Abstract
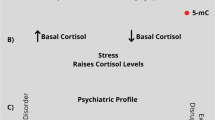
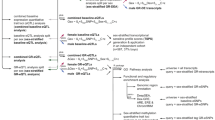
Highly prevalent stress-related disorders such as major depression (MD) are characterised by a dysregulation of the neuroendocrine system. Although heritability for these disorders is high, the role of genes in the underlying pathophysiology is poorly understood. Here, we show that polymorphic variations in genes coding for serotonin transporter (5-HTT), catechol-O-methyl transferase (COMT) and monoamine oxidase A (MAOA) as well as sex differences influence the regulation of hypothalamic–pituitary–adrenal (HPA)-axis response to acute psychological and endocrine challenges. In our sample, the effects of COMT on the release of adrenocorticotrophin hormone (ACTH) depend on the presence of the low-expression MAOA variant in the same individual. By including individuals varying in their degree of susceptibility to MD, we showed evidence of interactions between 5-HTT and MD susceptibility in baseline cortisol, and between MAOA and MD susceptibility in baseline ACTH measures, indicating a role for these genotypes in stable-state endocrine regulation. Collectively, these results indicate that the simultaneous investigation of multiple monoaminergic genes in interaction with gender have to be measured to understand the endocrine regulation of stress. These findings point towards a genetic susceptibility to stress-related disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Charmandari E, Tsigos C, Chrousos G . Endocrinology of the stress response. Annu Rev Physiol 2005; 67: 259–284.
McEwen BS . Protective and damaging effects of stress mediators. N Engl J Med 1998; 38: 171–179.
Charney DS . Psychobiological mechanisms of resilience and vulnerability: implications for successful adaptation to extreme stress. Am J Psychiatry 2004; 161: 195–216.
Sapolsky RM . Stress hormones: good and bad. Neurobiol Dis 2000; 7: 540–542.
Wong ML, Licinio J . Research and treatment approaches to depression. Nat Rev Neurosci 2001; 2: 343–351.
Moffitt TE, Caspi A, Rutter M . Strategy for investigating interactions between measured genes and measured environments. Arch Gen Psychiatry 2005; 62: 473–481.
Caspi A, Moffitt TE . Gene–environment interactions in psychiatry: joining forces with neuroscience. Nat Rev Neurosci 2006; 7: 583–590.
Meyer-Lindenberg A, Weinberger DR . Intermediate phenotypes and genetic mechanisms of psychiatric disorders. Nat Rev Neurosci 2006; 7: 818–827.
Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H et al. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 2003; 301: 386–389.
Kendler KS . ‘A gene for’: the nature of gene action in psychiatric disorders. Am J Psychiatry 2005; 162: 1243–1252.
Youdim MB, Edmondson D, Tipton KF . The therapeutic potential of monoamine oxidase inhibitors. Nat Rev Neurosci 2006; 7: 295–309.
Zubieta JK, Heitzeg MM, Smith YR, Bueller JA, Xu K, Xu Y et al. COMT val158met genotype affects mu-opioid neurotransmitter responses to a pain stressor. Science 2003; 299: 1240–1243.
Smolka MN, Schumann G, Wrase J, Grusser SM, Flor H, Mann K et al. Catechol-O-methyltransferase val158met genotype affects processing of emotional stimuli in the amygdala and prefrontal cortex. J Neurosci 2005; 25: 836–842.
Zhu QS, Grimsby J, Chen K, Shih JC . Promoter organization and activity of human monoamine oxidase (MAO) A and B genes. J Neurosci 1992; 12: 4437–4446.
Sabol SZ, Hu S, Hamer D . A functional polymorphism in the monoamine oxidase A gene promoter. Hum Genet 1998; 103: 273–279.
Lesch KP, Mossner R . Genetically driven variation in serotonin uptake: is there a link to affective spectrum, neurodevelopmental, and neurodegenerative disorders? Biol Psychiatry 1998; 44: 179–192.
Tunbridge EM, Harrison PJ, Weinberger DR . Catechol-o-methyltransferase, cognition, and psychosis: val158met and beyond. Biol Psychiatry 2006; 60: 141–151.
Kirschbaum C, Pirke KM, Hellhammer DH . The ‘Trier Social Stress Test’ – a tool for investigating psychobiological stress responses in a laboratory setting. Neuropsychobiology 1993; 28: 76–81.
Segman RH, Shefi N, Goltser-Dubner T, Friedman N, Kaminski N, Shalev AY . Peripheral blood mononuclear cell gene expression profiles identify emergent post-traumatic stress disorder among trauma survivors. Mol Psychiatry 2005; 10: 500–513.
de Kloet ER, Joels M, Holsboer F . Stress and the brain: from adaptation to disease. Nat Rev Neurosci 2005; 6: 463–475.
Heuser I, Yassouridis A, Holsboer F . The combined dexamethasone/CRH test: a refined laboratory test for psychiatric disorders. J Psychiatr Res 1994; 28: 341–356.
Schule C, Zill P, Baghai TC, Eser D, Zwanzger P, Wenig N et al. Brain-derived neurotrophic factor Val66Met polymorphism and dexamethasone/CRH test results in depressed patients. Psychoneuroendocrinology 2006; 31: 1019–1025.
Jabbi M, Kema IP, van der Pompe G, te Meerman GJ, Ormel J, den Boer JA . COMT polymorphism and susceptibility to major depression modulates psychological stress response. Psychiatr Genet (in press).
Landman-Peters KM, Hartman CA, van der Pompe G, den Boer JA, Minderaa RB, Ormel J . Gender differences in the relation between social support, problems in parent-offspring communication, and depression and anxiety. Soc Sci Med 2005; 60: 2549–2559.
Uhart M, McCaul ME, Oswald LM, Choi L, Wand GS . Gender differences in hypothalamic–pituitary–adrenal (HPA) axis reactivity. Psychoneuroendocrinology 2006; 31: 642–652.
Cook Jr EH, Courchesne R, Lord C, Cox NJ, Yan S, Lincoln A et al. Evidence of linkage between the serotonin transporter and autistic disorder. Mol Psychiatry 1997; 2: 247–250.
Pruessner JC, Kirschbaum C, Meinlschmid G, Hellhammer DH . Two formulas for computation of the area under the curve represent measures of total hormone concentration versus time-dependent change. Psychoneuroendocrinology 2003; 28: 916–931.
Plotsky PM, Owens MJ, Nemeroff CB . Psychoneuroendocrinology of depression. Hypothalamic–pituitary–adrenal axis. Psychiatr Clin North Am 1998; 21: 293–307.
Oswald LM, McCaul M, Choi L, Yang X, Wand GS . Catechol-O-methyltransferase polymorphism alters hypothalamic-pituitary-adrenal axis responses to naloxone: a preliminary report. Biol Psychiatr 2004; 55: 102–105.
Slawik M, Reisch N, Zwermann O, Maser-Gluth C, Stahl M, Klink A et al. Characterization of an adrenocorticotropin (ACTH) receptor promoter polymorphism leading to decreased adrenal responsiveness to ACTH. J Clin Endocrinol Metab 2004; 89: 3131–3137.
Dodson AM, Anderson GM, Rhoden KJ . Serotonin uptake and metabolism by cultured guinea pig airway smooth muscle cells. Pulm Pharmacol Ther 2004; 17: 19–25.
Acknowledgements
This work is funded by an NWO grant number 904-53-092. We especially thank Christian Keysers for valuable comments on earlier versions of the manuscript. We thank Iteke te Riet, Ruud Delissen and Roelie Nijzing for professional assistance, Magdalena Hubert, Astrid Brugman, Erik Nijboer, Janneke (DAJ) Dijck Brouwer, Henk Breukelman, Jan Koerts and colleagues for genotyping and endocrine analysis, our Ariadne colleagues for the HRP group selection, and Harm Jan Pot, Enno Hebekotte and Ernst Horwitz of the psychiatry department and our colleagues from GGZ Zuid and GGZ Stadskanaal for patient information. We acknowledge the individual participants and their families. We acknowledge Joop Clots, Ben Mulder and colleagues of the social sciences faculty for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
MJ, GvdP and IPK designed research. MJ performed research. IPK contributed reagents and performed genotyping. MJ and JK analysed the data. MJ wrote the paper with contributions of JK, IPK, CH, RBM, JO and JAdB.
Supplementary Information accompanies the paper on the Molecular Psychiatry website (http://www.nature.com/mp)
Supplementary information
Rights and permissions
About this article
Cite this article
Jabbi, M., Korf, J., Kema, I. et al. Convergent genetic modulation of the endocrine stress response involves polymorphic variations of 5-HTT, COMT and MAOA. Mol Psychiatry 12, 483–490 (2007). https://doi.org/10.1038/sj.mp.4001975
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.mp.4001975
Keywords
This article is cited by
-
Identification of stress resilience module by weighted gene co-expression network analysis in Fkbp5-deficient mice
Molecular Brain (2019)
-
Non-invasive genotyping with a massively parallel sequencing panel for the detection of SNPs in HPA-axis genes
Scientific Reports (2018)
-
Analysis of personality traits and their influence on the quality of life of postmenopausal women with regard to genetic factors
Annals of General Psychiatry (2016)
-
The effects of life stress and neural learning signals on fluid intelligence
European Archives of Psychiatry and Clinical Neuroscience (2015)
-
Combined Influences of Genes, Prenatal Environment, Cortisol, and Parenting on the Development of Children’s Internalizing Versus Externalizing Problems
Behavior Genetics (2015)