Abstract
Overexpression of the HER-2/neu oncogene appears to have prognostic significance in breast cancer. Recently, some have reported a relationship between increased immunohistochemical expression in osteosarcoma and poor clinical outcome. Despite limited data, a pilot trial of Herceptin, which targets the oncogene product, has been initiated for the therapy of some metastatic osteosarcomas (CCG-P9852). Archival formalin-fixed, paraffin-embedded tissue obtained from 41 patients diagnosed with osteosarcoma was examined immunohistochemically by 2 antibodies against the HER-2/neu oncogene product: CB-11 (monoclonal, 1/100) and Oncor (polyclonal, 1/200). All but one tumor (case of recurrent dedifferentiated parosteal osteosarcoma) represented primary tumor samples; when applicable, only prechemotherapy biopsies were analyzed. The study sample included the full spectrum of histologic subtypes and grades of osteosarcoma (25 conventional high grade; 3 telangiectatic; 1 small cell; 6 parosteal; 1 periosteal; and 5 low-grade intramedullary). A case of metastatic breast cancer with known overexpression of the HER-2/neu oncogene served as the positive control. Complete membranous positivity, considered prognostically significant in breast cancer, was not seen in any of our osteosarcoma cases. At least focal cytoplasmic positivity was documented in 40 (98%) tumors using the CB11 antibody and in 34 (83%) using the Oncor antibody. The intensity of the cytoplasmic staining (0, 1–3+) did not correlate with histologic subtype/grade, response to chemotherapy (<90% versus ≥90% necrosis), metastasis, or survival. Immunohistochemical overexpression of the HER-2/neu oncogene, defined as complete membranous positivity, is not present in our series of osteosarcomas. Cytoplasmic positivity is observed in most osteosarcomas, irrespective of histologic subtype/grade, and is not associated with response to preoperative chemotherapy or disease progression.
Similar content being viewed by others
INTRODUCTION
Recently, Gorlick (1) and colleagues analyzed paraffin-embedded tissue from 53 patients with high-grade osteosarcoma, diagnosed and treated at Memorial Sloan Kettering Cancer Center between 1986 and 1993. Immunoexpression of HER-2/neu was associated with a “significantly worse histologic response” and decreased survival. Similary, Onda and others (2), using immunoblotting, also found a correlation between decreased survival and “apparent ErbB-2 expression.” Despite limited data, especially when compared with breast cancer, pilot trials using Herceptin in recurrent osteosarcomas that overexpress HER-2/neu are now underway (CCG-P9852). Because of concerns raised in the breast cancer literature regarding methodologies as well as the reproducibility of the detection and prognostic significance of HER-2/neu, we report herein our clinicopathologic and immunohistochemical results among 41 patients with osteosarcoma using 2 different antibodies. We also sought to determine the significance of these findings among a variety of osteosarcoma subtypes (including surface forms) and grades, which to our knowledge, has not been reported.
MATERIALS AND METHODS
Archival formalin-fixed, paraffin-embedded material and medical records from 41 consecutive patients diagnosed with osteosarcoma were obtained from the files of the University of North Carolina (21 patients) and Wake Forest University Baptist Medical Center (20 patients). If applicable, only prechemotherapy tissue from the primary tumor (25 patients) was used. One locally recurrent, dedifferentiated parosteal osteosarcoma was examined. No metastases were evaluated. All were examined immunohistochemically by 2 antibodies against the HER-2/neu oncogene product. The antibodies and their specific methodologies are as follows.
For both antibodies, the procedure was similar, with the exception of the dilutions used and the fact that antigen retrieval was only used with the polyclonal Oncor antibody.
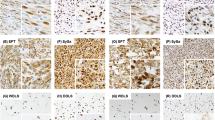
Five-micrometer sections were cut and mounted onto Probe On Plus slides (Fisher Scientific Biomedical Sciences, Inc., Swedesboro, NJ). After deparaffinization in xylene, the slides were rehydrated in a graded series of alcohol, rinsed under running water, and placed into a MicroProbe slide holder (Fisher). Slides requiring antigen retrieval (Oncor polyclonal) were immersed in a 1 × solution of Citra (BioGenex, San Ramon, CA) and placed in a Black and Decker steamer for 30 minutes, after which the slides were removed and allowed to cool at room temperature for 20 minutes. All slides were rinsed 4 times in 1 × Automation Buffer (Biomeda, Foster City, CA), and the endogenous peroxidase was quenched for 5 minutes at 37° C using Endogenous Blocking Reagent (Dakopatts, Carpinteria, CA). An additional 4 rinses with Automation Buffer were performed, followed by incubation in Protein Blocking Reagent (BioGenex) for 7 minutes at 37° C. The Protein Blocking Reagent was drained off, and the slides were incubated in either a 1:200 dilution of polyclonal anti-HER-2/neu (Oncor, Inc., Gaithersburg, MD, Lot 7C123) or a 1:100 dilution of mouse anti-HER-2/neu (BioGenex, clone CB11, Lot MU1340799) for 30 minutes at 37° C. The slides were then rinsed 4 times in Automation Buffer. Detection of the primary antibody involved the Super Sensitive Multi Link Detection Kit (Biogenex) using the avidin-biotin/horseradish peroxidase (HRP) methodology. The slides were incubated in the biotinylated secondary antibody for 7 minutes at 37° C, followed by 4 rinses with Automation Buffer. Next, the slides were incubated in the HRP label for 7 minutes at 37° C and again rinsed in Automation Buffer. 3,3′-diaminobenzidene tetrahydrochloride (3,3′-diaminobenzidine) chromogen (substrate) was used for the visualization of the antibody/enzyme complex. 3,3′-Diaminobenzidine was applied to the slides for 5 minutes at 37° C while development was visualized under a microscope intermittently. The slides were rinsed in Automation Buffer, removed from the MicroProbe slide holder, and placed in running tap water. The sections were then counterstained using Harris' hematoxylin (DAKO) for 5 seconds, rinsed in running tap water, and “blued” by placing them in Automation Buffer. The sections were again placed in running tap water, dehydrated in a graded series of alcohols, cleared in 2 changes of xylene, and permanently mounted with PerMount (Fisher). Appropriate positive and negative controls were used throughout the procedures. Sections of a case of breast cancer with known overexpression of HER-2/neu oncogene served as the positive control (Fig. 1).
All cases were examined for positivity (membranous versus cytoplasmic) and quantitated, if applicable. Cytoplasmic positivity was scored as follows: negative or 0 = no staining or staining in less than 10% of cells; 1+ = faint/barely perceptible staining in more than 10% of cells; 2+ = weak to moderate staining in more than 10% of cells; and 3+ = moderate to strong staining in more than 10% of cells. We purposely used only blocks of tissue with viable-appearing tumor, avoiding areas of necrosis.
Statistical analysis to evaluate the amount of agreement between the 2 antibodies was based on the kappa statistic. For survival analysis, the log-rank test statistic and Kaplan-Meier estimates were evaluated. To test the correlation of staining intensity with histologic subtype/grade, response to chemotherapy, and metastasis, the Mantel-Haenszel correlation statistic was used. Fisher's exact test was used to test for an association in general, not just a correlation. In summary, the Mantel–Haenszel nonzero correlation assumes the intensity of staining is ordinal, whereas the Fisher's exact test does not. The latter can be thought of as comparing among levels of staining without assuming that they are ordered.
RESULTS
The clinicopathologic features of the study cases and the scoring results of the 2 antibodies are summarized in Table 1. All but 1 patient (Patient 26), who was diagnosed in 1985, were initially diagnosed and treated between the years of 1990–2000. There were 22 males and 19 females ranging from 7 to 88 years of age (mean, 29 years). The study sample included the full spectrum of histologic subtypes and grades of osteosarcoma (25 conventional high grade; 3 telangiectatic; 1 small cell; 6 parosteal; 1 perisoteal; and 5 low-grade intramedullary forms). As stated in Table 1, chemotherapy refers only to neoadjuvant preoperative chemotherapy, not postoperative chemotherapy. Only two older adult patients with high-grade osteosarcoma (Patients 7 and 24) did not receive any chemotherapy because of preexisting health conditions and poor compliance, respectively. Complete membranous positivity was not seen in any of our osteosarcoma cases. At least focal cytoplasmic positivity (more than 10% of cells) was documented in 40 (98%) tumors using the CB11 antibody and in 34 (83%) using the Oncor antibody (Fig. 2). Osteoclast-type giant cells also showed cytoplasmic positivity. There was no difference observed among decalcification versus nondecalcification specimens. The weighted kappa statistic indicated that there was poor agreement between the 2 antibodies (weighted kappa =.079 with CI = −0.164, 0.321). With the values of the antibodies collapsed to positive (2–3+) vs. negative (0–1+), there remained poor agreement between the 2 measures (kappa =.093, 95% CI = −0.171, 0.357). This appears driven by the 9 cases that were positive for CB11 but negative for the Oncor polyclonal.
The intensity of the cytoplasmic staining for CB11 did not correlate with histologic subtype/grade (high vs. low, Mantel-Haenszel P value =.202; Fisher's exact test, P =.156), response to chemotherapy (less than 90% versus more than 90% necrosis for conventional high-grade subtype; Fisher's exact test, P =.320), metastasis (Mantel-Haenszel P value =.821; Fisher's exact test, P =.796), or survival (alive versus dead of disease; log-rank P value =.861).
Similarly, the intensity of the cytoplasmic staining for Oncor polyclonal was not significantly associated with response to chemotherapy (less than 90% vs. more than 90% necrosis for conventional high-grade subtype; Mantel-Haenszel P value =.818; Fisher's exact test, P =.157), metastasis (Mantel-Haenszel P value =.681; Fisher's exact test, P =.952), or survival (alive versus dead of disease; log-rank P value =.444). Kaplan-Meier survival estimates for levels of CB11 and Oncor are illustrated in Figure 3. For histologic grade, the Fisher's exact test did detect an association between the level of staining and the percentage of high-grade tumors (P =.019); however, the association is not linear. That is, the percentage of high grade does not increase linearly as the levels of staining increase; therefore, it is not detected (or significant) by the Mantel-Haenszel nonzero correlation test (P value =.341).
DISCUSSION
The HER-2/neu oncogene encodes a 185-kDa transmembrane protein with significant homology to epidermal growth factor receptor and is a member of the tyrosine kinase receptor family. Numerous studies have documented a significant relationship between amplification and overexpression of the HER-2/neu (c-erbB-2) oncogene and poor prognosis among patients with breast cancer (3, 4, 5, 6). Recent investigations have focused on HER-2/neu status in breast cancer as a predictor of response and/or resistance to certain chemotherapeutic agents, including Herceptin (trastuzumab), cisplatinum, cyclophosphamide, methotrexate, and doxorubicin (7, 8). However, because of various protocols, methodologies, and scoring systems for the detection of HER-2/neu, considerable controversy remains and some studies have found conflicting results, especially among patients with node-negative breast cancer (9, 10). Immunohistochemistry is the most common method for detection of HER-2/neu overexpression but is significantly affected by the type of tissue used (frozen versus fixed) and the sensitivity of different antibodies, reportedly ranging from 6–80% (11). In most series using immunohistochemistry, only a membranous pattern of staining has been considered specific for HER-2/neu and correlated with gene expression (12). Conversely, cytoplasmic staining is generally considered nonspecific. However, we should point out that cytoplasmic positivity has been reported as being prognostically significant in other carcinomas, such as bladder, nasopharyngeal, colorectal, and papillary thyroid carcinoma (13, 14, 15, 16). At the present time, a precise definition of HER-2/neu overexpression and its significance remains unclear, and there is no consensus as to the best method for determining HER-2/neu status (12). Against this backdrop of controversy regarding methodologies and the prognostic significance of HER-2/neu oncoprotein expression in breast cancer, we sought to examine the limited and recent evidence suggesting a relationship between HER-2/neu oncoprotein overexpression and survival in osteosarcoma.
In 1996, Onda et al. (2) documented “ErbB-2” expression in 11 of 26 osteosarcoma cases using immunoblot analysis and confirmed by immunohistochemical studies (CB11 antibody). Regarding the level of staining intensity, individual cases were qualitatively designated as little, weak, moderate, and high, yielding scores of 0, 1+, 2+, and 3+, respectively (2). No quantification (e.g., number or percentage of cells positive) requirement was documented. It appears that any membranous positivity (e.g., ≥1+) was considered significant, but it is unclear whether the authors required complete (or incomplete) membranous staining. However, most important, “neither amplification of the c-erbB-2 gene nor evidence of significant genetic mutation was found in these osteosarcomas” by Southern blot hybridization (2). The authors reported a strong correlation with immunoblotting and the development of pulmonary metastases. Gorlick and colleagues documented high levels of HER-2/neu expression in 20 (of 47) osteosarcoma samples. Positive immunoexpression was defined as membranous positivity and scored using the 5B5 antibody according to the following parameters: 0 (no staining), 1+ (1–25% staining), 2+ (26–50% staining), 3+ (51–75% staining), and 4+ (76–100% staining). According to the authors, greater than or equal to 2+ “correlated with at least 1+ staining using the controls and criteria provided in the HercepTest kit (1).” However, we should point out that using the criteria employed by the HercepTest kit (DAKO), scores of 0 (no staining or staining in less than 10% of cells) and 1+ (incomplete membranous staining more than 10% of cells) are considered negative, whereas scores of 2+ (mild to moderate complete membranous staining in more than 10% of cells) and 3+ (strong complete membranous staining in more than 10% of cells) are considered positive indicators of HER-2/neu oncogene overexpression (17). Furthermore, in breast cancer, those tumors with 2+ immunoexpression appear to be a heterogeneous group of tumors, with less than half showing true amplification of the HER-2/neu oncogene by FISH analysis (17). Recently, among 22 pretreatment osteosarcoma samples, Zhou and colleagues (18) reported a correlation between “cytoplasmic staining” of HER-2/neu and increased risk of pulmonary metastasis. Maitra and others (19) evaluated 18 high grade osteosarcomas using both immunohistochemistry and FISH analysis. Immunohistochemical overexpression of HER-2/neu was not observed. Furthermore, by FISH analysis, no evidence of amplification of the HER-2/neu oncogene was seen (19). In our series, we evaluated prechemotherapy samples (if applicable) from 41 osteosarcoma cases, 25 of which represented the conventional high-grade form. Using 2 antibodies to the HER-2/neu oncogene product, complete membranous staining in more than 10% of cells was not observed in any of the osteosarcomas. However, at least focal cytoplasmic positivity was seen in 40 (98%) tumors using the CB11 antibody and in 34 (83%) tumors using the Oncor polyclonal antibody. The intensity of staining (0, 1–3+) did not correlate with histologic subtype/grade, response to chemotherapy, metastasis, or survival.
Although immunohistochemical staining appears to be the predominant method for determining HER-2/neu oncogene overexpression, the results may be substantially affected by technical issues, including fixation duration and prolonged storage. Another potential problem for immunohistochemical determination is the type and sensitivities of the antibodies used. This may be even further compounded by the different scoring systems described. In most studies, the incidence of HER-2/neu oncogene overexpression in breast cancer ranges from 20–30% (12). Several investigators have reported variable results for HER-2/neu oncogene immunoexpression among different antibodies (20, 21). Using the scoring system of the HercepTest, Maia evaluated 69 breast cancers using the HercepTest kit and a polyclonal antibody from Oncor, Inc (20). Thirteen tumors (19%) appeared positive for overexpression with the Oncor antibody, whereas 34 tumors (50%) were positive with the HercepTest kit. Roche and Ingle evaluated tissue samples from 80 patients with resected node-positive breast cancer using the HercepTest kit and the monoclonal antibody CB11 (21). The incidence of HER-2/neu overexpression using CB11 was 26% (21 patients). However, 32 (54%) of the remaining 59 patients considered negative for overexpression by CB11 (0 or 1+) were found to be positive (2 or 3+) using the HercepTest kit (21). Statistically, in our series, there was also poor agreement among the 2 antibodies used. We documented six cases that were positive using CB11 but were completely negative (no staining, 0) for the Oncor polyclonal.
In conclusion, immunohistochemical expression of the HER-2/neu oncogene, defined as complete membranous positivity, is not present in our series of osteosarcomas. Cytoplasmic positivity is observed in most osteosarcomas, irrespective of histologic subtype/grade, and does not appear associated with response to preoperative chemotherapy or disease progression. Different antibodies as well as variable scoring systems may contribute to discrepant results among various studies. Additionally, our study was limited by a relatively small sample size. Before the acceptance of patients into trials for the humanized monoclonal antibody Herceptin (Genetech, San Francisco, CA) based solely on positive immunohistochemical results, a consensus should be reached regarding the most reliable and reproducible methods for determining HER-2/neu status. To date, no evidence of gene amplification or mutation of the HER-2/neu oncogene has been documented in osteosarcoma (2, 19).
References
Gorlick R, Huvos AG, Aledo A, Beardsley GP, Healey JH, Meyers PA . Expression of HER2/erbB-2 correlates with survival in osteosarcoma. J Clin Oncol 1999; 17: 2781–2788.
Onda M, Matsuda S, Higaki S, Iijima T, Fukushima J, Yokokura A, Kojima T, Horiuchi H, Kurokawa T, Yamamoto T . ErbB-2 expression is correlated with poor prognosis for patients with osteosarcoma. Cancer 1996; 77: 71–78.
Thor AD, Schwartz LH, Koerner FC, Edgerton SM, Skates SJ, Yin S, McKenzie SJ, Panicali DL, Marks PJ, Fingert HJ . Analysis of c-erbB-2 expression in breast cancer with clinical follow-up. Cancer Res 1989; 49: 2087–2090.
Tetu B, Brisson J . Prognostic significance of HER-2/neu oncoprotein expression in node-positive breast cancer: the influence of the pattern of immunostaining and adjuvant therapy. Cancer 1994; 73: 2359–2365.
Noguchi M, Koyasaki N, Ohta N, Kitagawa H, Earashi M, Thomas M, Miyazaki I, Mizukami Y . C-erbB-2 oncoprotein expression versus internal mammary lymph node metastases as additional prognostic factors in patients with axillary lymph node-positive breast cancer. Cancer 1992; 69: 2953–2960.
Toikkanen S, Helin H, Isola J, Joensuu H . Prognostic significance of HER-2 oncoprotein expression in breast cancer: a 30-year follow-up. J Clin Oncol 1992; 10: 1044–1048.
Cobleigh MA, Vogel CL, Tripathy D, Robert NJ, Scholl S, Fehrenbacher L, Wolter JM, Paton V, Shak S, Lieberman G, Slamon DJ . Multinational study of the efficacy and safety of humanized anti-HER2 monoclonal antibody in women who have HER2-overexpressing metastatic breast cancer that has progressed after chemotherapy for metastatic disease. J Clin Oncol 1999; 17: 2639–2648.
Pegram MD, Lipton A, Hayes DF, Weber BL, Baselga JM, Tripathy D, Baly D, Baughman SA, Twaddell T, Glaspy JA, Slamon DJ . Phase II study of receptor-enhanced chemosensitivity using recombinant humanized anti-p185HER2/neu monoclonal antibody plus cisplatinum in patients with HER2/neu-overexpressing metastatic breast cancer refractory to chemotherapy treatment. J Clin Oncol 1998; 16: 2659–2671.
Clark GM, McGuire WL . Follow-up study of HER-2/neu amplification in primary breast cancer. Cancer Res 1991; 51: 944–948.
Rosen PP, Lesser ML, Arroyo CD, Cranor M, Borgen P, Norton L . Immunohistochemical detection of HER-2/neu in patients with node-negative breast carcinoma. Cancer 1995; 75: 1320–1326.
Press MF, Hung G, Godolphin GW, Slamon DJ . Sensitivity of HER-2/neu antibodies in archival tissue samples: potential source of error in immunohistochemical studies of oncogene expression. Cancer Res 1994; 54: 2771–2777.
Hanna W, Kahn HJ, Trudeau M . Evaluation of HER-2/neu (erbB-2) status in breast cancer: from bench to bedside. Mod Pathol 1999; 12: 827–834.
Coombs LM, Pigott DA, Sweeney E, Proctor AJ, Eydmann ME, Parkinson C, Knowles MA . Amplification and overexpression of c-erbB-2 in transitional cell carcinoma of the urinary bladder. Br J Cancer 1991; 63: 601–608.
Kay WE, Mulchay H, Walsh CB, Leader M, O'Donoghue D . Cytoplasmic c-erbB-2 protein expression correlates with survival in Duke's B colorectal carcinoma. Histopathology 1994; 25: 455–461.
Roychowdhury DF, Tseng A Jr, Fu KK, Weinberg V, Weidner N . New prognostic factors in nasopharyngeal carcinoma: tumor angiogenesis and c-erbB-2 expression. Cancer 1996; 77: 1419–1426.
Akslen LA, Varhaug JE . Oncoprotein and tumor progression in papillary thyroid carcinoma: presence of epidermal growth factor receptor, c-erbB-2 protein, estrogen receptor-related protein, p21 ras protein, and proliferation indictors in relation to tumor recurrences and patient survival. Cancer 1995; 76: 1643–1654.
Ridolfi RL, Jamehdor MR, Arber JM . HER-2/neu testing in breast carcinoma: a combined immunohistochemical and fluorescence in situ hybridization approach. Mod Pathol 2000; 13: 866–873.
Zhou H, Randall LR, Goldsby R, Smith L, Coffin CM . HER-2/neu staining in osteosarcoma: association with increased risk of metastasis [abstract]. Mod Pathol 2001; 14: 19A.
Maitra A, Wanzer DM, Saboorian H, Weinberg AG, Ashfaq R . Amplification of the Her-2/Neu ocogene is uncommon in pediatric osteosarcomas [abstract]. Mod Pathol 2001; 14: 4P.
Maia DM . Immunohistochemical assays for HER2 overexpression [letter]. J Clin Oncol 1999; 17: 1650.
Roche PC, Ingle JN . Increased incidence of HER2 overexpression with FDA-approved antibody in breast caner patients [letter]. J Clin Oncol 1999; 17: 434.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was presented in part at the Annual Meeting of the Society for Pediatric Pathology, Atlanta, Georgia, March 3–4, 2001.
Rights and permissions
About this article
Cite this article
Kilpatrick, S., Geisinger, K., King, T. et al. Clinicopathologic Analysis of HER-2/neu Immunoexpression among Various Histologic Subtypes and Grades of Osteosarcoma. Mod Pathol 14, 1277–1283 (2001). https://doi.org/10.1038/modpathol.3880474
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3880474
Keywords
This article is cited by
-
Role of TKI for Metastatic Osteogenic Sarcoma
Current Treatment Options in Oncology (2020)
-
CDC20 and its downstream genes: potential prognosis factors of osteosarcoma
International Journal of Clinical Oncology (2019)
-
Bone Sarcomas in Pediatrics: Progress in Our Understanding of Tumor Biology and Implications for Therapy
Pediatric Drugs (2015)
-
Comments on Liu Y et al. “Effect of c-erbB2 overexpression on prognosis in osteosarcoma: evidence from eight studies”
Tumor Biology (2015)
-
Translational biology of osteosarcoma
Nature Reviews Cancer (2014)