancer. AA was withdrawn in 38 Japanese patients with prostate cancer who had undergone this therapy for various periods. Patients were stratified into those who had undergone AA for less than 24 months (Group 1, n=12) and those with longer periods of AA (Group 2, n=26). Serial changes in hormones and prostate-specific antigen (PSA) were prospectively monitored following cessation of AA. The median durations of AA in the two groups were 8.5 and 54.5 months, respectively. Levels of total testosterone (T), luteinizing hormone and PSA increased significantly with time. At the end of 2 y, 30/38 patients (78.9%) had T levels above 50 ng/dl and 19/38 (50%) had levels above 320 ng/dl. Patients in Group 2 required significantly longer duration for T recovery. Complete T recovery is not always accompanied by rising PSA. Recovery of T levels is often slow following cessation of prolonged AA. Expression of PSA after AA is often variable and unpredictable. Thus, interpretation of outcomes in clinical trials incorporating AA needs caution and careful consideration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Huggins C, Hodges CV . Studies on prostatic cancer. I. The effect of castration, of estrogen, and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res 1941; 1: 293–297.
Egawa S et al. A pilot study of intermittent androgen ablation in advanced prostate cancer in Japanese men. Jpn J Clin Oncol 2000; 30: 21–26.
Akakura K et al. Effects of intermittent androgen suppression on androgen-dependent tumours: apoptosis and serum prostate specific antigen. Cancer 1993; 71: 2782–2790.
Gleave M, Bruchovsky N, Goldenberg SL, Rennie P . Intermittent androgen suppression: rationale and clinical experience. In: Schroder T-H. (ed). Recent Advances in Prostate Cancer and BPH. Parthenon: New York, 1997, pp 109–120.
Moul JW . Contemporary hormonal management of advanced prostate cancer. Oncology 1998; 12: 499–505.
Pickles T et al. Testosterone recovery following prolonged adjuvant androgen ablation for prostate carcinoma. Cancer 2002; 94: 362–367.
Bolla M et al. Improved survival in patients with locally advanced prostate cancer treated with radiotherapy and goserelin. N Engl J Med 1997; 337: 295–300.
Oefelein MG . Serum testosterone-based luteinizing hormone-releasing hormone agonist redosing schedule for chronic androgen ablation: a phase I assessment. Urology 1999; 54: 694–699.
Hall MC et al. Prospective determination of the hormonal response after cessation of luteinizing hormone-releasing hormone agonist treatment in patients with prostate cancer. Urology 1999; 53: 898–903.
Nejat RJ et al. A prospective analysis of time to normalization of serum testosterone after withdrawal of androgen deprivation therapy. J Urol 2000; 164: 1891–1894.
Oefelein MG . Time to normalization of serum testosterone after 3-month luteinizing hormone-releasing hormone agonist administered in the neoadjuvant setting: implications for dosing schedule and neoadjuvant study consideration. J Urol 1998; 160: 1685–1688.
Kuber W, Viehberger G, Zeillinger R, Spona J . Effects of the duration of therapy with the LHRH agonist D-ser (BUT)6 Azgly10-LHRH (ICI 118-630) on the steroid hormone content and the morphology of human testicular tissue in the treatment of patients with advanced prostate cancer. Urol Res 1991; 19: 19–24.
Lunglmayr G et al. Effects of long term GnRH analogue treatment on hormone levels and spermatogenesis in patients with carcinoma of the prostate. Urol Res 1988; 16: 315–319.
Tomic B, Bergman B . Hormonal effects of cessation of estrogen treatment for prostatic carcinoma. J Urol 1987; 138: 801–803.
Hoffman MA, DeWolf WC, Morgentaler A . Is low serum free testosterone a marker for high grade prostate cancer? J Urol 2000; 163: 824–827.
Vermeulen A, Kaufman JM . Ageing of the hypothalamo-pituitary-testicular axis in men. Horm Res 1995; 43: 25–28.
Acknowledgements
We thank WA Thomasson, PhD, for expert editorial assistance. This work was supported in part by a Grant from the Ministry of Health and Welfare of Japan and the Foundation for Promotion of Cancer Research in Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Egawa, S., Okusa, H., Matsumoto, K. et al. Changes in prostate-specific antigen and hormone levels following withdrawal of prolonged androgen ablation for prostate cancer. Prostate Cancer Prostatic Dis 6, 245–249 (2003). https://doi.org/10.1038/sj.pcan.4500675
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.pcan.4500675
Keywords
This article is cited by
-
Accuracy of serum luteinizing hormone and serum testosterone measurements to assess the efficacy of medical castration in prostate cancer patients
Journal of Biomedical Science (2017)