Abstract
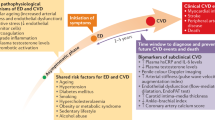
The association of erectile dysfunction (ED) and cardiovascular disease is well-documented in the literature and both conditions share risk factors. Therefore, it is difficult to distinguish the effect of underlying disease and adverse effects of the drugs and/or interactions between ED drugs and drugs implemented for cardiovascular disease. The known interactions of systemic administered drugs for ED with drugs for cardiovascular disease are mainly pharmacodynamic. Thus, nitrates enhance the production of cyclic GMP and combined with phosphodiesterase type-5 inhibitors this can lead to severe hypotension. The same is the case for the treatment with phentolamine in patients treated with β-adrenoceptor antagonists. Due to increased partial thromboplastin time, the risk of bleeding is enhanced for intracavernous alprostadil injection in heparin-treated patients. Pharmacokinetic interactions of clinical importance have been described for ED drugs with other therapeutic groups such as sildenafil with the antifungal drug, ketoconazole, and apomorphine with the antiparkinson drug, entacapon. Although sildenafil and antihypertensive dihydropyridines like amlodipine are metabolized by the same cytochrome P450 enzyme, CYP3A4 in the liver, the combination of these drugs does not exhibit a synergistic blood pressure lowering action. Unfortunately documentation concerning drug interactions is often poor and occasional.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Feldman HA et al. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study J Urol 1994 151: 54–61
Martin-Morales A et al. Prevalence and independent risk factors for erectile dysfunction in Spain: results of the Epidemiologia de la Disfuncion Erectil Masculina Study J Urol 2001 166: 569–574
Johannes CB et al. Incidence of erectile dysfunction in men 40 to 69 y old: longitudinal results from the Massachusetts male aging study J Urol 2000 163: 460–463
Gundle MJ et al. Psychosocial outcome after coronary artery surgery Am J Psychiatry 1980 137: 1591–1594
Wabrek AJ, Burchell RC . Male sexual dysfunction associated with coronary heart disease Arch Sex Behav 1980 9: 69–75
Chang M, Hahn RA, Teutsch SM, Hutwagner LC . Multiple risk factors and population attributable risk for ischemic heart disease mortality in the United States, 1971–1992 J Clin Epidemiol 2001 54: 634–644
Virag R, Bouilly P, Frydman D . Is impotence an arterial disorder? A study of arterial risk factors in 440 impotent men Lancet 1985 1: 181–184
Shabsigh R, Fishman IJ, Schum C, Dunn JK . Cigarette smoking and other vascular risk factors in vasculogenic impotence Urology 1991 38: 227–231
Morley JE et al. Relationship of penile brachial pressure index to myocardial infarction and cerebrovascular accidents in older men Am J Med 1988 84: 445–448
Jensen J et al. The prevalence and etiology of impotence in 101 male hypertensive outpatients Am J Hypertens 1999 12: 271–275
Fattinger K et al. Epidemiology of drug exposure and adverse drug reactions in two Swiss departments of internal medicine Br J Clin Pharmacol 2000 49: 158–167
1999 World Health Organization-International Society of Hypertension Guidelines for the Management of Hypertension. Guidelines Subcommittee J Hypertens 1999 17: 151–183
McCarron DA . Diuretic therapy for mild hypertension: the ‘real’ cost of treatment Am J Cardiol 1984 53: 9A–11A
MRC trial of treatment of mild hypertension: principal results. Medical Research Council Working Party Br Med J (Clin Res Ed) 1985 291: 97–104
Chang SW et al. The impact of diuretic therapy on reported sexual function Arch Intern Med 1991 151: 2402–2408
Grimm RH Jr et al. Long-term effects on sexual function of five antihypertensive drugs and nutritional hygienic treatment in hypertensive men and women. Treatment of Mild Hypertension Study (TOMHS) Hypertension 1997 29: 8–14
Bulpitt CJ et al. The effects of anti-hypertensive drugs on sexual function in men and women: a report from the DHSS Hypertension Care Computing Project (DHCCP) J Hum Hypertens 1989 3: 53–56
Prisant LM et al. Self reported sexual dysfunction in men and women treated with bisoprolol, hydrochlorothiazide, enalapril, amlodipine, placebo, or bisoprolol/hydrochlorothia-zide J Clin Hypertens (Greenwich) 1999 1: 22–26
Burchardt M et al. Hypertension is associated with severe erectile dysfunction J Urol 2000 164: 1188–1191
Cohen JS . Adverse drug effects, compliance, and initial doses of antihypertensive drugs recommended by the Joint National Committee vs the Physicians' Desk Reference Arch Intern Med 2001 161: 880–885
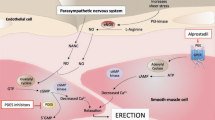
Andersson KE, Wagner G . Physiology of penile erection Physiol Rev 1995 75: 191–236
Simonsen U et al. Adrenoceptor-mediated regulation of the contractility in horse penile resistance arteries J Vasc Res 1997 34: 90–102
Becker AJ et al. Plasma levels of cavernous and systemic norepinephrine and epinephrine in men during different phases of penile erection J Urol 2000 164: 573–577
Adverse reactions to bendrofluazide and propranolol for the treatment of mild hypertension. Report of Medical Research Council Working Party on Mild to Moderate Hypertension Lancet 1981 2: 539–543
Croog SH et al. The effects of antihypertensive therapy on the quality of life New Engl J Med 1986 314: 1657–1664
Broekman CP, Haensel SM, Van de Ven LL, Slob AK . Bisoprolol and hypertension: effects on sexual functioning in men J Sex Marital Ther 1992 18: 325–331
Fletcher AE et al. The effects of verapamil and propranolol on quality of life in hypertension J Hum Hypertens 1989 3: 125–130
Hong CY et al. Calcium antagonists stimulate sperm motility in ejaculated human semen Br J Clin Pharmacol 1985 19: 45–49
Fogari R et al. Sexual function in hypertensive males treated with lisinopril or atenolol: a cross-over study Am J Hypertens 1998 11: 1244–1247
Fogari R et al. Sexual activity in hypertensive men treated with valsartan or carvedilol: a crossover study Am J Hypertens 2001 14: 27–31
Kaplan SA et al. Combination therapy using oral alpha-blockers and intracavernosal injection in men with erectile dysfunction Urology 1998 52: 739–743
Marquer C, Bressolle F . Moxisylyte: a review of its pharmacodynamic and pharmacokinetic properties, and its therapeutic use in impotence Fundam Clin Pharmacol 1998 12: 377–387
Jaffe A et al. Erectile dysfunction in hypertensive subjects. Assessment of potential determinants Hypertension 1996 28: 859–862
Azadzoi KM, Goldstein I . Erectile dysfunction due to atherosclerotic vascular disease: the development of an animal model J Urol 1992 147: 1675–1681
Ari G, Vardi Y, Finberg JP . Nitric oxide and penile erection in streptozotocin-diabetic rats Clin Sci (Colch) 1999 96: 365–371
Martinez C, Simonsen U . Decreased erectile function in renal hypertensive rats Pharmacol Toxicol 2001 89: 122
Saenz dT I et al. Impaired neurogenic and endothelium-mediated relaxation of penile smooth muscle from diabetic men with impotence New Engl J Med 1989 320: 1025–1030
Garban H et al. Effect of aging on nitric oxide-mediated penile erection in rats Am J Physiol 1995 268: H467–H475
Azadzoi KM et al. Relationship between cavernosal ischemia and corporal veno-occlusive dysfunction in an animal model J Urol 1997 157: 1011–1017
Okabe H et al. The penis is not protected—in hypertension there are vascular changes in the penis which are similar to those in other vascular beds Int J Impot Res 1999 11: 133–140
Toblli JE et al. Morphological changes in cavernous tissue in spontaneously hypertensive rats Am J Hypertens 2000 13: 686–692
Hale TM, Okabe H, Heaton JP, Adams MA . Antihypertensive drugs induce structural remodeling of the penile vasculature J Urol 2001 166: 739–745
Davies MK, Gibbs CR, Lip GY . ABC of heart failure. Management: diuretics, ACE inhibitors, and nitrates Br Med J 2000 320: 428–431
Packer M et al. The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. US Carvedilol Heart Failure Study Group New Engl J Med 1996 334: 1349–1355
Pitt B et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators New Engl J Med 1999 341: 709–717
Squire IB, Barnett DB . The rational use of beta-adrenoceptor blockers in the treatment of heart failure. The changing face of an old therapy Br J Clin Pharmacol 2000 49: 1–9
Cohn JN et al. A comparison of enalapril with hydralazine-isosorbide dinitrate in the treatment of chronic congestive heart failure New Engl J Med 1991 325: 303–310
The effect of digoxin on mortality and morbidity in patients with heart failure. The Digitalis Investigation Group New Engl J Med 1997 336: 525–533
Gupta S et al. A possible mechanism for alteration of human erectile function by digoxin: inhibition of corpus cavernosum sodium/potassium adenosine triphosphatase activity J Urol 1998 159: 1529–1536
Prieto D, Simonsen U, Hernandez M, Garcia-Sacristan A . Contribution of K+ channels and ouabain-sensitive mechanisms to the endothelium-dependent relaxations of horse penile small arteries Br J Pharmacol 1998 123: 1609–1620
Gupta S et al. Possible role of Na(+)-K(+)-ATPase in the regulation of human corpus cavernosum smooth muscle contractility by nitric oxide Br J Pharmacol 1995 116: 2201–2206
Neri A et al. The effect of long-term administration of digoxin on plasma androgens and sexual dysfunction J Sex Marital Ther 1987 13: 58–63
Frick MH et al. Helsinki Heart Study: primary-prevention trial with gemfibrozil in middle-aged men with dyslipidemia. Safety of treatment, changes in risk factors, and incidence of coronary heart disease New Engl J Med 1987 317: 1237–1245
Staels B et al. Mechanism of action of fibrates on lipid and lipoprotein metabolism Circulation 1998 98: 2088–2093
Randomised trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S) Lancet 1994 344: 1383–1389
Shepherd J et al. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group New Engl J Med 1995 333: 1301–1307
Wei M et al. Total cholesterol and high density lipoprotein cholesterol as important predictors of erectile dysfunction Am J Epidemiol 1994 140: 930–937
Schneider J, Kaffarnik H . Impotence in patients treated with clofibrate Atherosclerosis 1975 21: 455–457
Bain SC, Lemon M, Jones AF . Gemfibrozil-induced impotence Lancet 1990 336: 1389
Bruckert E, Giral P, Heshmati HM, Turpin G . Men treated with hypolipidaemic drugs complain more frequently of erectile dysfunction J Clin Pharm Ther 1996 21: 89–94
Kersten S, Wahli W . Peroxisome proliferator activated receptor agonists EXS 2000 89: 141–151
Xu S et al. PPARalpha-dependent induction of liver microsomal esterification of estradiol and testosterone by a prototypical peroxisome proliferator Endocrinology 2001 142: 3554–3557
Jackson G . Simvastatin and impotence Br Med J 1997 315: 31–32
Kostis JB, Rosen RC, Wilson AC . Central nervous system effects of HMG CoA reductase inhibitors: lovastatin and pravastatin on sleep and cognitive performance in patients with hypercholesterolemia J Clin Pharmacol 1994 34: 989–996
Pedersen TR, Faergemann O . Simvastatin seems unlikely to cause impotence Br Med J 1999 318: 192
De Siati M et al. Priapism as a complication of heparin therapy Arch Ital Urol Androl 1999 71: 201–202
Kulmala RV, Lehtonen TA, Tammela TL . Preservation of potency after treatment for priapism Scand J Urol Nephrol 1996 30: 313–316
Helgason AR et al. Factors associated with waning sexual function among elderly men and prostate cancer patients J Urol 1997 158: 155–159
Danjou P et al. Assessment of erectogenic properties of apomorphine and yohimbine in man Br J Clin Pharmacol 1988 26: 733–739
Segraves RT, Bari M, Segraves K, Spirnak P . Effect of apomorphine on penile tumescence in men with psychogenic impotence J Urol 1991 145: 1174–1175
Heaton JP et al. Recovery of erectile function by the oral administration of apomorphine Urology 1995 45: 200–206
Heaton JP . Key issues from the clinical trials of apomorphine SL World J Urol 2001 19: 25–31
Seeman P, Van Tol HH . Dopamine receptor pharmacology Trends Pharmacol Sci 1994 15: 264–270
Fagan TC et al. Cardiovascular safety of sublingual apomorphine in patients on stable doses of oral antihypertensive agents and nitrates Am J Cardiol 2001 88: 760–766
Dula E et al. Efficacy and safety of fixed-dose and dose-optimization regimens of sublingual apomorphine versus placebo in men with erectile dysfunction. The Apomorphine Study Group Urology 2000 56: 130–135
Montastruc P, Damase-Michel C, Montastruc JL . Apomorphine potentiates vagal bradycardia Eur J Pharmacol 1989 166: 511–514
Dlewati A, Watkins HO, Lokhandwala MF . Effects of SK&F 85174, a DA-1/DA-2 receptor agonist, on pre- and postganglionic sympathetic neurotransmission to the heart Eur J Pharmacol 1989 164: 197–203
Mukai M et al. The inhibition of ganglionic transmission via presynaptic dopamine DA1 and postsynaptic DA2 receptor activation in the canine cardiac sympathetic ganglia J Pharmacol Exp Ther 1996 279: 822–829
MicroMedex Incorporated. www.smi.dk 2000
Argiolas A, Hedlund H . The pharmacology and clinical pharmacokinetics of apomorphine SL BJU Int 2001 88: (Suppl 3) 18–21
Traish A et al. Phentolamine mesylate relaxes penile corpus cavernosum tissue by adrenergic and non-adrenergic mechanisms Int J Impot Res 1998 10: 215–223
Goldstein I . Oral phentolamine: an alpha-1, alpha-2 adrenergic antagonist for the treatment of erectile dysfunction Int J Impot Res 2000 12: (Suppl 1) S75–S80
Andersson KE, Stief C . Oral alpha adrenoceptor blockade as a treatment of erectile dysfunction World J Urol 2001 19: 9–13
Seideman P et al. Prazosin first dose phenomenon during combined treatment with a beta- adrenoceptor blocker in hypertensive patients Br J Clin Pharmacol 1982 13: 865–870
Tam SW, Worcel M, Wyllie M . Yohimbine: a clinical review Pharmacol Ther 2001 91: 215–243
Winter JC, Rabin RA . Yohimbine as a serotonergic agent: evidence from receptor binding and drug discrimination J Pharmacol Exp Ther 1992 263: 682–689
Ernst E, Pittler MH . Yohimbine for erectile dysfunction: a systematic review and meta-analysis of randomized clinical trials J Urol 1998 159: 433–436
Tallentire D et al. Modulation of sexual behaviour in the rat by a potent and selective alpha 2-adrenoceptor antagonist, delequamine (RS-15385-197) Br J Pharmacol 1996 118: 63–72
Mathes CW, Smith ER, Popa BR, Davidson JM . Effects of intrathecal and systemic administration of buspirone on genital reflexes and mating behavior in male rats Pharmacol Biochem Behav 1990 36: 63–68
Mansoor GA . Herbs and alternative therapies in the hypertension clinic Am J Hypertens 2001 14: 971–975
Lacomblez L et al. Effect of yohimbine on blood pressure in patients with depression and orthostatic hypotension induced by clomipramine Clin Pharmacol Ther 1989 45: 241–251
Andersson KE . Pharmacology of penile erection Pharmacol Rev 2001 53: 417–450
Langtry HD, Markham A . Sildenafil: a review of its use in erectile dysfunction Drugs 1999 57: 967–989
Goldstein I et al. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group New Engl J Med 1998 338: 1397–1404
FDA. FDA Postmarketing safety of sildenafil citrate (Viagra) Food and Drug Administration Website: www.fda.gov 1998
Feenstra J, Drie-Pierik RJ, Lacle CF, Stricker BH . Acute myocardial infarction associated with sildenafil Lancet 1998 352: 957–958
Conti CR, Pepine CJ, Sweeney M . Efficacy and safety of sildenafil citrate in the treatment of erectile dysfunction in patients with ischemic heart disease Am J Cardiol 1999 83: 29C–34C
Herrmann HC, Chang G, Klugherz BD, Mahoney PD . Hemodynamic effects of sildenafil in men with severe coronary artery disease New Engl J Med 2000 342: 1622–1626
Przyklenk K, Kloner RA . Sildenafil citrate (Viagra) does not exacerbate myocardial ischemia in canine models of coronary artery stenosis J Am Coll Cardiol 2001 37: 286–292
Wallis RM, Corbin JD, Francis SH, Ellis P . Tissue distribution of phosphodiesterase families and the effects of sildenafil on tissue cyclic nucleotides, platelet function, and the contractile responses of trabeculae carneae and aortic rings in vitro Am J Cardiol 1999 83: 3C–12C
Webb DJ et al. Sildenafil citrate potentiates the hypotensive effects of nitric oxide donor drugs in male patients with stable angina J Am Coll Cardiol 2000 36: 25–31
Ishikura F et al. Effects of sildenafil citrate (Viagra) combined with nitrate on the heart Circulation 2000 102: 2516–2521
Zusman RM, Prisant LM, Brown MJ . Effect of sildenafil citrate on blood pressure and heart rate in men with erectile dysfunction taking concomitant antihypertensive medication. Sildenafil Study Group J Hypertens 2000 18: 1865–1869
Ballard SA et al. Effects of sildenafil on the relaxation of human corpus cavernosum tissue in vitro and on the activities of cyclic nucleotide phosphodiesterase isozymes J Urol 1998 159: 2164–2171
McAuley IW, Kim NN, Min K, Goldstein I, Traish AM . Intracavernosal sildenafil facilitates penile erection independent of the nitric oxide pathway J Androl 2001 22: 623–628
Zhao L et al. Sildenafil inhibits hypoxia-induced pulmonary hypertension Circulation 2001 104: 424–428
Geelen P et al. Sildenafil (Viagra) prolongs cardiac repolarization by blocking the rapid component of the delayed rectifier potassium current Circulation 2000 102: 275–277
Medina P et al. Inhibition of neuroeffector transmission in human vas deferens by sildenafil Br J Pharmacol 2000 131: 871–874
Stief CG et al. Effects of sildenafil on cAMP and cGMP levels in isolated human cavernous and cardiac tissue Urology 2000 55: 146–150
Levesque PC et al. Anion and cation modulation of the guinea-pig ventricular action potential during beta-adrenoceptor stimulation Pflugers Arch 1993 424: 54–62
Sugiyama A et al. Cardiac electrophysiologic and hemodynamic effects of sildenafil, a PDE5 inhibitor, in anesthetized dogs J Cardiovasc Pharmacol 2001 38: 940–946
Warrington JS, Shader RI, von Moltke LL, Greenblatt DJ . In vitro biotransformation of sildenafil (Viagra): identification of human cytochromes and potential drug interactions Drug Metab Dispos 2000 28: 392–397
Dresser GK, Spence JD, Bailey DG . Pharmacokinetic-pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition Clin Pharmacokinet 2000 38: 41–57
Merry C et al. Interaction of sildenafil and indinavir when co-administered to HIV-positive patients AIDS 1999 13: F101–F107
Zusman RM, Morales A, Glasser DB, Osterloh IH . Overall cardiovascular profile of sildenafil citrate Am J Cardiol 1999 83: 35C–44C
Webb DJ, Freestone S, Allen MJ, Muirhead GJ . Sildenafil citrate and blood-pressure-lowering drugs: results of drug interaction studies with an organic nitrate and a calcium antagonist Am J Cardiol 1999 83: 21C–28C
Buvat J et al. Double-blind multicenter study comparing alprostadil alpha-cyclodextrin with moxisylyte chlorhydrate in patients with chronic erectile dysfunction J Urol 1998 159: 116–119
Linet OI, Ogrinc FG . Efficacy and safety of intracavernosal alprostadil in men with erectile dysfunction. The Alprostadil Study Group New Engl J Med 1996 334: 873–877
Porst H . The rationale for prostaglandin E1 in erectile failure: a survey of worldwide experience J Urol 1996 155: 802–815
Porst H et al. Intracavernous Alprostadil Alfadex—an effective and well tolerated treatment for erectile dysfunction. Results of a long-term European study Int J Impot Res 1998 10: 225–231
Limoge JP, Olins E, Henderson D, Donatucci CF . Minimally invasive therapies in the treatment of erectile dysfunction in anticoagulated cases: a study of satisfaction and safety J Urol 1996 155: 1276–1279
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Simonsen, U. Interactions between drugs for erectile dysfunction and drugs for cardiovascular disease. Int J Impot Res 14, 178–188 (2002). https://doi.org/10.1038/sj.ijir.3900846
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3900846
Keywords
This article is cited by
-
Aging and pathogenesis of erectile dysfunction
International Journal of Impotence Research (2004)
-
Disfunzione erettile
L'Endocrinologo (2002)