Abstract
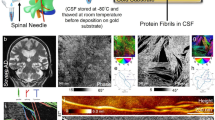
The diagnostic potential of large Aβ-peptide binding particles (LAPs) in the cerebrospinal fluid (CSF) of Alzheimer's dementia (AD) patients and non-AD controls (nAD) was evaluated. LAPs were detected by confocal spectroscopy in both groups with high inter-individual variation in number. Molecular imaging by confocal microscopy revealed that LAPs are heterogeneous superaggregates that could be subdivided morphologically into four main types (LAP1–4). LAP-4 type, resembling a ‘large chain of pearls’, was detected in 42.1% of all nAD controls but it was virtually absent in AD patients. LAP-4 type could be selectively removed by protein A beads, a clear indication that it contained immunoglobulins in addition to beta-amyloid peptides (Aβ1-42). We observed a close correlation between LAPs and immunoglobulin G (IgG) concentration in CSF in controls but not in AD patients. Double labeling of LAPs with anti-Aβ and anti-IgG antibodies confirmed that LAP-4 type consisted of Aβ and IgG aggregates. Our results assign a central role to the immune system in regulating Aβ1-42 homeostasis by clustering this peptide in immunocomplexes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lewczuk P, Esselmann H, Bibl M, Beck G, Maler JM, Otto M et al. Tau protein phosphorylated at threonine 181 in CSF as a neurochemical biomarker in Alzheimer's disease: original data and review of the literature. J of Mol Neurosci 2004; 23: 115–122.
Kropp S, Schlimme J, Bleich S, Wiltfang J, Dietrich DE, Emrich HM . Diagnostic steps in Alzheimer dementia before treatment with new antidimentives. Fortschr Neurol Psychiatr 2000; 68: 257–261.
Verbeek MM, De Jong D, Kremer HP . Brain-specific proteins in cerebrospinal fluid for the diagnosis of neurodegenerative diseases. Ann Clin Biochem 2003; 40 (Part 1): 25–40.
Andreasen N, Blennow K . CSF biomarkers for mild cognitive impairment and early Alzheimer's disease. Clin Neurol Neurosurg 2005; 107: 165–173.
Wiltfang J, Esselmann H, Smirnov A, Bibl M, Cepek L, Steinacker P et al. Beta-amyloid peptides in cerebrospinal fluid of patients with Creutzfeldt–Jakob disease. Ann Neurol 2003; 54: 263–267.
Hulstaert F, Blennow K, Ivanoiu A, Schoonderwaldt HC, Riemenschneider M, De Deyn PP et al. Improved discrimination of AD patients using beta-amyloid(1–42) and tau levels in CSF. Neurology 1999; 52: 1555–1562.
Otto M, Esselmann H, Schulz-Shaeffer W, Neumann M, Schroter A, Ratzka P et al. Decreased beta-amyloid1-42 in cerebrospinal fluid of patients with Creutzfeldt-Jakob disease. Neurology 2000; 54: 1099–1102.
Selkoe DJ . Cell biology of the amyloid beta-protein precursor and the mechanism of Alzheimer's disease. Annu Rev Cell Biol 1994; 10: 373–403.
Antzutkin ON BJ, Tycko R . Site-specific identification of non-beta-strand conformations in Alzheimer's beta-amyloid fibrils by solid-state NMR. Biophys J 2003; 84: 3326–3335.
Pitschke M, Prior R, Haupt M, Riesner D . Detection of single amyloid beta-protein aggregates in the cerebrospinal fluid of Alzheimer's patients by fluorescence correlation spectroscopy. Nat Med 1998; 4: 832–834.
Atwood CS, Martins RN, Smith MA, Perry G . Senile plaque composition and posttranslational modification of amyloid-beta peptide and associated proteins. Peptides 2002; 23: 1343–1350.
Schwille P, Bieschke J, Oehlenschlager F . Kinetic investigations by fluorescence correlation spectroscopy: the analytical and diagnostic potential of diffusion studies. Biophys Chem 1997; 66: 211–228.
Du Y, Dodel R, Hampel H, Buerger K, Lin S, Eastwood B et al. Reduced levels of amyloid beta-peptide antibody in Alzheimer disease. Neurology 2001; 57: 801–805.
Schenk D, Barbour R, Dunn W, Gordon G, Grajeda H, Guido T et al. Immunization with amyloid-beta attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature 1999; 400: 173–177.
Hock C, Konietzko U, Papassotiropoulos A, Wollmer A, Streffer J, von Rotz RC et al. Generation of antibodies specific for beta-amyloid by vaccination of patients with Alzheimer disease. Nat Med 2002; 8: 1270–1275.
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM . Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease. Neurology 1984; 34: 939–944.
Lewczuk P, Kornhuber J, Wiltfang J . The German Competence Net Dementias: standard operating procedures for the neurochemical dementia diagnostics. J Neural Transm 2006; 113: 1075–1080.
Dittrich PS, Schwille P . An integrated microfluidic system for reaction, high-sensitivity detection, and sorting of fluorescent cells and particles. Anal Chem 2003; 75: 5767–5774.
Dittrich PS, Schwille P . Spatial two-photon fluorescence cross-correlation spectroscopy for controlling molecular transport in microfluidic structures. Anal Chem 2002; 74: 4472–4479.
Ljunghusen O, Johansson A, Skogh T . Hepatic immune complex elimination studied with FITC-labelled antigen. J Immunol Methods 1990; 128: 1–7.
Citron M . Strategies for disease modification in Alzheimer's disease. Nat Rev Neurosci 2004; 5: 677–685.
Hock C, Konietzko U, Streffer JR, Tracy J, Signorell A, Muller-Tillmanns B et al. Antibodies against beta-amyloid slow cognitive decline in Alzheimer's disease. Neuron 2003; 38: 547–554.
Hyman BT, Smith C, Buldyrev I, Whelan C, Brown H, Tang MX et al. Autoantibodies to amyloid-beta and Alzheimer's disease. Ann Neurol 2001; 49: 808–810.
Pawelec G, Adibzadeh M, Pohla H, Schaudt K . Immunosenescence: ageing of the immune system. Immunol Today 1995; 16: 420–422.
Acknowledgements
This work was supported by grants from the German Federal Ministry of Education and Research (Competence Net Dementia, grant 01 GI 0420 to JW, JK, Creutzfeldt-Jacob Disease Net, grant JK 01 GI 0301 to JW, JK and HBPP-NGFN2 01 GR 0447 to JW, PL). Experiments were conducted by AWH and PSD at Experimental Biophysics Group (head: P Schwille) and at Membrane Biophysics Group (head: E Neher), both at the Max-Planck-Institute for Biophysical Chemistry, Göttingen, Germany. We thank G Beck from the Laboratory of Clinical Neurochemistry of the University Hospital of Erlangen-Nuremberg for the quantification of immunoglobulins and proteins in CSF. We thank our technician S Paul for expertise comments to the experimental setup and U Reulbach for valuable assistance on statistical calculations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Henkel, A., Dittrich, P., Groemer, T. et al. Immune complexes of auto-antibodies against Aβ1-42 peptides patrol cerebrospinal fluid of non-Alzheimer's patients. Mol Psychiatry 12, 601–610 (2007). https://doi.org/10.1038/sj.mp.4001947
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.mp.4001947
Keywords
This article is cited by
-
Exploiting microglial and peripheral immune cell crosstalk to treat Alzheimer’s disease
Journal of Neuroinflammation (2019)
-
Biomarker Modelling of Early Molecular Changes in Alzheimer’s Disease
Molecular Diagnosis & Therapy (2014)
-
Potential sources of interference on Abeta immunoassays in biological samples
Alzheimer's Research & Therapy (2012)
-
Multidimensional plasma protein separation technique for identification of potential Alzheimer’s disease plasma biomarkers: a pilot study
Journal of Neural Transmission (2012)