Abstract
Omi/HtrA2 is a mitochondrial serine protease that is released into the cytosol during apoptosis and promotes cytochrome c (Cyt c)dependent caspase activation by neutralizing inhibitor of apoptosis proteins (IAPs) via its IAP-binding motif. The protease activity of Omi/HtrA2 also contributes to the progression of both apoptosis and caspase-independent cell death. In this study, we found that wild-type Omi/HtrA2 is more effective at caspase activation than a catalytically inactive mutant of Omi/HtrA2 in response to apoptotic stimuli, such as UV irradiation or tumor necrosis factor. Although similar levels of Omi/HtrA2 expression, XIAP-binding activity, and Omi/HtrA2 mitochondrial release were observed among cells transfected with catalytically inactive and wild-type Omi/HtrA2 protein, XIAP protein expression after UV irradiation was significantly reduced in cells transfected with wild-type Omi/HtrA2. Recombinant Omi/HtrA2 was observed to catalytically cleave IAPs and to inactivate XIAP in vitro, suggesting that the protease activity of Omi/HtrA2 might be responsible for its IAP-inhibiting activity. Extramitochondrial expression of Omi/HtrA2 indirectly induced permeabilization of the outer mitochondrial membrane and subsequent Cyt c-dependent caspase activation in HeLa cells. These results indicate that protease activity of Omi/HtrA2 promotes caspase activation through multiple pathways.
Similar content being viewed by others
Introduction
Apoptosis is a physiological cell suicide program critical to the development and homeostasis of all animals.1 Abnormal inhibition of apoptosis is a hallmark of cancer and autoimmune disease, whereas excessive cell death has been implicated in a number of neurodegenerative disorders.2 The caspases, a family of intracellular cysteine proteases, are the central executioners of apoptosis.3 Effector caspases, such as caspase-3 and -7, are activated by initiator caspases, such as caspase-9, through proteolytic cleavage. Once activated, effector caspases are responsible for the proteolytic cleavage of a diverse array of structural and regulatory proteins, resulting in an apoptotic phenotype.3
Inhibitor of apoptosis proteins (IAPs), originally found in baculoviruses, are conserved in a number of species, ranging from insects to humans, and play a role in regulating apoptosis.4,5 Several members of the human IAP family of proteins, including X chromosome-linked IAP (XIAP), cellular IAP (c-IAP1) and c-IAP2, are potent direct inhibitors of caspase-3, -7, and -9.6,7,8,9 Among these, XIAP is the most potent inhibitor of caspases and apoptosis.5,10
Two IAP inhibitors, second mitochondria-derived activator of caspase (Smac)/direct IAP binding protein with low pI (DIABLO) and Omi/ high temperature requirement A2 (HtrA2), have been identified.11,12,13,14,15,16,17 Both of these proteins have mitochondrial localization signals (MLS) at their N-termini and are transported to the mitochondria where their MLS sequences are cleaved and the mature proteins are generated. The mature proteins have IAP-binding motifs (IBMs) at their N-termini, which enable them to bind IAPs and eliminate their caspase-inhibitory activity.18 In response to various apoptotic stimuli, these mature proteins are released from the mitochondria into the cytosol, where they interact with and inhibit IAPs. In this regard, Smac/DIABLO and Omi/HtrA2 resemble each other. However, unlike Smac/DIABLO, Omi/HtrA2 has protease activity. When expressed extramitochondrially, Omi/HtrA2 induces caspase-independent cell death. This is thought to depend on the protease activity of Omi/HtrA2 alone, independent of IAP inhibition, because an Omi/HtrA2 mutant lacking the IBM has also shown to induce caspase-independent cell death.13,14 On the other hand, there is evidence to suggest that both the serine protease activity and IAP-binding activity of Omi/HtrA2 contribute to its proapoptotic function.14,15
Recently, Smac/DIABLO-deficient mice have been engineered.19 Several different Smac/DIABLO-deficient cells respond normally to various apoptotic stimuli, suggesting the existence of a redundant molecule or molecules compensating for a loss of Smac/DIABLO function.19 In contrast, although Omi/HtrA2-deficient mice have not been reported, Omi/HtrA2-knockdown cells, in which antisense Omi/HtrA2 RNA was expressed, or to which synthetic double-stranded small interfering RNA (siRNA) oligonucleotides were introduced, have shown to be less susceptible to induction of apoptosis.14,16 These data suggest a nonredundant essential function of Omi/HtrA2 in the induction of apoptosis.
In this study, we provide evidence that the protease activity of Omi/HtrA2 contributes to its ability to potentiate caspase activation and apoptosis via at least two different mechanisms. Omi/HtrA2 cleaves and/or degrades IAPs and an unidentified substrate(s), resulting in inactivation of IAPs and permeabilization of the outer mitochondrial membrane followed by Cyt c-dependent caspase activation, respectively. Our data suggest that the protease activity of Omi/HtrA2 is important for its ability to potentiate caspase activation and the induction of apoptosis.
Results and Discussion
Catalytically inactive Omi/HtrA2 mutant is less effective than wild-type Omi/HtrA2 at potentiating caspase activation and apoptosis in HeLa cells
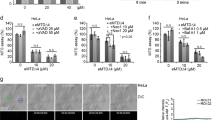
Both IAP-binding and serine protease activity of Omi/HtrA2 contributes to its proapoptotic function. Verhagen et al.15 have shown that mutation of either the catalytic serine residue, or a critical alanine residue required for XIAP binding, reduces, but does not eliminate, the pro-apoptotic activity of Omi/HtrA2, whereas mutation of both sites negates the ability of Omi/HtrA2 to potentiate cell death.15 These results suggest that Omi/HtrA2 protease activity is responsible for potentiation of caspase-3-like DEVDase activation. To verify this possibility, we investigated DEVDase activation in HeLa cells transfected with wild-type Omi/HtrA2, as well as a catalytically inactive mutant of Omi/HtrA2, in response to apoptotic stimuli, such as ultraviolet (UV) irradiation or tumor necrosis factor combined with cycloheximide (TNF/CHX). Following exposure to UV-irradiation, wild-type Omi/HtrA2 (HtrA2 WT) potentiated DEVDase activity to a similar extent as wild-type Smac/DIABLO (Smac WT), and to a greater extent than the catalytically inactive mutant of Omi/HtrA2 (HtrA2 S/A) (Figure 1a). Similar results were obtained in TNF/CHX-treated cells (Figure 1a). The immunoblot and glutathione S-transferase (GST)-pull down assay results revealed that the protein expression and XIAP-binding activity of the mature HtrA2 S/A mutant were similar to that of mature HtrA2 WT (Figure 1b). We also observed that HtrA2 WT potentiated DEVDase activity and apoptosis in the presence of exogenously expressed XIAP, indicating that inhibition of XIAP by HtrA2 WT occurs (Figure 1c and d). Interestingly, the HtrA2 S/A mutant was less effective at inhibiting XIAP-mediated suppression of DEVDase activity and apoptosis (Figure 1c and d). These results are consistent with those of a previous report in which the protease activity of Omi/HtrA2 was observed to enhance its ability to potentiate apoptosis.15
The catalytically inactive HtrA2 mutant is less effective than wild-type HtrA2 at potentiating caspase activation and apoptosis in HeLa cells. (a) HeLa cells transiently transfected with the indicated C-terminally Myc-tagged expression constructs were treated with 20 or 100 mJ/cm2 of UV irradiation (UV 20, UV 100), or 30 ng/ml TNF combined with 1 μg/ml cycloheximide (CHX), for 4 h to induce apoptosis. Cell lysate samples were prepared from adherent and floating cells, after which caspase (DEVDase) activity was measured in each lysate sample, as described in Experimental procedures. The data presented here are the mean+s.e. of triplicate samples. (b) Binding between the indicated proteins and XIAP. Cell lysate samples prepared as outlined in (a) were pulled down with GST-tagged recombinant XIAP protein as described in the Experimental procedures. Whole-cell lysate (WCL) and samples precipitated with XIAP (Pull down) were subjected to immunoblot analysis using antibodies against Myc (IB: Myc) or GST (IB: GST). (c) HeLa cells transfected with the indicated expression constructs together with the N-terminally FLAG-tagged XIAP expression construct were treated with 100 mJ/cm2 of UV irradiation for 4 h to induce apoptosis, after which DEVDase activity was measured. The DEVDase activity of each sample was quantified as a percentage of control DEVDase activity (Ctrl: UV-irradiated mock-transfected cells). (d) HeLa cells transfected with the indicated expression constructs together with the EGFP expression construct were treated with 100 mJ/cm2 of UV. At 5 h after incubation, the number of green apoptotic cells exhibiting membrane blebbing were counted. The results are reported as the percentage of green cells with membrane blebbing in total green cells
Catalytically inactive HtrA2 mutant is also released from the mitochondria
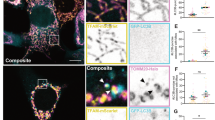
We observed Omi/HtrA2 in the intermembrane space of mitochondria within HeLa cells by immunoelectron microscopy using antibody against Omi/HtrA2 (Figure 2a), leading us to question whether or not mutant HtrA2 S/A is also released from the mitochondria. To test this possibility, we examined the mitochondrial release of mutant HtrA2 S/A in an in vitro system. The release of both HtrA2 WT and HtrA2 S/A mutant protein from isolated HeLa mitochondria transfected with Myc-tagged constructs was induced in a concentration-dependent manner by recombinant truncated Bid (tBid) (Figure 2b). The mitochondrial release of WT and S/A mutant HtrA2 did not significantly differ (Figure 2b). Coimmunostaining with antibodies against Cyt c and Myc-epitope tag, which detects exogenously expressed C-terminally Myc-tagged Omi/HtrA2, revealed that both HtrA2 S/A mutant and HtrA2 WT protein were released from mitochondria along with Cyt c in response to UV irradiation (Figure 2c). These results indicate that the catalytically inactive HtrA2 mutant is also released from the mitochondria, suggesting that the catalytic activity of HtrA2 might enhance caspase activation following its release from the mitochondria.
Catalytically inactive HtrA2 mutant is also released from the mitochondria. (a) Ultrastructural localization of endogenous HtrA2 protein in the mitochondrion of HeLa cells. A magnified image of the area enclosed in the box in the left panel is shown in the right panel. The arrowheads indicate the immunogold particles crossreact with anti-HtrA2 antibody, indicating the presence of HtrA2 protein within the mitochondrial intermembrane space. Scale bar, 0.2 and 0.1 μm. (b) HtrA2 release from isolated mitochondria. Isolated mitochondria were prepared from HeLa cells transfected with C-terminally Myc-tagged wild-type HtrA2 (HtrA2 WT), or its catalytically inactive mutant (HtrA2 S/A), as described in the Experimental procedures. Isolated mitochondria were incubated with the indicated concentrations of recombinant tBid at 25°C for 30 min, after which the reaction mixtures were centrifuged. The supernatant (sup) and precipitated pellet (ppt) were subjected to immunoblot analysis with antibodies against Myc-tag, Cyt c, or Hsp60. (c) Coimmunostaining of wild-type HtrA2 HtrA2 WT, or its catalytically inactive mutant (HtrA2 S/A), with Cyt c was done as described in the Experimental procedures. HeLa cells transfected with C-terminally Myc-tagged HtrA2 expression constructs were treated without or with 100 mJ/cm2 of UV irradiation. After 2.5 h of incubation, the cells were fixed and coimmunostained with anti-Myc polyclonal antibody (green) and anti-Cyt c monoclonal antibody (red). The merged images are shown here
HtrA2 cleaves and inactivates XIAP
The protease activity of Omi/HtrA2 enhances its ability to potentiate caspase-3 like DEVDase activation. HtrA2 WT was more effective than HtrA2 S/A in this regard. We examined the protein expression of XIAP in the same lysate samples discussed in Figure 1a. A significant reduction in endogenous XIAP protein was observed in cells transfected with HtrA2 WT only after treatment of the cells with UV irradiation (Figure 3a, upper panel). Immunoblot analysis with anti-Hsc70 antibody revealed that equivalent amounts of cell lysate were applied to each lane (Figure 3a, lower panel). It has been reported that caspases cleave XIAP.20 However, the observed reduction in XIAP in cells transfected with HtrA2 WT may not have been solely due to caspase-dependent cleavage of XIAP, since a slower reduction of XIAP was observed in cells transfected with Smac WT than with HtrA2 WT (Figure 3a, upper panel), even though they demonstrated comparable DEVDase activity (Figure 1a).
HtrA2 cleaves and inactivates XIAP. (a) HeLa cells transfected with the indicated expression constructs were treated with or without (Ctrl) 100 mJ/cm2 of UV for 4 h. The cell lysate samples were then subjected to immunoblot analysis with antibodies against XIAP or Hsc70. (b) HeLa cells transfected with the indicated expression constructs together with XIAP expression constructs were treated without (–) or with 100 mJ/cm2 of UV irradiation, 30 ng/ml TNF combined with 1 μg/ml CHX, or 0.5 μM of staurosporine (STS), for 4 h. Cell lysate samples were prepared from adherent and floating cells and then subjected to immunoblot analysis with antibodies against XIAP. (c) Recombinant HtrA2 cleaves IAP family proteins in vitro. N-terminally FLAG-tagged proteins were translated in vitro in the presence of 35S-methionine, after which these proteins were purified by immunoprecipitation with anti-FLAG antibody. The purified proteins were incubated in the absence (–) or presence (+) of recombinant HtrA2 protein (100 nM) at 37°C for 5 h. Samples were subjected to SDS-polyacrylamide gel electrophoresis (PAGE) and visualized by autoradiography. (d) Inactivation of XIAP by an HtrA2 mutant that does not bind to XIAP in vitro. HeLa S100 fraction recombinant proteins were prepared as described in the Experimental procedures. A recombinant HtrA2 mutant that does not bind to XIAP (HtrA2ΔN134, 1 μM), and a catalytically inactive form of the mutant (HtrA2ΔN134 S/A, 1 μM), were pre-incubated with GST-XIAP (100 nM) at 37°C for the indicated time periods. Then the HeLa S100 fraction (70 μg), XIAP (30 nM), HtrA2 (300 nM) Cyt c (10 μM), and dATP (1 mM) were mixed on ice in a final volume of 20 μl, followed by incubation at 37°C. After 1 h of incubation, DEVDase activity was measured. The DEVDase activity of each sample was expressed as a percentage of the control value (Ctrl: without XIAP and HtrA2)
We next examined the protein expression of XIAP under similar conditions to those shown in Figure 1c. Exogenously expressed XIAP protein was markedly reduced in cells transfected with HtrA2 WT after the cells were treated with different inducers of apoptosis (Figure 3b). The observed reduction in XIAP expression among cells transfected with HtrA2 WT in the absence of apoptotic stimuli might reflect leakage of exogenously expressed HtrA2 protein from the mitochondria (Figure 3b, upper panel). Although a similar reduction in XIAP protein expression was observed among Smac WT-transfected and mock-transfected cells treated with inducers of apoptosis, significantly less reduction of XIAP expression was observed in HtrA2 S/A-transfected cells (Figure 3b). This suggests that HtrA2 S/A might compete with endogenous HtrA2 or other proteases, thereby inhibiting their ability to reduce XIAP protein expression. These results indicate that HtrA2 protease activity might reduce XIAP expression, suggesting that the release of HtrA2 from mitochondria might catalytically cleave and inactivate IAP family proteins, including XIAP.
To examine whether HtrA2 directly cleaves IAP proteins, we performed an in vitro protease assay using a C-terminally His6-tagged recombinant mature form of HtrA2 (residues 134–458) in the presence of in vitro translated 35S-labeled IAP proteins (Figure 3c). Since the IAP proteins used in this experiment had ubiquitin (Ub) ligase function, they were autoubiquitylated and/or degraded during the prolonged incubation time. To remove components of the Ub–proteasome system from the rabbit reticulocyte lysate, we used N-terminally FLAG-tagged expression constructs in the in vitro translation reaction. After the reaction, the translated protein was immunoprecipitated with anti-FLAG M2 antibody, after which the precipitate was incubated with or without recombinant HtrA2 protein for 5 h at 37°C. The in vitro translated IAPs, including several human IAPs (XIAP, c-IAP1, and c-IAP2) and a Drosophila IAP (DIAP1), were cleaved/degraded by the recombinant HtrA2 protein, whereas in vitro translated Hop (Hsp70/Hsp90 organizing protein), which was used as a negative control, was not (Figure 3c). This result demonstrates that HtrA2 is capable of cleaving/degrading IAP proteins.
We further examined whether the cleaved fragments or degradation products of XIAP were able to inhibit caspase-9 and -3. Thus, we made four different recombinant HtrA2 proteins: HtrA2ΔN133, an enzymatically active protein with an intact IAP-binding motif (IBM); HtrA2ΔN133 S/A, an enzymatically inactive protein with an intact IBM; HtrA2ΔN134, an enzymatically active protein with a disrupted IBM; and HtrA2ΔN134 S/A, an enzymatically inactive protein with a disrupted IBM. The desired characteristics of these proteins were confirmed by in vitro protease assays using beta-casein as the substrate, as well as GST-pull down assays using GST-XIAPΔRING, which lacks a C-terminal RING finger domain (data not shown). We tested the ability of these proteins to potentiate Cyt c-dependent activation of DEVDase in S100 extracts with recombinant XIAP protein (Figure 3d). In the absence of XIAP, Cyt c-dependent activation of DEVDase was observed, whereas activation of DEVDase was inhibited by the addition of XIAP (Figure 3d). XIAP-mediated inhibition of DEVDase activity was attenuated by the addition of enzymatically active and inactive HtrA2 proteins with intact IBM (Figure 3d). Although recombinant HtrA2 proteins lacking intact IBMs did not attenuate XIAP-mediated inhibition of DEVDase activity, pre-incubation with XIAP enable only HtrA2ΔN134 to potentiate the DEVDase activation inhibited by XIAP (Figure 3d). These results indicate that Omi/HtrA2 enzymatically cleaves and inactivates XIAP to promote caspase activation.
The caspase-inhibitory activity of IAPs is subject to multiple regulatory mechanisms. The mitochondrial IAP inhibitors, Smac/DIABLO and Omi/HtrA2, can neutralize IAPs using their IBM. In Drosophila, IAP inhibitors, such as Reaper and HID, promote Ub-dependent degradation of DIAP1.21,22,23 Moreover, Reaper and Grim have been shown to decrease the level of DIAP1 through general suppression of protein translation.21,23 At least three proteases (caspase, proteasome, and calpain) have been observed to cleave and inactivate XIAP.20,24,25 A recent report suggests that Omi/HtrA2 cleaves c-IAP1.26 Interestingly, our results provide evidence that Omi/HtrA2 might inactivate XIAP by catalytic cleavage, as well as by direct binding. During the preparation of this manuscript, Yang et al.27 and Srinivasula et al.28 published reports indicating that Omi/HtrA2 cleaves and inactivates IAPs,27,28 which is consistent with the data presented in this report.
Effect of extramitochondrially expressed Omi/HtrA2 and Smac/DIABLO mutants on caspase activation
To investigate the contribution of protease and IAP-binding activity to Omi/HtrA2-induced caspase activation without any contribution of the mitochondrial proapoptotic factors, we made expression constructs with IAP-binding activity that were expressed extramitochondrially. We previously reported on an artificial construct containing an initiation codon (ATG) and the mature Omi/HtrA2 sequence (134–458). This construct was expressed extramitochondrially but showed little to no XIAP binding,13 presumably due to post-translational modification. Two experimental approaches have been devised to enable extramitochondrial expression of mature Smac/DIABLO with an intact IBM.29,30 Of these approaches, we chose the Ub fusion technique (Figure 4a).30,31 When expressed in HeLa cells, the Ub molecules of these fusion proteins were almost completely cleaved by endogenous Ub-specific proteases 24 h after transfection, generating ΔN forms with or without intact IBMs (Figure 4b). Binding between XIAP and each of the mature proteins produced was confirmed by a GST-pull down assay, the results of which indicate that XIAP binds only to proteins with intact IBMs (Figure 4b). Likewise, TNF markedly potentiated caspase activation in the presence of SmacΔN with an intact IBM, but not in the presence of SmacΔN with a disrupted IBM, compared with mock-transfected cells (Figure 4c). This indicates that Smac potentiates TNF-induced caspase activation by neutralizing IAPs using its IBM. On the other hand, catalytically inactive HtrA2ΔN (HtrA2ΔN S/A) did not significantly potentiate TNF-induced caspase activation, despite its ability to bind to IAPs, for at least 4 h after treatment (Figure 4c). This indicates that HtrA2ΔN has less potent IAP-inhibitory activity than SmacΔN. Surprisingly, exogenously expressed HtrA2ΔN activated caspase, regardless of whether or not it had IAP-binding activity (Figure 4c), suggesting that the protease activity of Omi/HtrA2 plays an important role in caspase activation.
Effect of extramitochondrially expressed HtrA2 or Smac mutants on caspase activation. (a) Schematic diagram of the Ub-fusion constructs of HtrA2 and Smac. Cleavage at the end of the Ub molecule generates proteins with intact (IBM, AVP) or mutated (IBM, MVP) IAP-binding motifs. The mutated IBMs lacked N-terminal mitochondria localization signals. S306 indicates the presence of a serine residue in the active site of HtrA2, which is mutated to alanine in the catalytically inactive mutant. (b) Binding between the indicated proteins and XIAP. Cell lysate samples prepared from HeLa cells transfected with the indicated expression constructs were pulled down with GST-tagged recombinant XIAP protein as described in the Experimental procedures. Whole-cell lysate (WCL) and XIAP-precipitated samples (PD) were subjected to immunoblot analysis using antibodies against Myc (IB: Myc) or GST (IB: GST). A and M indicate expression constructs with, or without, intact IBMs, respectively, as shown in (a). (c) HeLa cells transfected with the indicated expression constructs were treated with or without (–) 100 ng/ml TNF for 4 h. Cell lysate samples were prepared from adherent and floating cells, after which the caspase (DEVDase) activity of each lysate sample was measured as described in the Experimental Procedures
A feedback loop for amplification of mitochondrial Cyt c release contributes to Omi/HtrA2-induced caspase activation
We previously reported that the extramitochondrially expressed HtrA2 mutant induces little to no caspase activation in HEK293 cells. To elucidate differences between HeLa and HEK293 cells with respect to caspase activation, we investigated the effect of various proapoptotic molecules on caspase activation in these cell lines. The same HtrA2 expression constructs as described by our group in a previous report13 were used. When expressed extramitochondrially, HtrA2ΔN markedly activated caspase in HeLa, but not HEK293, cells (Figure 5a). The effect of HtrA2ΔN was observed to depend on protease activity, since the catalytically inactive mutant (HtrA2ΔN S/A) was not capable of caspase activation and HtrA2ΔN showed limited XIAP-binding (Figure 5a and data not shown). These results are consistent with those published in a previous report by our group13 and the data shown in Figure 4c. Although we tried to examine HtrA2ΔN-induced caspase activation in various cell lines, such as Neuro2A and SH-SY5Y cells, substantial activation of caspase was not observed in these cell lines (data not shown). However, induction of caspase by exogenously expressed HtrA2 has been observed in MCF7 cells.32 Both Bax and caspase-9 caused marked activation of caspase in HEK293 and HeLa cells (Figure 5a), indicating that HEK293 cells are capable of responding to caspase-activating molecules. However, tBid did not activate caspase in HEK293 cells, although it did in HeLa cells (Figure 5a). Thus, it appears that the response to tBid differs among these two cell lines. We further examined the response of isolated mitochondria from each of these cell lines to recombinant tBid and found that the mitochondria from HEK293 cells also did not respond to recombinant tBid (Figure 5b). These results suggest that tBid might be involved in HtrA2ΔN-induced caspase activation. However, recombinant HtrA2ΔN alone was not capable of cleaving either a recombinant precursor of Bid or endogenous Bid in a cytosolic S100 extract from HeLa cells (data not shown). In mammals, BH3-only proapoptotic Bcl-2 family proteins, including Bid, activate the multidomain proapoptotic members, Bax and Bak, to trigger mitochondrial release of Cyt c release.33 However, the relative insensitivity of HEK293 mitochondria to tBid cannot be attributed to an absence of Bax and/or Bak, since expression of these two proteins was detected in HEK293 cells (data not shown).
A feedback loop for amplification of mitochondrial Cyt c release contributes to HtrA2ΔN-induced caspase activation. (a) Extramitochondrially expressed HtrA2 mutant induces caspase activation in HeLa cells, but not HEK293, cells. HeLa cells and HEK293 cells were transfected with the indicated expression constructs. At 16–18 h after transfection, cell lysate samples were prepared, after which the caspase (DEVDase) activity of each lysate sample was measured as described in the Experimental procedures. HtrA2ΔN lacked the N-terminal mitochondria localization signal and was expressed outside of the mitochondria. (b) Recombinant tBid induces Cyt c release from mitochondria isolated from HeLa, but not HEK293, cells. The mitochondria were prepared from HeLa or HEK293 cells as described in the Experimental procedures. The isolated mitochondria were then incubated with 1 μM of recombinant tBid at 25°C for 30 min, after which the reaction mixtures were centrifuged. The supernatant (sup) and precipitated pellet (ppt) were subjected to immunoblot analysis with antibodies against c (Cyt c), or cytochrome oxidase subunit IV (COX). (c) HtrA2ΔN-induced caspase activation is blocked by Bcl-xL. HeLa cells were transfected with the indicated expression constructs. At 16–18 h after transfection, cell lysate samples were prepared, after which the caspase (DEVDase) activity of each lysate sample was measured as described in the Experimental procedures. The DEVDase activity of each sample was compared to the amount of DEVDase activity of the control (Ctrl: mock-transfected cells without Bcl-xL). (d) HtrA2ΔN-induced mitochondrial Cyt c release is blocked by Bcl-xL. HeLa cells were transfected in combination with HtrA2ΔN and/or Bcl-xL expression constructs as indicated. During transfection and following incubation, 50 μM of a broad-range caspase inhibitor (BAF) was added to one of the samples as indicated. At 12 h after transfection, cytosolic extracts were prepared, and then subjected to immunoblot analysis using antibodies against Cyt c or Actin as a control for cytosolic protein. (e) Recombinant HtrA2 did not directly induce Cyt c release from the isolated mitochondria. The mitochondria were prepared from HeLa cells as described in the Experimental procedures. The isolated mitochondria were incubated with 1 μM of the indicated recombinant proteins at 25°C for 30 min, after which the reaction mixtures were centrifuged. The supernatant (sup) and precipitated pellet (ppt) were subjected to immunoblot analysis with antibodies against Cyt c
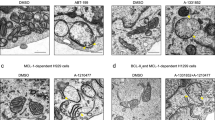
It has been reported that exogenously expressed HID, a Drosophila proapoptotic protein, induces apoptosis in HeLa, but not HEK293, cells, and that HID-induced apoptosis is inhibited by Bcl-xL.34 In light of the fact that Bcl-xL neutralizes some BH3-only proteins, including tBid, and since it inhibits Cyt c release from mitochondria and subsequent caspase activation,33 it is possible that Bcl-xL inhibits HtrA2ΔN-induced caspase activation. As expected, Bcl-xL suppressed HtrA2ΔN-, as well as tBid-, but not caspase-9- induced caspase activation (Figure 5c). Interestingly, Bcl-xL did not significantly suppress HtrA2ΔN-induced morphological changes, including cell rounding and shrinkage (data not shown), suggesting that these might result from induction of caspase-independent cell death by HtrA2 in HeLa cells. HtrA2ΔN also induced Cyt c release from the mitochondria, which was inhibited in the presence of Bcl-xL (Figure 5d). A broad-spectrum caspase inhibitor, Boc-Asp fluoromethyl ketone (BAF), could not suppress HtrA2ΔN-induced Cyt c release (Figure 5d), suggesting that caspases inhibitable by BAF might not be responsible for the release of Cyt c. We further examined whether HtrA2ΔN-induced Cyt c release resulted from direct cleavage of a mitochondrial protein (Figure 5e). When isolated HeLa mitochondria were incubated with recombinant tBid or caspase-3, Cyt c release was observed (Figure 5e). The observed release of Cyt c following exposure to recombinant caspase-3 was inconsistent with the results of a previous report,35 perhaps due to the large amount of caspase-3 added to the mitochondria, which might have induced a non-specific effect. However, despite using high concentrations, HtrA2, Smac, and caspase-8 did not induce Cyt c release (Figure 5e), indicating that Omi/HtrA2 does not directly induce Cyt c release in HeLa cells. Omi/HtrA2 might cleave one or more proteins from compartments other than the mitochondria, somehow resulting in mitochondrial Cyt c release.
The Bcl-2 family proteins play a pivotal role in induction of Cyt c release from mitochondria and they are regulated by transcription and post-translational modification, including phosphorylation and proteolytic processing. Bid is cleaved by caspase-3, as well as caspase-8, which mediates feedback amplification of mitochondrial Cyt c release.35 As described previously, HtrA2 was not observed to cleave Bid. Caspase-3 was observed to cleave Bcl-2 and Bcl-xL and convert them into Bax-like proapoptotic molecules.36,37,38 However, HtrA2 did not induce Cyt c release from isolated mitochondria (Figure 5e). Bax protein resides in the cytoplasm of living cells and translocates to the mitochondria in response to apoptotic stimuli,39 where it directly induces Cyt c release from mitochondria.40 HtrA2 may indirectly regulate the translocation of Bax by cleaving one or more Bax inhibitor proteins. Under normal conditions, the BH3-only protein, Bmf, is sequestered within the actin cytoskelton-based myosin V motor complex; however, Bmf may be released from the complex in response to stressful stimuli, such as detachment of adherent cells from their substratum (anoikis).41 HtrA2ΔN induced morphological changes resembling anoikis,13 which was not suppressed by cotransfection of Bcl-xL (described previously), implying involvement of Bmf in this process.
In this study, we provide evidence that the protease activity of Omi/HtrA2 enhances caspase activation and subsequent apoptosis by at least two different mechanisms, including direct cleavage and/or degradation of IAPs leading to inactivation of IAPs, as well as cleavage of an unidentified substrate(s) leading to permeabilization of the mitochondrial outer membrane and release of Cyt c (Figure 6). In light of the finding that Smac/DIABLO-deficient cells respond normally to various apoptotic stimuli, while Omi/HtrA2-knockdown cells are resistant to induction of apoptosis, the essential role of Omi/HtrA2 in apoptosis may dependent on its protease activity.
Materials and Methods
Plasmids, antibodies, and reagents
The plasmids encoding C-terminally Myc-tagged full-length human HtrA2 (pcDNA3-HtrA2-Myc), and the N-terminal 133 residue-deleted form of HtrA2 (pcDNA3-HtrA2ΔN-Myc), as well as C-terminally Myc-tagged full-length human Smac (pcDNA3-Smac-Myc), and N-terminally FLAG-tagged human IAPs (pcDNA3-FLAG-XIAP, -cIAP1, -cIAP2), pcDNA3-Bax, pGEX4T1-XIAP, -XIAPΔRING, pET28a-HtrA2ΔN133 (mature HtrA2 AVPS), along with -HtrA2ΔN134 (mature HtrA2 MVPS), have all been described previously.13,42 The cDNAs encoding human Bid and truncated Bid (tBid, residues 61–195) were generated from HeLa mRNA by reverse transcription-PCR (RT-PCR). The EcoRI–XhoI and NdeI–XhoI fragments of Bid and tBid were cloned in pcDNA3-HA-N2 (pcDNA3-HA-Bid and -tBid) and in pET28a (pET28-Bid and -tBid). pcDNA3-FLAG-Hop (Hsp70/Hsp90-organizing protein) was made by ligating the EcoRI–XhoI fragment of the RT-PCR product into pcDNA3-FLAG-N. pcDNA3-FLAG-DIAP1 was made by ligating the EcoRI–XhoI fragment of pUAS-DIAP1 (a generous gift from Dr. Masayuki Miura) into pcDNA3. The Ub-fusion constructs illustrated in Figure 4a were made by overlapping PCR, after which BamHI–XhoI fragments of Ub-Smac, and KpnI–XhoI fragments of Ub-HtrA2, were ligated into pcDNA3-Myc-C. pcDNA3-mouse Bcl-xL was a generous gift from Dr Yuzuru Imai. pcDNA3-Myc-caspase-9 was made by ligating the EcoRI–SalI fragment of the PCR product into the EcoRI–XhoI site of pcDNA3-Myc-N. pET15b-His6-caspase-8 (214–496) and pET23b-Caspase-3-His6 were generously provided by Dr Guy Salvesen.43 pET28a-SmacΔN55-His6 was made by ligating the NcoI-XhoI fragment of the PCR product into pET28a. The point mutants were generated using the QuikChange mutagenesis kit (Stratagene). Proper construction of all the plasmids was confirmed by DNA sequencing. The antisera against HtrA2 and XIAP have been described previously.13 The antibodies against Myc-tag (A-14), GST (B-14), and Hsc70 (K-19) were purchased from Santa Cruz Biotechnology. The antibodies against Cyt c (7H8.2C12 and 6H2.B4), Hsp60 (LK1), COX IV (20E8-C12), and Actin (C4) were purchased from BD PharMingen, SIGMA, Molecular Probes, and CHEMICON, respectively. TNF-alpha (TNF), CHX, Staurosporine, and BOC-Asp-FMK (BAF) were purchased from CHEMICON, Aldrich, Alomone labs, and Enzyme System Products, respectively.
Cell culture, transfection, binding assay, DEVDase activity, and immunoblot analysis
Human cervical carcinoma HeLa cells and human embryonic kidney (HEK) 293 cells were maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS). HEK293 and HeLa cells were transiently transfected using LipofectAMINE 2000 or LipofectAMINE Plus Reagents (Invitrogen), according to the manufacturer's instructions. The total amount of plasmid DNA used for transfection was kept constant by adding an appropriate amount of empty vector. In order to measure DEVDase activity and perform the apoptosis assay, 700 ng of plasmid DNA were transfected into each well (six- well plate). At 3–4 h after transfection, the medium containing the DNA/reagent mixture was replaced with fresh complete medium. At 16–18 h after transfection, the cells were treated with or without the indicated apoptosis-inducers. For UV treatment, the culture medium was replaced with 1 ml PBS and the cells were irradiated with a UV crosslinker (Stratalinker; Stratagene). The treated cells were then further cultured in fresh complete medium for several hours as indicated, after which cell lysate samples were prepared from adherent and floating cells. A GST-pull down assay was performed using GST-XIAPΔRING (20 pmol) with HeLa cell lysate (30 μg), as described previously.44 Assessment of DEVDase activity and the immunoblot analysis were performed as described previously.13,42
Recombinant proteins
GST-XIAP (1–497), GST-XIAPΔRING (1–437), HtrA2ΔN133-His6, HtrA2ΔN134-His6, and caspase-3-His6 have been described previously.13,42,44,45 The catalytically inactive HtrA2ΔN S306A mutant was made in the same way as previously described.13,45 HtrA2ΔN proteins were purified by nickel affinity chromatography and underwent HiTrap Desalting (Amersham Bioscience) in order to replace the buffer with Tris buffer (20 mM Tris-HCl (pH 8.0)) containing 50 mM NaCl. Then the eluate was loaded onto an HiTrap Q column (Amersham Bioscience) equilibrated with Tris buffer containing 50 mM NaCl. After washing the column with 10-column volumes of Tris buffer containing 50 mM NaCl, the bound protein was eluted with Tris buffer containing 150 mM NaCl. His6-caspase-8 (214–496), His6-Bid, and SmacΔN55-His6 were produced in the BL21 (DE3) pLysS Escherichia coli strain (Novagen) by incubating with 1 mM Isopropyl-1-thio-B-D-galactopyranoside (IPTG) for 3 h at 18°C, followed by purification with Ni-NTA agarose (Qiagen). His6-Bid was cleaved by recombinant caspase-8 to generate tBid, and the mixture of tBid/caspase-8 was treated with a broad-range caspase inhibitor (BAF), after which any free BAF was removed by ultrafiltration.
Immunoelectron microscopy
HeLa cells were washed and harvested with PBS. The cell pellets were suspended and fixed in a mixture of 4% paraformaldehyde and 0.1% glutaraldehyde in 0.1 M phosphate buffer (PB) for 4 h at 4°C. After washing in PB, the cell pellets were cut into ca. 0.5 mm3 blocks, and infiltrated with a mixture of 20% polyvinylpyrolidone and 1.8 M sucrose in PB overnight at 4°C. Ultrathin frozen sections were cut by Reichert Ultracut FCS cryoattachment at –110°C and collected on formvar film-coated EM grids with a drop of 2.3 M sucrose. For immunostaining, ultrathin sections were incubated with 5% normal goat serum in TBS (0.1 M Tris-buffered saline, pH 7.4, containing 1% bovine serum albumin and 0.1% gelatin) for 20 min, and then with primary antibody against HtrA2 (1 : 500) overnight at 4°C. The sections were incubated with 5 nm colloidal gold-conjugated secondary antibody for 2 h. The sections were then negatively stained with 2% polyvinylalcohol and 0.2% uranyl acetate and examined electron microscopically (LEO 912AB, LEO, Germany).
Immunostaining
For immunocytochemical analysis, HeLa cells were plated on eight-well chamber slides and cultivated for 18 h, followed by transfected with the indicated HtrA2 expression constructs. At 24 h after transfection, the cells were treated with or without UV irradiation as described above. At 2.5 h after treatment, the cells were washed with PBS, fixed with 4% paraformaldehyde, permeabilized with 0.2% Triton X-100, and then incubated with rabbit anti-Myc antibody (A-14, 1 : 200 dilution) and mouse anti-Cyt c antibody (6H2.B4, 1 : 500 dilution). The primary antibodies were detected by secondary antibodies conjugated to Alexa 488 or 546 (Molecular Probes). Stained cells were mounted in SlowFade (Molecular Probes), and then analyzed with a confocal laser scanning microscope system (TCS SP2, Leica).
Preparation of the S100 fraction and mitochondria
HeLa cells cultured in monolayer at 70% confluence were washed once with PBS and harvested by centrifugation at 800 g for 3 min at 4°C. The cell pellets were re-suspended in three volumes of buffer A (20 mM HEPES-KOH (pH 7.5), 10 mM KCl, 1.5 mM MgCl2, 1 mM EDTA, 1 mM EGTA, and 1 mM dithiotheitol (DTT)) containing 250 mM sucrose and 1 × Complete Protease inhibitor (Roche Diagnostics). After being kept on ice for 15 min, the cells were disrupted using a Teflon homogenizer on ice. The nuclei and mitochondria were pelleted at 8000 g for 10 min at 4°C. The supernatant was further centrifuged at 100 000 g for 10 min at 4°C. The resulting supernatant (S100 fraction, 6 mg protein/ml) was stored at –80°C and used for in vitro assay.
Mitochondria were isolated from HeLa or HEK293 cells as described previously46 with some modification. HeLa or HEK293 cells cultured in monolayer at 70% confluence were washed once with PBS and harvested by centrifugation at 800 g for 3 min at 4°C. The cell pellets were resuspended in 5 volumes of mitochondrial isolation buffer (MIB) (20 mM HEPES-KOH (pH 7.5), 10 mM KCl, 10 mM sodium succinate, 0.2 mM EGTA, 60 mM sucrose, 210 mM mannitol, 1 mM ADP, and 0.5 mM DTT) containing 1 × Complete Protease inhibitor. After being kept on ice for 10 min, the cells were disrupted by 100 strokes of a Teflon homogenizer. The unbroken cells and nuclei were pelleted at 600 g for 5 min, and the supernatant was centrifuged for 10 min at 5500 × g in order to pellet the mitochondria. The pellets were then resuspended in MIB containing 80 mM KCl and aliquots were used for the in vitro assays.
Abbreviations
- IAP:
-
inhibitor of apoptosis protein
- XIAP:
-
X chromosome-linked IAP
- c-IAP:
-
cellular IAP
- Smac:
-
second mitochondria-derived activator of caspase
- DIABLO:
-
direct IAP-binding protein with low pI
- HtrA2:
-
high temperature requirement A2
- IBM:
-
IAP-binding motif
- UV:
-
ultraviolet
- TNF:
-
tumor necrosis factor
- Cyt c:
-
cytochrome c
- DEVD-MCA:
-
acetyl-Asp-Glu-Val-Asp-4-methyl-coumaryl-7-amide
- AMC:
-
7-amino-4-methyl coumarin
- BAF:
-
Boc-Asp fluoromethyl ketone
- GST:
-
glutathione S-transferase
- DTT:
-
dithiothreitol
- PAGE:
-
polyacrylamide gel electrophoresis
References
Jacobson MD, Weil M and Raff MC (1997) Programmed cell death in animal development. Cell 88: 347–354
Thompson CB (1995) Apoptosis in the pathogenesis and treatment of disease. Science 267: 1456–1462
Thornberry NA and Lazebnik Y (1998) Caspases: enemies within. Science 281: 1312–1316
Miller LK (1999) An exegesis of IAPs: salvation and surprises from BIR motifs. Trends Cell Biol. 9: 323–328
Deveraux QL and Reed JC (1999) IAP family proteins – suppressors of apoptosis. Genes Dev. 13: 239–252
Deveraux QL, Takahashi R, Salvesen GS and Reed JC (1997) X-linked IAP is a direct inhibitor of cell-death proteases. Nature 388: 300–304
Deveraux QL, Roy N, Stennicke HR, Van Arsdale T, Zhou Q, Srinivasula SM, Alnemri ES, Salvesen GS and Reed JC (1998) IAPs block apoptotic events induced by caspase-8 and cytochrome c by direct inhibition of distinct caspases. EMBO J. 17: 2215–2223
Roy N, Deveraux QL, Takahashi R, Salvesen GS and Reed JC (1997) The c-IAP-1 and c-IAP-2 proteins are direct inhibitors of specific caspases. EMBO J. 16: 6914–6925
Takahashi R, Deveraux Q, Tamm I, Welsh K, Assa-Munt N, Salvesen GS and Reed JC (1998) A single BIR domain of XIAP sufficient for inhibiting caspases. J. Biol. Chem. 273: 7787–7790
Salvesen GS and Duckett CS (2002) IAP proteins: blocking the road to death's door. Nat. Rev. Mol. Cell Biol. 3: 401–410
Du C, Fang M, Li Y, Li L and Wang X (2000) Smac, a mitochondrial protein that promotes cytochrome c-dependent caspase activation by eliminating IAP inhibition. Cell 102: 33–42
Verhagen AM, Ekert PG, Pakusch M, Silke J, Connolly LM, Reid GE, Moritz RL, Simpson RJ and Vaux DL (2000) Identification of DIABLO, a mammalian protein that promotes apoptosis by binding to and antagonizing IAP proteins. Cell 102: 43–53
Suzuki Y, Imai Y, Nakayama H, Takahashi K, Takio K and Takahashi R (2001) A serine protease, HtrA2, is released from the mitochondria and interacts with XIAP, inducing cell death. Mol. Cell 8: 613–621
Hegde R, Srinivasula SM, Zhang Z, Wassell R, Mukattash R, Cilenti L, DuBois G, Lazebnik Y, Zervos AS, Fernandes-Alnemri T and Alnemri ES (2002) Identification of Omi/HtrA2 as a mitochondrial apoptotic serine protease that disrupts inhibitor of apoptosis protein–caspase interaction. J. Biol. Chem. 277: 432–438
Verhagen AM, Silke J, Ekert PG, Pakusch M, Kaufmann H, Connolly LM, Day CL, Tikoo A, Burke R, Wrobel C, Moritz RL, Simpson RJ and Vaux DL (2002) HtrA2 promotes cell death through its serine protease activity and its ability to antagonize inhibitor of apoptosis proteins. J. Biol. Chem. 277: 445–454
Martins LM, Iaccarino I, Tenev T, Gschmeissner S, Totty NF, Lemoine NR, Savopoulos J, Gray CW, Creasy CL, Dingwall C and Downward J (2002) The serine protease Omi/HtrA2 regulates apoptosis by binding XIAP through a reaper-like motif. J. Biol. Chem. 277: 439–444
van Loo G, van Gurp M, Depuydt B, Srinivasula SM, Rodriguez I, Alnemri ES, Gevaert K, Vandekerckhove J, Declercq W and Vandenabeele P (2002) The serine protease Omi/HtrA2 is released from mitochondria during apoptosis. Omi interacts with caspase-inhibitor XIAP and induces enhanced caspase activity. Cell Death Differ. 9: 20–26
Wu G, Chai J, Suber TL, Wu JW, Du C, Wang X and Shi Y (2000) Structural basis of IAP recognition by Smac/DIABLO. Nature 408: 1008–1012
Okada H, Suh WK, Jin J, Woo M, Du C, Elia A, Duncan GS, Wakeham A, Itie A, Lowe SW, Wang X and Mak TW (2002) Generation and characterization of Smac/DIABLO-deficient mice. Mol. Cell. Biol. 22: 3509–3517
Deveraux QL, Leo E, Stennicke HR, Welsh K, Salvesen GS and Reed JC (1999) Cleavage of human inhibitor of apoptosis protein XIAP results in fragments with distinct specificities for caspases. EMBO J. 18: 5242–5251
Yoo SJ, Huh JR, Muro I, Yu H, Wang L, Wang SL, Feldman RM, Clem RJ, Muller HA and Hay BA (2002) Hid, Rpr and Grim negatively regulate DIAP1 levels through distinct mechanisms. Nat. Cell Biol. 4: 416–424
Ryoo HD, Bergmann A, Gonen H, Ciechanover A and Steller H (2002) Regulation of Drosophila IAP1 degradation and apoptosis by reaper and ubcD1. Nat. Cell Biol. 4: 432–438
Holley CL, Olson MR, Colon-Ramos DA and Kornbluth S (2002) Reaper eliminates IAP proteins through stimulated IAP degradation and generalized translational inhibition. Nat. Cell Biol. 4: 439–444
Yang Y, Fang S, Jensen JP, Weissman AM and Ashwell JD (2000) Ubiquitin protein ligase activity of IAPs and their degradation in proteasomes in response to apoptotic stimuli. Science 288: 874–877
Kobayashi S, Yamashita K, Takeoka T, Ohtsuki T, Suzuki Y, Takahashi R, Yamamoto K, Kaufmann SH, Uchiyama T, Sasada M and Takahashi A (2002) Calpain-mediated X-linked inhibitor of apoptosis degradation in neutrophil apoptosis and its impairment in chronic neutrophilic leukemia. J. Biol. Chem. 277: 33968–33977
Jin S, Kalkum M, Overholtzer M, Stoffel A, Chait BT and Levine AJ (2003) CIAP1 and the serine protease HTRA2 are involved in a novel p53-dependent apoptosis pathway in mammals. Genes Dev. 17: 359–367
Yang QH, Church-Hajduk R, Ren J, Newton ML and Du C (2003) Omi/HtrA2 catalytic cleavage of inhibitor of apoptosis (IAP) irreversibly inactivates IAPs and facilitates caspase activity in apoptosis. Genes Dev. 17: 1487–1496
Srinivasula SM, Gupta S, Datta P, Zhang Z, Hegde R, Cheong N, Fernandes-Alnemri T and Alnemri ES (2003) Inhibitor of apoptosis proteins are substrates for the mitochondrial serine protease Omi/HtrA2. J. Biol. Chem. 278: 31469–31472
Srinivasula SM, Datta P, Fan XJ, Fernandes-Alnemri T, Huang Z and Alnemri ES (2000) Molecular determinants of the caspase-promoting activity of Smac/DIABLO and its role in the death receptor pathway. J. Biol. Chem. 275: 36152–36157
Hunter AM, Kottachchi D, Lewis J, Duckett CS, Korneluk RG and Liston P (2003) A novel ubiquitin fusion system bypasses the mitochondria and generates biologically active Smac/DIABLO. J. Biol. Chem. 278: 7494–7499
Varshavsky A (2000) Ubiquitin fusion technique and its descendants. Methods Enzymol. 327: 578–593
Li W, Srinivasula SM, Chai J, Li P, Wu JW, Zhang Z, Alnemri ES and Shi Y (2002) Structural insights into the pro-apoptotic function of mitochondrial serine protease HtrA2/Omi. Nat. Struct. Biol. 9: 436–441
Cheng EH, Wei MC, Weiler S, Flavell RA, Mak TW, Lindsten T and Korsmeyer SJ (2001) BCL-2, BCL-X(L) sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol. Cell 8: 705–711
Haining WN, Carboy-Newcomb C, Wei CL and Steller H (1999) The proapoptotic function of Drosophila Hid is conserved in mammalian cells. Proc. Natl. Acad. Sci. USA 96: 4936–4941
Bossy-Wetzel E and Green DR (1999) Caspases induce cytochrome c release from mitochondria by activating cytosolic factors. J. Biol. Chem. 274: 17484–17490
Cheng EH, Kirsch DG, Clem RJ, Ravi R, Kastan MB, Bedi A, Ueno K and Hardwick JM (1997) Conversion of Bcl-2 to a Bax-like death effector by caspases. Science 278: 1966–1968
Clem RJ, Cheng EH, Karp CL, Kirsch DG, Ueno K, Takahashi A, Kastan MB, Griffin DE, Earnshaw WC, Veliuona MA and Hardwick JM (1998) Modulation of cell death by Bcl-XL through caspase interaction. Proc. Natl. Acad. Sci. USA 95: 554–559
Kirsch DG, Doseff A, Chau BN, Lim DS, de Souza-Pinto NC, Hansford R, Kastan MB, Lazebnik YA and Hardwick JM (1999) Caspase-3-dependent cleavage of Bcl-2 promotes release of cytochrome c. J. Biol. Chem. 274: 21155–21161
Wolter KG, Hsu YT, Smith CL, Nechushtan A, Xi XG and Youle RJ (1997) Movement of Bax from the cytosol to mitochondria during apoptosis. J. Cell Biol. 139: 1281–1292
Jurgensmeier JM, Xie Z, Deveraux Q, Ellerby L, Bredesen D and Reed JC (1998) Bax directly induces release of cytochrome c from isolated mitochondria. Proc. Natl. Acad. Sci. USA 95: 4997–5002
Puthalakath H, Villunger A, O'Reilly LA, Beaumont JG, Coultas L, Cheney RE, Huang DC and Strasser A (2001) Bmf: a proapoptotic BH3-only protein regulated by interaction with the myosin V actin motor complex, activated by anoikis. Science 293: 1829–1832
Suzuki Y, Nakabayashi Y and Takahashi R (2001) Ubiquitin–protein ligase activity of X-linked inhibitor of apoptosis protein promotes proteasomal degradation of caspase-3 and enhances its anti-apoptotic effect in Fas-induced cell death. Proc. Natl. Acad. Sci. USA 98: 8662–8667
Zhou Q, Snipas S, Orth K, Muzio M, Dixit VM and Salvesen GS (1997) Target protease specificity of the viral serpin CrmA. Analysis of five caspases. J. Biol. Chem. 272: 7797–7800
Suzuki Y, Nakabayashi Y, Nakata K, Reed JC and Takahashi R (2001) X-linked inhibitor of apoptosis protein (XIAP) inhibits caspase-3 and -7 in distinct modes. J. Biol. Chem. 276: 27058–27063
Savopoulos JW, Carter PS, Turconi S, Pettman GR, Karran EH, Gray CW, Ward RV, Jenkins O and Creasy CL (2000) Expression, purification, and functional analysis of the human serine protease HtrA2. Protein Expr. Purif. 19: 227–234
von Ahsen O, Renken C, Perkins G, Kluck RM, Bossy-Wetzel E and Newmeyer DD (2000) Preservation of mitochondrial structure and function after Bid- or Bax-mediated cytochrome c release. J. Cell Biol. 150: 1027–1036
Acknowledgements
We thank Dr. Guy Salvesen for the caspase plasmids, Dr. Masayuki Miura for the DIAP1 cDNA, and Dr. Imai Yuzuru for the Bcl-xL plasmid. We also thank all of our laboratory members for their helpful discussion. This work was supported in part by a research grant from RIKEN BSI to RT, President's Special Research Fund grant from RIKEN to RT, a Grant-in-Aid for Scientific Research from the Japanese ministry of Education, Culture, Sports, Science, and Technology to RT and YS, and a grant from the Special Postdoctoral Researchers Program of RIKEN to YS
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by H Ichijo
Rights and permissions
About this article
Cite this article
Suzuki, Y., Takahashi-Niki, K., Akagi, T. et al. Mitochondrial protease Omi/HtrA2 enhances caspase activation through multiple pathways. Cell Death Differ 11, 208–216 (2004). https://doi.org/10.1038/sj.cdd.4401343
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cdd.4401343
Keywords
This article is cited by
-
A literature review of microRNA and gene signaling pathways involved in the apoptosis pathway of lung cancer
Respiratory Research (2023)
-
The role of Ubiquitination in Apoptosis and Necroptosis
Cell Death & Differentiation (2022)
-
Prevention of acquired sensorineural hearing loss in mice by in vivo Htra2 gene editing
Genome Biology (2021)
-
An updated review of mechanistic potentials of melatonin against cancer: pivotal roles in angiogenesis, apoptosis, autophagy, endoplasmic reticulum stress and oxidative stress
Cancer Cell International (2021)
-
Engorgement of Rhipicephalus haemaphysaloides ticks blocked by silencing a protein inhibitor of apoptosis
Experimental and Applied Acarology (2021)