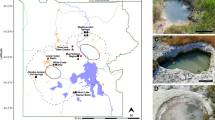
Abstract
The search for extraterrestrial life may be facilitated if ecosystems can be found on Earth that exist under conditions analogous to those present on other planets or moons. It has been proposed, on the basis of geochemical and thermodynamic considerations, that geologically derived hydrogen might support subsurface microbial communities on Mars and Europa in which methanogens form the base of the ecosystem1,2,3,4,5. Here we describe a unique subsurface microbial community in which hydrogen-consuming, methane-producing Archaea far outnumber the Bacteria. More than 90% of the 16S ribosomal DNA sequences recovered from hydrothermal waters circulating through deeply buried igneous rocks in Idaho are related to hydrogen-using methanogenic microorganisms. Geochemical characterization indicates that geothermal hydrogen, not organic carbon, is the primary energy source for this methanogen-dominated microbial community. These results demonstrate that hydrogen-based methanogenic communities do occur in Earth's subsurface, providing an analogue for possible subsurface microbial ecosystems on other planets.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Boston, P. M., Ivanov, M. V. & McKay, C. P. On the possibility of chemosynthetic ecosystems in subsurface habitats on Mars. Icarus 95, 300–308 (1992).
McCollom, T. M. Methanogenesis as a potential source of chemical energy for primary biomass production by autotrophic organisms in hydrothermal systems on Europa. J. Geophys. Res. 104 (E12), 30729–30742 (1999).
Carr, M. H. in Evolution of Hydrothermal Ecosystems on Earth (and Mars?) (eds Bock, G. R. & Goode, J. A.) 249–265 (Wiley, Chichester, 1996).
McKay, C. P. in Subsurface Microbiology and Biogeochemistry (eds Fredrickson, J. K. & Fletcher, M.) 315–327 (Wiley, New York, 2001).
Fisk, M. R. & Giovannoni, S. J. Sources of nutrients and energy for a deep biosphere on Mars. J. Geophys. Res. 104 (E12), 11805–11815 (1999).
Shock, E. L. in Evolution of Hydrothermal Ecosystems on Earth (and Mars?) (eds Bock, G. R. & Goode, J. A.) 40–60 (Wiley, Chichester, 1996).
Farmer, J. D. Hydrothermal systems: Doorways to early biosphere evolution. GSA Today 10 (7), 1–9 (2000).
Embree, G. F., McBroome, L. A. & Soherty, D. J. Preliminary stratigraphic framework of the Pliocene and Miocene rhyolite, Eastern Snake River Plain. Idaho Bur. Mines Geol. Bull. 26, 333–346 (1982).
Sugisaki, R. & Sugiura, T. Gas anomalies at three mineral springs and a fumarole before an inland earthquake, Central Japan. J. Geophys. Res. 91, 12296–12304 (1986).
Lilley, M. D. & Olson, E. J. in Abstr. 11th Annu. V. M. Goldschmidt Conf. Abstract no. 3682, LPI contribution no. 1088 (Lunar and Planetary Institute, Houston, 2001) (CD-ROM).
Wakita, H. et al. Hydrogen release: New indicator of fault activity. Science 210, 188–190 (1980).
Pierce, K. L. & Morgan, L. A. The track of the Yellowstone hot spot: Volcanism, faulting, and uplift. Geol. Soc. Am. Mem. no. 179 (1992).
Knobel, L. L. et al. Chemical constituents in ground water from 39 selected sites with an evaluation of quality assurance data, Idaho National Engineering and Environmental Laboratory and vicinity, Idaho. US Geol. Surv. Op. File Rep. no. 99-246 (1999).
Lovley, D. R. & Goodwin, S. Hydrogen concentrations as an indicator of the predominant terminal electron accepting reactions in aquatic sediments. Geochim. Cosmochim. Acta 52, 2993–3003 (1988).
Orphan, V. J., Taylor, L. T., Hafenbrandl, D. & Delong, E. F. Culture-dependent and culture-independent characterization of microbial assemblages associated with high-temperature petroleum reservoirs. Appl. Environ. Microbiol. 66, 700–711 (2000).
Picard, C. et al. Detection and enumeration of bacteria in soil by direct DNA extraction and polymerase chain reaction. Appl. Environ. Microbiol. 58, 2717–2722 (1992).
Klappenbach, J. A. et al. rrndb: the ribosomal RNA operon copy number database. Nucleic Acids Res. 29, 181–184 (2001).
Amann, R. I., Ludwid, W. & Schleifer, K. H. Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol. Rev. 59, 143–169 (1995).
Massana, R., Murray, A. E., Preston, C. M. & Delong, E. F. Vertical distribution and phylogenetic characterization of marine planktonic Archaea in the Santa Barbara channel. Appl. Environ. Microbiol. 63, 50–56 (1997).
Moyer, C. L., Dobbs, F. C. & Karl, D. M. Estimation of diversity and community structure through restriction fragment length polymorphism distribution analysis of bacterial 16S rRNA genes from a microbial mat at an active hydrothermal vent system, Loihi Seamount, Hawaii. Appl. Environ. Microbiol. 60, 871–879 (1994).
Barns, S. M. et al. Remarkable archaeal diversity detected in a Yellowstone National Park hot spring environment. Proc. Natl Acad. Sci. USA 91, 1609–1613 (1994).
Stevens, T. O. & McKinley, J. P. Lithoautotrophic microbial ecosystems in deep basalt aquifers. Science 270, 450–454 (1995).
Fry, N. K. et al. Population structure of microbial communities associated with two deep, anaerobic, alkaline aquifers. Appl. Environ. Microbiol. 63, 1498–1504 (1997).
Anderson, R. T., Chapelle, F. H. & Lovley, D. R. Evidence against hydrogen-based microbial ecosystems in basalt aquifers. Science 281, 976–977 (1998).
Chapelle, F. H., Vroblesky, D. A., Woodward, J. C. & Lovley, D. R. Practical considerations for measuring hydrogen concentrations in groundwater. Environ. Sci. Tech. 31, 2873–2877 (1997).
Hobbie, J. E., Daley, R. J. & Jasper, S. Use of Nuclepore filters for counting bacteria by fluorescence microscopy. Appl. Environ. Microbiol. 33, 1225–1228 (1977).
Acknowledgements
This research was supported by the US Geological Survey and a grant from the LexEn programme of the National Science Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Chapelle, F., O'Neill, K., Bradley, P. et al. A hydrogen-based subsurface microbial community dominated by methanogens. Nature 415, 312–315 (2002). https://doi.org/10.1038/415312a
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/415312a
This article is cited by
-
The biomass and biodiversity of the continental subsurface
Nature Geoscience (2018)
-
Microbial Composition and Diversity Patterns in Deep Hyperthermal Aquifers from the Western Plain of Romania
Microbial Ecology (2018)
-
Factors influencing hydrogen yield in water radiolysis and implications for hydrocarbon generation: a review
Arabian Journal of Geosciences (2018)
-
Reconstructing a hydrogen-driven microbial metabolic network in Opalinus Clay rock
Nature Communications (2016)
-
Arsenic release metabolically limited to permanently water-saturated soil in Mekong Delta
Nature Geoscience (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.