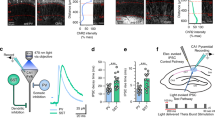
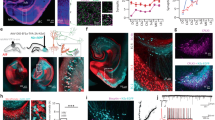
Abstract
The hippocampus is necessary for the acquisition and retrieval of declarative memories1,2. The best-characterized sensory input to the hippocampus is the perforant path projection from layer II of entorhinal cortex (EC) to the dentate gyrus3,4. Signals are then processed sequentially in the hippocampal CA fields before returning to the cortex via CA1 pyramidal neuron spikes. There is another EC input—the temporoammonic (TA) pathway—consisting of axons from layer III EC neurons that make synaptic contacts on the distal dendrites of CA1 neurons3,5,6. Here we show that this pathway modulates both the plasticity and the output of the rat hippocampal formation. Bursts of TA activity can, depending on their timing, either increase or decrease the probability of Schaffer-collateral (SC)-evoked CA1 spikes. TA bursts can also significantly reduce the magnitude of synaptic potentiation at SC–CA1 synapses. The TA–CA1 synapse itself exhibits both long-term depression (LTD) and long-term potentiation (LTP). This capacity for bi-directional plasticity can, in turn, regulate the TA modulation of CA1 activity: LTP or LTD of the TA pathway either enhances or diminishes the gating of CA1 spikes and plasticity inhibition, respectively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Morris, R. G. M., Garrud, P., Rawlins, J. N. P. & O'Keefe, J. Place navigation impaired in rats with hippocampal lesions. Nature 297, 681–683 (1982).
Eichenbaum, H. A cortical–hippocampal system for declarative memory. Nature Rev. Neurosci. 1, 41–50 (2001).
Witter, M. P., Groenewegen, H. J., Silva, F. H. L. D. & Lohman, A. H. Functional organization of the extrinsic and intrinsic circuitry of the parahippocampal region. Prog. Neurobiol. 33, 161–253 (1989).
Amaral, D. G. & Witter, M. P. The three-dimensional organisation of the hippocampus: a review of the anatomical data. Neuroscience 31, 571–591 (1989).
Cajal, S. R. The Structure of Ammon's Horn (ed. Thomas, C. C.) (Thomas, Springfield, Illinois, 1968).
Steward, O. & Scoville, S. A. Cells of origin of entorhinal cortical affferents to the hippocampus and fascia dentate of the rat. J. Comp. Neurol. 169, 347–370 (1976).
Dvorak-Carbone, H. & Schuman, E. M. Long-term depression of temporoammonic-CA1 hippocampal synaptic transmission. J. Neurophysiol. 81, 1036–1044 (1999).
Dvorak-Carbone, H. & Schuman, E. M. Patterned activity in stratum lacunosum moleculare inhibits CA1 pyramidal neuron firing. J. Neurophysiol. 82, 3213–3222 (1999).
Empson, R. M. & Heineman, U. The perforant path projection to hippocampal area CA1 in the rat hippocampal–entorhinal cortex combined slice. J. Physiol. (Lond.) 484, 707–720 (1995).
Levy, W. B., Desmond, N. L. & Zhang, D. X. Perforant path activation modulates the induction of long-term potentiation of the Schaffer collateral–hippocampal CA1 response: theoretical and experimental analyses. Learn. Mem. 4, 510–518 (1998).
Chrobak, J. J. & Buzsaki, G. Gamma oscillations in the entorhinal cortex of the freely behaving rat. J. Neurosci. 18, 388–398 (1998).
Chrobak, J. J., Lorincz, A. & Buzsaki, G. Physiological patterns in the hippocampo-entorhinal cortex system. Hippocampus 10, 457–465 (2000).
Colbert, C. M. & Levy, W. B. Electrophysiological and pharmacological characterization of perforant path synapses in CA1: mediation by glutamate receptors. J. Neurophysiol. 68, 1–8 (1992).
Maccaferri, G. & McBain, C. J. Passive propagation of LTD to stratum oriens-alveus inhibitory neurons modulates the temporoammonic input to the hippocampal CA1 region. Neuron 15, 137–145 (1995).
Jarrard, L. E., Okaichi, H., Steward, O. & Goldschmidt, R. B. On the role of hippocampal connections in the performance of place and cue tasks: comparisons with damage to hippocampus. Behav. Neurosci. 98, 946–954 (1984).
Jarrard, L. E. What does the hippocampus really do? Behav. Brain. Res. 71, 1–10 (1995).
Muller, R. A quarter of a century of place cells. Neuron 17, 813–822 (1996).
McNaughton, B. L., Barnes, C. A., Meltzer, J. & Sutherland, R. J. Hippocampal granule cells are necessary for normal spatial learning but not for spatially-selective pyramidal cell discharge. Exp. Brain. Res. 76, 485–496 (1989).
Brun, V. H., Otnæss, M. K., Witter, M. P., Moser, M. B. & Moser, E. I. Place representation in hippocampal area CA1 in the absence of input from area CA3. Soc. Neurosci. Abstr. 31 (2001).
Acknowledgements
We thank G. Laurent for critial reading of the manuscript. This work was supported by Fundacao para a Ciencia e Tecnologia (FCT)—Portugal and the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests
Supplementary information
Rights and permissions
About this article
Cite this article
Remondes, M., Schuman, E. Direct cortical input modulates plasticity and spiking in CA1 pyramidal neurons. Nature 416, 736–740 (2002). https://doi.org/10.1038/416736a
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/416736a
This article is cited by
-
Heterosynaptic plasticity-induced modulation of synapses
The Journal of Physiological Sciences (2023)
-
Long-term potentiation of the nucleus reuniens and entorhinal cortex to CA1 distal dendritic synapses in mice
Brain Structure and Function (2020)
-
Leptin Regulation of Synaptic Function at Hippocampal TA-CA1 and SC-CA1 Synapses: Implications for Health and Disease
Neurochemical Research (2019)
-
HCN1 channels reduce the rate of exocytosis from a subset of cortical synaptic terminals
Scientific Reports (2017)
-
Interaction of nucleus reuniens and entorhinal cortex projections in hippocampal field CA1 of the rat
Brain Structure and Function (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.