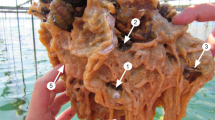
An uncanny similarity in the karyotype of these malignant tumours means that they could be infective.
Abstract
The Tasmanian devil, a large carnivorous Australian marsupial, is under threat from a widespread fatal disease in which a malignant oral–facial tumour obstructs the animal's ability to feed1. Here we show that the chromosomes in these tumours have undergone a complex rearrangement that is identical for every animal studied. In light of this remarkable finding and of the known fighting behaviour of the devils2, we propose that the disease is transmitted by allograft, whereby an infectious cell line is passed directly between the animals through bites they inflict on one another.
Similar content being viewed by others
Main
The cancer, known as devil facial-tumour disease, now affects devils (Sarcophilus harrisii) in more than half of Tasmania1. The growth of the tumours, which ulcerate and become friable, eventually causes the devils to starve. As the tumour cells are easily dislodged and because almost all bites from the devils' frequent fighting occur around the mouth2, we investigated whether the disease might be transmitted by allograft between animals. We studied tumours that included early neoplasms, huge primary cancers and secondary cancers. The cancers were sampled from animals throughout eastern Tasmania, Australia, over a 12-month period (for methods, see supplementary information).
The normal number of chromosomes in the devil is 14, including the XX or XY sex chromosomes (Fig. 1a). We found that the facial tumours contained only 13 chromosomes and that these were grossly abnormal (Fig. 1b). The number and appearance of the chromosomes (the karyotype) indicated that both sex chromosomes, both chromosomes 2 and one chromosome 6 were absent. There was also a deletion of the long arm of one chromosome 1, and four unidentified marker chromosomes were present. Most important, these anomalies were the same in the facial tumours from every animal (n = 11).
a, Normal karyotype for a male Tasmanian devil (14 chromosomes, including XY). b, Karyotype of cancer cells found in each of the facial tumours of all 11 animals studied (13 chromosomes, with no sex chromosomes, no chromosome-2 pair and only one chromosome 6; the long arm of one chromosome 1 was deleted; four additional marker chromosomes were present (M1–M4).
These rearrangements are complex, but no intermediate stages were found between normal and tumour chromosomes, even in small primary cancers. In human cancers, there is generally a common breakpoint (first event)3, irrespective of whether the neoplasm is caused by viral insertion (as in Burkitt's lymphoma4) or arises spontaneously (as in Ewing's sarcoma3); complex rearrangements occur in solid tumours as a result of further clonal evolution5. However, the identical chromosomal rearrangements that we found in the facial tumours of each devil are too complex for a common breakpoint to have occurred3. Indeed, the rearrangements do not conform to any human model, particularly given the loss of sex chromosomes in all the tumours of devils of both sexes.
Further support for the allograft theory of disease transmission derives from the serendipitous observation of a pericentric inversion of chromosome 5 in the constitutional karyotype of one animal. This constitutional anomaly was found in all cultures of that devil's normal tissues, but was not present in either of the chromosomes 5 in his facial-tumour cells, where it would have been found had the neoplasm arisen from his own tissue.
Cases of transmissible venereal sarcoma in dogs6 also show similar chromosomal defects among tumours, leading to the proposition that this sarcoma may develop from “a clone capable of a parasitic existence”7 — a description that also fits the features of the devil's facial tumour. We suggest that the devils' cancer (like the dogs') is infective and that the infective agent is a rogue cell line that initially evolved in a tumour of unknown origin.
Humans, too, can accidentally infect each other with cancer, through cell implantation in patients that have received organ transplants8; such cancers then develop according to their usual course9. Organ transplants are less likely to be rejected if the donor is a close relative who has a matching tissue type; by analogy, the low genetic diversity and high degree of kinship among devils10 might help to reduce their immune response to cancer cells implanted during biting. Although the devil's immune system is poorly understood, preliminary investigations indicate that there is little immune reaction between lymphocytes taken from devils from within and outside local populations (G. Woods, personal communication).
To obtain further insight into the transmission of the devil's facial-tumour disease, it will be necessary to DNA-fingerprint tumours and clarify their derivation by using whole-chromosome painting probes, as well as searching for oncogenes. This should reveal the disease's toxicology, progression and epidemiology.
References
Hawkins, C. E. et al. Biol. Cons. (submitted).
Hamede, R. Thesis, Univ. Tasmania, (Hobart, 2004).
Mitelman, F. Mutat. Res. 462, 247–253 (2000).
Zech, L., Haglund, U., Nilsson, K. & Klein, G. Int. J. Cancer 17, 47–56 (1976).
Mitelman, F., Johansson, B., Mandahl, N. & Mertens, F. Cancer Genet. Cytogenet. 95, 1–8 (1997).
Das, U. & Das, A. K. Vet. Res. Commun. 24, 545–556 (2000).
Murray, M., James, Z. H. & Martin, W. B. Res. Vet. Sci. 10, 565–568 (1969).
Kauffman, H. M. & McBride, M. A. Transplantation 74, 358–362 (2002).
Forbes, G. B. et al. J. Clin. Pathol. 34, 109–115 (1981).
Jones, M. E., Paetkau, D., Geffen, E. & Moritz, C. Mol. Ecol. 13, 2197–2209 (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
(DOC 25 kb)
Rights and permissions
About this article
Cite this article
Pearse, AM., Swift, K. Transmission of devil facial-tumour disease. Nature 439, 549 (2006). https://doi.org/10.1038/439549a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/439549a
This article is cited by
-
Living in human-modified landscapes narrows the dietary niche of a specialised mammalian scavenger
Scientific Reports (2023)
-
Centuries of genome instability and evolution in soft-shell clam, Mya arenaria, bivalve transmissible neoplasia
Nature Cancer (2023)
-
Game-theoretical description of the go-or-grow dichotomy in tumor development for various settings and parameter constellations
Scientific Reports (2023)
-
Somatic evolution of marine transmissible leukemias in the common cockle, Cerastoderma edule
Nature Cancer (2023)
-
Population genetics of clonally transmissible cancers
Nature Ecology & Evolution (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.