Abstract
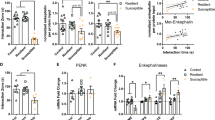
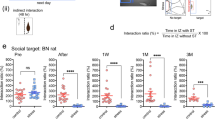
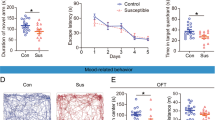
Cholecystokinin (CCK) involvement in depression-like disorders is poorly documented. Here, we investigated whether CCKergic neurotransmission is relevant to depressive-like symptoms and antidepressant therapy using a novel preclinical model based on repeated social defeat over 4 weeks in rats. Repeated social defeat triggers changes that could be considered as behavioral and biological correlates of depressive symptoms in humans, such as a hyperactivity of hypothalamic–pituitary–adrenal (HPA) axis (increase of serum corticosterone levels and of adrenal gland weight), increased immobility time in the forced swimming test (FST), decrease of body weight and of sweet water consumption and reduction of hippocampal volume associated with a decreased cell proliferation in the dentate gyrus. In addition, in vivo microdialysis showed that cortical CCK release was tonically increased in defeated rats. Chronic imipramine treatment (16 mg kg−1 per day for 25 days) prevented both the repeated social defeat-induced alterations of biological and behavioral parameters and the associated increase of cortical CCK release. Chronic blockade of CCK2 receptors by the specific antagonist CI-988 (1 mg kg−1 per day for 25 days) also normalized immobility time in the FST and prevented HPA axis hyperactivity, reduction of hippocampal volume and cell proliferation and decreased sweet water intake normally evoked by repeated social defeat. These data showed that the repeated social-defeat paradigm can be considered as a suitable model of ‘depression’ in rats. The causal link between social defeat-evoked (1) increase in cortical CCKergic neurotransmission and (2) depression-like symptoms that we highlighted here strongly suggests that CCKergic systems may be a relevant target for novel antidepressant therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Willner P, Muscat R, Papp M . Chronic mild stress-induced anhedonia: a realistic animal model of depression. Neurosci Biobehav Rev 1992; 16: 525–534.
Cryan JF, Slattery DA . Animal models of mood disorders: recent developments. Curr Opin Psychiatry 2007; 20: 1–7.
Koolhaas JM, Hermann PM, Kemperman C, Bohus B, van den Hoofdakker RH, Beersma DGM . Single social defeat in male rats induces a gradual but long lasting behavioural change: a model of depression? Neurosci Res Commun 1990; 7: 35–41.
Koolhaas JM, Meerlo P, De Boer SF, Strubbe JH, Bohus B . Social stress in rats: an animal model of depression? Acta Neuropsychiatrica 1995; 7: 27–29.
Tornatzky W, Miczek KA . Long-term impairment of autonomic circadian rhythms after brief intermittent social stress. Physiol Behav 1993; 53: 983–993.
Tornatzky W, Miczek KA . Behavioral and autonomic responses to intermittent social stress: differential protection by clonidine and metoprolol. Psychopharmacology (Berl) 1994; 116: 346–356.
Tornatzky W, Miczek KA . Alcohol, anxiolytics and social stress in rats. Psychopharmacology (Berl) 1995; 121: 135–144.
Meerlo P, De Boer SF, Koolhaas JM, Daan S, Van den Hoofdakker RH . Changes in daily rhythms of body temperature and activity after a single social defeat in rats. Physiol Behav 1996; 59: 735–739.
Tidey JW, Miczek KA . Social defeat stress selectively alters mesocorticolimbic dopamine release: an in vivo microdialysis study. Brain Res 1996; 721: 140–149.
Von Frijtag JC, Reijmers LG, Van der Harst JE, Leus IE, Van den Bos R, Spruijt BM . Defeat followed by individual housing results in long-term impaired reward- and cognition-related behaviours in rats. Behav Brain Res 2000; 117: 137–146.
Becker C, Thiébot M-H, Touitou Y, Hamon M, Cesselin F, Benoliel J-J . Enhanced cortical extracellular levels of cholecystokinin-like material in a model of anticipation of social defeat in the rat. J Neurosci 2001; 21: 262–269.
Buwalda B, Kole MH, Veenema AH, Huininga M, de Boer SF, Korte SM et al. Long-term effects of social stress on brain and behavior: a focus on hippocampal functioning. Neurosci Biobehav Rev 2005; 29: 83–97.
Kudryavtseva NN, Bakshtanovskaya IV, Koryakina LA . Social model of depression in mice of C57BL/6J strain. Pharmacol Biochem Behav 1991; 38: 315–320.
Kudryavtseva NN, Avgustinovich DF . Behavioural and physiological markers of experimental depression induced by social conflicts. Aggress Behav 1998; 24: 271–286.
Berton O, McClung CA, Dileone RJ, Krishnan V, Renthal W, Russo SJ et al. Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science 2006; 311: 864–868.
Rygula R, Abumaria N, Flugge G, Hiemke C, Fuchs E, Ruther E et al. Citalopram counteracts depressive-like symptoms evoked by chronic social stress in rats. Behav Pharmacol 2006; 17: 19–29.
Kalueff AV, Avgustinovich DF, Kudryavtseva NN, Murphy DL . BDNF in anxiety and depression. Science 2006; 312: 1598–1599.
Berton O, Krishnan V, Nestler EJ . BDNF in anxiety and depression. Science 2006; 312: 1598–1599.
Berton O, Nestler EJ . New approaches to antidepressant drug discovery: beyond monoamines. Nat Rev Neurosci 2006; 7: 137–151.
Beinfeld MC, Meyer DK, Eskay RL, Jensen RT, Brownstein MJ . The distribution of cholecystokinin immunoreactivity in the central nervous system of the rat as determined by radioimmunoassay. Brain Res 1981; 212: 51–57.
Hökfelt T, Blacker D, Broberger C, Herrera-Marschitz M, Snyder G, Fisone G et al. Some aspects on the anatomy and function of central cholecystokinin systems. Pharmacol Toxicol 2002; 91: 382–386.
Shlik J, Vasar E, Bradwejn J . Cholecystokinin and psychiatric disorders. Role in aetiology and potential of receptor antagonists in therapy. CNS Drugs 1997; 8: 134–152.
Nevo I, Becker C, Hamon M, Benoliel J-J . Stress- and yohimbine-induced release of cholecystokinin in the frontal cortex of freely moving rat: prevention by diazepam but not ondansetron. J Neurochem 1996; 66: 2041–2049.
Bradwejn J, Koszycki D . Cholecystokinin and panic disorder: past and future clinical research strategies. Scand J Clin Lab Invest Suppl 2001; 234: 19–27.
Brodin K, Rosen A, Iwarsson K, Ogren SO, Brodin E . Increased levels of substance P and cholecystokinin in rat cerebral cortex following repeated electroconvulsive shock and subchronic treatment with a serotonin uptake inhibitor. Acta Physiol Scand 1989; 136: 613–614.
Harro J, Haidkind R, Harro M, Modiri AR, Gillberg PG, Pahkla R et al. Chronic mild unpredictable stress after noradrenergic denervation: attenuation of behavioural and biochemical effects of DSP-4 treatment. Eur Neuropsychopharmacol 1999; 10: 5–16.
Kim H, Whang WW, Kim HT, Pyun KH, Cho SY, Hahm DH et al. Expression of neuropeptide Y and cholecystokinin in the rat brain by chronic mild stress. Brain Res 2003; 983: 201–208.
Hernando F, Fuentes JA, Roques BP, Ruiz-Gayo M . The CCKB receptor antagonist, L-365,260, elicits antidepressant-type effects in the forced-swim test in mice. Eur J Pharmacol 1994; 261: 257–263.
Koszycki D, Copen J, Bradwejn J . Sensitivity to cholecystokinin-tetrapeptide in major depression. J Affect Disord 2004; 80: 285–290.
Damasio AR . Towards a neuropathology of emotion and mood. Nature 1997; 386: 769–770.
Noble F, Wank SA, Crawley JN, Bradwejn J, Seroogy KB, Hamon M et al. International union of pharmacology. XXI. Structure, distribution, and functions of cholecystokinin receptors. Pharmacol Rev 1999; 51: 745–781.
André J, Zeau B, Pohl M, Cesselin F, Benoliel J-J, Becker C . Involvement of cholecystokininergic systems in anxiety-induced hyperalgesia in male rats: behavioral and biochemical studies. J Neurosci 2005; 25: 7896–7904.
Porsolt RD, Le Pichon M, Jalfre M . Depression: a new animal model sensitive to antidepressant treatments. Nature 1977; 266: 730–732.
Calil CM, Marcondes FK . The comparison of immobility time in experimental rat swimming models. Life Sci 2006; 79: 1712–1719.
Paxinos P, Watson C . The Rat Brain in Stereotaxic Coordinates, 2nd edn. Academic Press: Sydney, 1986.
Malberg JE, Eisch AJ, Nestler EJ, Duman RS . Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J Neurosci 2000; 20: 9104–9110.
Yap JJ, Takase LF, Kochman LJ, Fornal CA, Miczek KA, Jacobs BL . Repeated brief social defeat episodes in mice: effects on cell proliferation in the dentate gyrus. Behav Brain Res 2006; 172: 344–350.
Uylings HB, van Eden CG . Qualitative and quantitative comparison of the prefrontal cortex in rat and in primates including humans. Prog Brain Res 1990; 85: 31–62.
Beaufour CC, Ballon N, Le Bihan C, Hamon M, Thiebot MH . Effects of chronic antidepressants in an operant conflict procedure of anxiety in the rat. Pharmacol Biochem Behav 1999; 62: 591–599.
Costall B, Domeney AM, Hughes J, Kelly ME, Naylor RJ, Woodruff GN . Anxiolytic effects of CCK-B antagonists. Neuropeptides 1991; 19: 65–73.
Xu XJ, Wiesenfeld-Hallin Z, Hughes J, Horwell DC, Hökfelt T . CI988, a selective antagonist of cholecystokininB receptors, prevents morphine tolerance in the rat. Br J Pharmacol 1992; 105: 591–596.
Benoliel J-J, Bourgoin S, Mauborgne A, Pohl M, Legrand JC, Hamon M et al. GABA, acting at both GABAA and GABAB receptors, inhibits the release of cholecystokinin-like material from the rat spinal cord in vitro. Brain Res 1992; 590: 255–262.
Koolhaas JM, Meerlo P, De Boer SF, Strubbe JH, Bohus B . The temporal dynamics of the stress response. Neurosci Biobehav Rev 1997; 21: 775–782.
Blanchard RJ, Hebert M, Sakai RR, McKittrick C, Henrie A, Yudko E et al. Chronic social stress: changes in behavioral and physiological indices of emotion. Aggress Behav 1998; 24: 307–321.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th edn. American Psychatric Press: Washington, DC, 1994.
Nikisch G, Mathe AA, Czernik A, Thiele J, Bohner J, Eap CB et al. Long-term citalopram administration reduces responsiveness of HPA axis in patients with major depression: relationship with S-citalopram concentrations in plasma and cerebrospinal fluid (CSF) and clinical response. Psychopharmacology (Berl) 2005; 181: 751–760.
Kunugi H, Ida I, Owashi T, Kimura M, Inoue Y, Nakagawa S et al. Assessment of the dexamethasone/CRH test as a state-dependent marker for hypothalamic–pituitary–adrenal (HPA) axis abnormalities in major depressive episode: a multicenter study. Neuropsychopharmacology 2006; 31: 212–220.
Muscat R, Willner P . Suppression of sucrose drinking by chronic mild unpredictable stress: a methodological analysis. Neurosci Biobehav Rev 1992; 16: 507–517.
Van De Poll NE, De Jonge F, Van Oyen HG, Van Pelt J . Aggressive behavior in rats: effects of winning or losing on subsequent aggressive interactions. Behav Process 1982; 7: 143–145.
Willner P, D’Aquila PS, Coventry T, Brain P . Loss of social status: preliminary evaluation of a novel animal model of depression. J Psychopharmacol 1995; 9: 207–213.
Muscat R, Papp M, Willner P . Reversal of stress-induced anhedonia by the atypical antidepressants, fluoxetine and maprotiline. Psychopharmacology (Berl) 1992; 109: 433–438.
Sheline YI, Wang PW, Gado MH, Csernansky JG, Vannier MW . Hippocampal atrophy in recurrent major depression. Proc Natl Acad Sci USA 1996; 93: 3908–3913.
Bremner JD, Narayan M, Anderson ER, Staib LH, Miller HL, Charney DS . Hippocampal volume reduction in major depression. Am J Psychiatry 2000; 157: 115–118.
Czeh B, Michaelis T, Watanabe T, Frahm J, de Biurrun G, van Kampen M et al. Stress-induced changes in cerebral metabolites, hippocampal volume, and cell proliferation are prevented by antidepressant treatment with tianeptine. Proc Natl Acad Sci USA 2001; 98: 12796–12801.
Duman RS . Depression: a case of neuronal life and death? Biol Psychiatry 2004; 56: 140–145.
Gould E, McEwen BS, Tanapat P, Galea LA, Fuchs E . Neurogenesis in the dentate gyrus of the adult tree shrew is regulated by psychosocial stress and NMDA receptor activation. J Neurosci 1997; 17: 2492–2498.
Gould E, Tanapat P, McEwen BS, Flugge G, Fuchs E . Proliferation of granule cell precursors in the dentate gyrus of adult monkeys is diminished by stress. Proc Natl Acad Sci USA 1998; 95: 3168–3171.
Tanapat P, Hastings NB, Rydel TA, Galea LA, Gould E . Exposure to fox odor inhibits cell proliferation in the hippocampus of adult rats via an adrenal hormone-dependent mechanism. J Comp Neurol 2001; 437: 496–504.
Malberg JE, Duman RS . Cell proliferation in adult hippocampus is decreased by inescapable stress: reversal by fluoxetine treatment. Neuropsychopharmacology 2003; 28: 1562–1571.
Pham K, Nacher J, Hof PR, McEwen BS . Repeated restraint stress suppresses neurogenesis and induces biphasic PSA-NCAM expression in the adult rat dentate gyrus. Eur J Neurosci 2003; 17: 879–886.
Gould E, Tanapat P . Stress and hippocampal neurogenesis. Biol Psychiatry 1999; 46: 1472–1479.
Mischoulon D . Why do antidepressants take so long to work? Am Soc Clin Psychopharmacol Progr Notes 1997; 8: 9–11.
Von Frijtag JC, Van den Bos R, Spruijt BM . Imipramine restores the long-term impairment of appetitive behavior in socially stressed rats. Psychopharmacol (Berl) 2002; 162: 232–238.
Rose C, Vargas F, Facchinetti P, Bourgeat P, Bambal RB, Bishop PB et al. Characterization and inhibition of a cholecystokinin-inactivating serine peptidase. Nature 1996; 380: 403–409.
Lodge DJ, Lawrence AJ . The neurochemical effects of anxiolytic drugs are dependent on rearing conditions in fawn-Hooded rats. Prog Neuropsychopharmacol Biol Psychiatry 2003; 27: 451–458.
Acknowledgements
This research was supported by grants from Institut National de la Santé et de la Recherche Médicale and Université Pierre et Marie Curie (Paris 6). We thank Dr A Bogdan for especially helpful statistical assistance and Eleni Paizanis (INSERM U677) and Aurélien Dauphin (Plateforme d’Imagerie Cellulaire-PS) for their invaluable recommendations and help for immunofluorescence experiments. We are grateful to Pfizer Laboratories for generous gift of CI-988.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Becker, C., Zeau, B., Rivat, C. et al. Repeated social defeat-induced depression-like behavioral and biological alterations in rats: involvement of cholecystokinin. Mol Psychiatry 13, 1079–1092 (2008). https://doi.org/10.1038/sj.mp.4002097
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.mp.4002097
Keywords
This article is cited by
-
Cholecystokinin B receptor antagonists for the treatment of depression via blocking long-term potentiation in the basolateral amygdala
Molecular Psychiatry (2023)
-
Electroacupuncture improves repeated social defeat stress-elicited social avoidance and anxiety-like behaviors by reducing Lipocalin-2 in the hippocampus
Molecular Brain (2021)
-
Potential gut–brain mechanisms behind adverse mental health outcomes of bariatric surgery
Nature Reviews Endocrinology (2021)
-
Chronic harmine treatment has a delayed effect on mobility in control and socially defeated rats
Psychopharmacology (2020)
-
Structural MRI at 7T reveals amygdala nuclei and hippocampal subfield volumetric association with Major Depressive Disorder symptom severity
Scientific Reports (2019)