Abstract
Fibrolamellar carcinoma arises in noncirrhotic livers of young individuals and has been considered to be less aggressive than conventional hepatocellular carcinoma. This study compares survival and clinicopathologic features of fibrolamellar carcinoma with hepatocellular carcinoma arising in noncirrhotic and cirrhotic livers. Clinical and pathologic features including age, gender, tumor size, stage and survival were recorded in 20 resected cases of fibrolamellar carcinoma. Survival was compared with resected hepatocellular carcinoma without (n=32) and with cirrhosis (n=30). Proliferative activity was determined by immunohistochemistry for Ki-67. In all, 12 (60%) patients with fibrolamellar carcinoma died during follow-up; the 5-year survival was 45%. Mortality in fibrolamellar carcinoma was higher with metastatic disease at presentation (6/7, 86% vs 5/13, 39%, P=0.06). Age, gender and tumor size did not correlate with survival. The 5-year (45 vs 56%, P=0.4) as well as overall survival (40 vs 56.3%, P=0.3) was similar in fibrolamellar carcinoma and hepatocellular carcinoma without cirrhosis. The 5-year and overall survival in hepatocellular carcinoma with cirrhosis was 27 and 23.3%, respectively, which was not significantly different compared to fibrolamellar carcinoma (P=0.2). Among the cases without metastases at presentation, 5-year survival in fibrolamellar carcinoma (62%) and hepatocellular carcinoma without cirrhosis (57%) was significantly better (P=0.03) than hepatocellular carcinoma with cirrhosis (27%). The mean Ki-67 index was similar in all three groups (P=0.1). In conclusion, fibrolamellar carcinoma is an aggressive neoplasm with 45% 5-year survival and overall mortality of 60%. Nearly half the patients develop lymph node or distant metastasis. The prognosis of fibrolamellar carcinoma is similar to conventional hepatocellular carcinoma. Among nonmetastatic cases, the prognosis is better in fibrolamellar carcinoma and hepatocellular carcinoma without cirrhosis compared to hepatocellular carcinoma with cirrhosis. The better outcome in fibrolamellar carcinoma appears to be due to the absence of cirrhosis rather than its distinct clinicopathologic features.
Similar content being viewed by others
Main
Fibrolamellar hepatocellular carcinoma is a variant of hepatocellular carcinoma with distinct clinicopathologic features. It occurs at a young age1, 2, 3 and lacks common risk factors for hepatocellular carcinoma like viral hepatitis and cirrhosis.4, 5, 6 Elevated alpha fetoprotein (AFP) levels are infrequent.2, 6 The defining feature of fibrolamellar carcinoma is its triad of histologic characteristics, viz. tumor cells with deeply eosinophilic cytoplasm, presence of macronucleoli and abundant fibrous stroma arranged in thin parallel lamellae around the tumor cells.1, 2, 4, 5, 7
Many studies have advocated that fibrolamellar carcinoma is less aggressive than conventional hepatocellular carcinoma.1, 3, 4, 5, 8, 9, 10, 11, 12, 13, 14 Several pathology and hepatology texts mention that fibrolamellar carcinoma is associated with a favorable prognosis.15, 16, 17, 18 However, other studies have failed to confirm the observation of a better outcome in fibrolamellar carcinoma.2, 6, 19, 20, 21 The World Health Organization blue book lists fibrolamellar carcinoma as a distinct variant of hepatocellular carcinoma, but refrains from any comment about its outcome compared to conventional hepatocellular carcinoma.22 In our experience, fibrolamellar carcinoma is an aggressive tumor and we have observed recurrence and metastasis in many cases of fibrolamellar carcinoma after initial resection.
Cirrhosis is a well-established poor prognostic factor in hepatocellular carcinoma.23, 24 Since fibrolamellar carcinoma almost always arises in noncirrhotic liver, the apparent better outcome in fibrolamellar carcinoma may be related to lack of cirrhosis. Approximately 20% of conventional hepatocellular carcinoma arise in the absence of cirrhosis.25 The survival in fibrolamellar carcinoma cases should be compared to conventional hepatocellular carcinoma arising in noncirrhotic liver to avoid the confounding factor of cirrhosis. There have been few studies that have simultaneously compared fibrolamellar carcinoma with hepatocellular carcinoma with and without cirrhosis.6, 12, 19 The goal of this study was to compare the survival and clinicopathologic features of fibrolamellar carcinoma with hepatocellular carcinoma arising in noncirrhotic and cirrhotic livers.
Materials and methods
The study population comprised of 20 patients with resected fibrolamellar carcinoma and 62 patients with conventional hepatocellular carcinoma (30 arising in cirrhotic liver and 32 in noncirrhotic liver). The cases were retrieved from UCSF Medical Center, Mayo Clinic and Yale New Haven Hospital by searching for ‘fibrolamellar carcinoma’ in the diagnosis line of liver resection specimens from 1987 to 2000. The slides were reviewed to confirm the diagnosis. Four cases originally diagnosed as fibrolamellar carcinoma lacked the typical histological features and were reclassified as scirrhous hepatocellular carcinoma. These cases are not included in the study group of 20 cases. All tumors classified as fibrolamellar carcinoma showed abundant granular cytoplasm, enlarged nucleus with a prominent nucleolus and lamellar plate-like fibrosis (Figure 1). Clinical and pathologic features including age, gender, tumor size, stage and survival data were recorded. Among the hepatocellular carcinoma cases arising in cirrhotic liver, the etiology of cirrhosis was hepatitis B (n=7), hepatitis C (n=11), hereditary hemochromatosis (n=1) and cryptogenic (n=11). Additional histologic features were recorded for fibrolamellar carcinoma cases including presence of bile, cytoplasmic hyaline globules and pale bodies. Rhodanine stain for copper was performed in 12 cases of fibrolamellar carcinoma. Positive results were scored as mild, moderate or marked. The clinicopathologic features and survival in fibrolamellar carcinoma was compared with hepatocellular carcinoma arising in noncirrhotic and cirrhotic liver.
The fibrolamellar carcinoma cases were examined by paraffin immunohistochemistry using rabbit polyclonal antibody against AFP (1:500, Dako, Carpinteria, CA, USA) and mouse monoclonal antibodies against chromogranin (1:75, Boehringer Mannheim, Petersburg, VA, USA) and synaptophysin (1:40, MP Biomedicals, Irvine, CA, USA). Proliferation activity in both fibrolamellar carcinoma and hepatocellular carcinoma was determined by using mouse monoclonal antibody against Ki-67 (1:100, Dako, Carpinteria, CA, USA). Deparaffinized slides were microwaved in 10 mM citrate buffer for 10 min for antigen retrieval. The endogenous peroxidase activity was blocked by 3% H2O2. The sections were incubated overnight with the primary antibody at 4°C Biotinylated horse anti-mouse antibody (Vector Labs, Burlingame, CA, USA) was used as the secondary antibody and diacetyl benzidine was used for color development. The Ki-67 index was determined by counting the number of positive cells in five high-power fields and expressing the result as a percentage. Fisher's exact test and χ2 test were used for statistical analysis. Log-rank test and Kaplan–Meier curves were used for survival analysis.
Results
Fibrolamellar Carcinoma
The mean age based on 19 cases of fibrolamellar carcinoma (age not known in one case) was 27 years (range 16–47 years, 11 male, nine female). In all, 16 (84%) cases were below 40 years of age. Multiple primary tumors were present in two cases. Serum AFP was elevated in 3/13 (23%) patients; levels were not known in the other cases. The levels were <100 ng/ml in two cases and 612 ng/ml in one case. Stainable copper was present in 9/12 (75%) cases examined. The staining was mild in six cases and marked in three cases. AFP was negative by immunohistochemistry in all 14 cases examined. Immunohistochemistry for synaptophysin and chromogranin was negative in all 12 cases tested.
A total of 12 (60%) patients died of the disease; the 5-year survival was 45%. Seven of the 20 (35%) patients had lymph node metastasis at presentation. The 5-year survival was higher in patients with localized disease at initial presentation (8/13, 62% vs 1/7, 14%, P=0.06). Age, gender and tumor size did not correlate with survival (Table 1).
Comparison with Conventional Hepatocellular Carcinoma
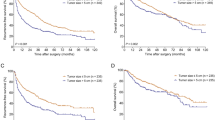
Fibrolamellar carcinoma occurred at a younger age and was associated with a larger tumor size compared to HCC (Table 2). Cytoplasmic pale bodies were identified in eight (40%) fibrolamellar carcinoma cases, but were not seen in hepatocellular carcinoma (Figure 2). Survival analysis among all cases irrespective of stage showed that the 5-year (45 vs 56%, P=0.4) as well as overall survival (40 vs 56%, P=0.3) was similar in fibrolamellar carcinoma and hepatocellular carcinoma without cirrhosis. The 5-year and overall survival in hepatocellular carcinoma with cirrhosis was 27 and 23%, respectively, which was worse compared to fibrolamellar carcinoma (Figure 3), but the difference was not statistically significant (P=0.2).
When survival analysis was performed in cases with same-stage disease by examining tumors localized to the liver at presentation (Table 1, Figure 4), the 5-year survival in fibrolamellar carcinoma (62%) and hepatocellular carcinoma without cirrhosis (58%) was similar, but survival in both groups was significantly better than hepatocellular carcinoma with cirrhosis (27%).
The mean Ki-67 index was 5.0 (range 0.2–17) in fibrolamellar carcinoma, 7.8 (range 0.1–27) in hepatocellular carcinoma without cirrhosis and 9.5 (range 0.5–26) in hepatocellular carcinoma with cirrhosis (P=0.1). The Ki-67 index did not correlate with survival.
Discussion
Fibrolamellar carcinoma is an uncommon variant of hepatocellular carcinoma with distinctive morphology and clinical setting. It has been widely regarded that fibrolamellar carcinoma has relatively favorable natural history and is associated with better outcome compared to conventional hepatocellular carcinoma.
This study confirms the distinctive clinicopathologic features of fibrolamellar carcinoma as reported in literature. The tumors in fibrolamellar carcinoma cases were significantly larger than conventional hepatocellular carcinoma; similar findings have been reported in literature.5, 9, 11 Majority of the tumors occurred in patients less than 40 years old, with only 15% above the age of 40 years. A review of some large series reveals that 80% of fibrolamellar carcinoma patients present before 35 years and only 11% after the age of 40 years. It is important to strictly use established criteria for the diagnosis of fibrolamellar carcinoma, especially in older individuals. In ambiguous cases, the presence of ‘pale bodies’ would strongly favor fibrolamellar carcinoma, although they have been described in conventional hepatocellular carcinoma, especially the scirrhous variant.22 Stainable copper was found in 75% of fibrolamellar carcinoma in our series. The presence of copper and copper-binding protein was cited as a specific feature of fibrolamellar carcinoma,26, 27 but was later described in conventional HCC as well.28 Serum AFP was elevated in 3/18 (16.7%) cases, although levels were less than 70 ng/ml except in one case. A prior review noted elevated AFP in 7% of fibrolamellar carcinoma compared to 83% of conventional hepatocellular carcinoma.2 Synaptophysin and chromogranin were negative in all our cases. Nonspecific enolase and neurotensin have been reported to be positive in fibrolamellar carcinoma.29, 30 Structures resembling neurosecretory granules have been noticed in some29 but not all ultrastructural studies.1, 31 The significance of this ‘neuroendocrine differentiation’ remains unclear, but is unlikely to be helpful in distinction from conventional HCC as up to one-fourth of the latter may also show ‘neuroendocrine differentiation’.30, 32, 33 Immunohistochemistry for AFP was negative in all cases tested, similar to other studies,8 although focal positive reaction in small groups of cells has been reported.31
Several studies have emphasized the better survival in fibrolamellar carcinoma compared to conventional hepatocellular carcinoma.1, 3, 4, 5, 8, 9, 10, 11, 12, 13, 14 However, these results could not be confirmed by many authors.2, 6, 19, 20 The present study provides further evidence that fibrolamellar carcinoma is an aggressive neoplasm associated with 5-year survival of less than 50%. Although the majority of cases occurred in patients less than 40 years old, nearly 40% of patients died even when fibrolamellar carcinoma was localized to the liver at presentation. The outcome was particularly dismal (5-year survival ∼15%) in patients with metastatic disease at presentation. The 5-year survival in resected fibrolamellar carcinoma in various reports ranges from <25 to 75%1, 4, 5, 6, 8, 9, 11, 12, 14, 18, 19, 21 The aggressive nature of fibrolamellar carcinoma is apparent from the fact that in several studies in which fibrolamellar carcinoma had a better outcome compared to conventional hepatocellular carcinoma, the 5-year survival was only 20–40%.1, 8, 12 Ploidy studies have shown that all fibrolamellar carcinoma cases are aneuploid or polyploid, which is in keeping with an aggressive tumor.34 Similarly, chromosomal genomic hybridization has shown chromosomal gains and losses that are similar to conventional hepatocellular carcinoma.35
Several reasons could explain the discrepant results reported in literature. Fibrolamellar carcinoma is a rare tumor and majority of the reported series are small (Table 3). This may lead to lack of sufficient power for statistical analysis. Another reason may be the criteria used for the diagnosis of fibrolamellar carcinoma. It has been advocated that a triad of histological features be used for diagnosis—abundant eosinophilic cytoplasm, plate-like lamellar fibrosis and large nuclei with marginalized chromatin and prominent nucleolus.7 Reliance on two features may lead to erroneous diagnosis of some hepatocellular carcinoma variants (like scirrhous variant and hepatocellular carcinoma with oncocytic cytoplasm) as fibrolamellar carcinoma. Many studies have not used this diagnostic triad to identify cases of fibrolamellar carcinoma; in some series, the diagnostic criteria are not clearly outlined12 or the original histological diagnosis was not reviewed before being included in the study.3 Some studies have included cases that occurred in cirrhotic liver and are unlikely to be fibrolamellar carcinoma.1 In some series, tumors with fibrolamellar carcinoma-like areas were included even though other areas showed features typical of conventional hepatocellular carcinoma.5 In our experience, the clinicopathologic features of these tumors like advanced age and background of cirrhosis resemble conventional hepatocellular carcinoma rather than fibrolamellar carcinoma (unpublished observations). These ‘mixed’ tumors have also been reported in association with hepatitis C and cirrhosis, which supports the view that they should be classified as conventional hepatocellular carcinoma.36 In some studies, lack of adequate follow-up may contribute to the conclusion of better prognosis in fibrolamellar carcinoma. In a series reported by Starzl et al,10 12/14 fibrolamellar carcinoma patients were alive after transplantation or resection. However, 5-year follow-up was available in only two of these 12 patients.
Perhaps the most important reason for the varying results in literature is that fibrolamellar carcinoma has been compared to hepatocellular carcinoma as a whole. Hepatocellular carcinoma is not a prognostically homogeneous entity but can be divided into several subgroups, which may have distinct biological behavior. Examples of these subgroups include hepatocellular carcinoma arising in noncirrhotic liver, small encapsulated hepatocellular carcinoma and well-differentiated hepatocellular carcinoma. However, the overwhelming majority of hepatocellular carcinoma (>80%) arise in cirrhotic liver.25 Cirrhosis is a well-known adverse prognostic feature in hepatocellular carcinoma.23, 24 Hence, when survival in fibrolamellar carcinoma is compared to hepatocellular carcinoma as a whole, the poor outcome in the latter group as a result of cirrhosis is likely to skew the results. In a vast majority of studies, fibrolamellar carcinoma has been compared to hepatocellular carcinoma as a whole (Table 3). The largest and most recent population-based study of 68 cases of fibrolamellar carcinoma using information collected from the Surveillance, Epidemiology and End Results (SEER) program showed superior survival in fibrolamellar carcinoma, but compared it to hepatocellular carcinoma as a group without separately analyzing hepatocellular carcinoma with and without cirrhosis.3 The study also relies on the original pathology diagnosis and cases were not reviewed to confirm the diagnosis of fibrolamellar carcinoma before inclusion in the study.
There are few studies in literature that have compared fibrolamellar carcinoma to hepatocellular carcinoma by separating the latter cases into those arising in noncirrhotic liver and cirrhotic liver. Nagorney et al19 reported no significant difference in survival in 12 cases of resected fibrolamellar carcinoma compared to hepatocellular carcinoma in noncirrhotic liver. Ruffin in a series of five fibrolamellar carcinoma case reports suggested that the differences between fibrolamellar carcinoma and comparable subgroups of hepatocellular carcinoma are less distinct.2 In a study of 20 cases of fibrolamellar carcinoma, Ringe et al6 reported a 5-year survival of 37% and did not find a better prognosis compared to conventional hepatocellular carcinoma. Our study supports these findings and fails to confirm the assertion that fibrolamellar carcinoma has a favorable outcome. We found that survival was similar in resected fibrolamellar carcinoma and hepatocellular carcinoma in noncirrhotic liver among the cases confined to the liver at presentation. The survival in both these groups was significantly better than hepatocellular carcinoma arising in cirrhotic liver. It has been argued that better survival in fibrolamellar carcinoma may be related to higher resectability rate.3, 19 Since our analysis includes only resected cases of fibrolamellar carcinoma and hepatocellular carcinoma, this issue cannot be addressed by our study.
In conclusion, our results indicate that fibrolamellar carcinoma is an aggressive tumor associated with less than 50% 5-year survival even in resectable cases. When same-stage disease is considered, the survival in fibrolamellar carcinoma is similar to hepatocellular carcinoma arising in noncirrhotic liver, and better than hepatocellular carcinoma arising in cirrhotic liver. The better survival in fibrolamellar carcinoma is likely to be related to the absence of cirrhosis rather than the unique clinicopathologic features of the tumor.
References
Craig JR, Peters RL, Edmondson HA, et al. Fibrolamellar carcinoma of the liver: a tumor of adolescents and young adults with distinctive clinico-pathologic features. Cancer 1980;46:372–379.
Ruffin IV MT . Fibrolamellar hepatoma. Am J Gastroenterol 1990;85:577–581.
El-Serag HB, Davila JA . Is fibrolamellar carcinoma different from hepatocellular carcinoma? A US population-based study. Hepatology 2004;39:798–803.
Hodgson HJ . Fibrolamellar cancer of the liver. J Hepatol 1987;5:241–247.
Berman MA, Burnham JA, Sheahan DG . Fibrolamellar carcinoma of the liver: an immunohistochemical study of nineteen cases and a review of the literature. Hum Pathol 1988;19:784–794.
Ringe B, Wittekind C, Weimann A, et al. Results of hepatic resection and transplantation for fibrolamellar carcinoma. Surg Gynecol Obstet 1992;175:299–305.
Burgart LJ, Martinez CJM, Batts KP . Fibrolamellar hepatoma—importance of using a strict definition (abstract). Mod Pathol 1994;7:129A.
Lack EE, Neave C, Vawter GF . Hepatocellular carcinoma. Review of 32 cases in childhood and adolescence. Cancer 1983;52:1510–1515.
Farhi DC, Shikes RH, Murari PJ, et al. Hepatocellular carcinoma in young people. Cancer 1983;52:1516–1525.
Starzl TE, Iwatsuki S, Shaw Jr BW, et al. Treatment of fibrolamellar hepatoma with partial or total hepatectomy and transplantation of the liver. Surg Gynecol Obstet 1986;162:145–148.
Soreide O, Czerniak A, Bradpiece H, et al. Characteristics of fibrolamellar hepatocellular carcinoma. A study of nine cases and a review of the literature. Am J Surg 1986;151:518–523.
Wood WJ, Rawlings M, Evans H, et al. Hepatocellular carcinoma: importance of histologic classification as a prognostic factor. Am J Surg 1988;155:663–666.
Okuda K . Natural history of hepatocellular carcinoma including fibrolamellar and hepato-cholangiocarcinoma variants. J Gastroenterol Hepatol 2002;17:401–405.
Pinna AD, Iwatsuki S, Lee RG, et al. Treatment of fibrolamellar hepatoma with subtotal hepatectomy or transplantation. Hepatology 1997;26:877–883.
Everson GT, Trotter JF . Transplantation of the liver. In: Schiff ER, Sorrell MF, Maddrey WC (eds). Diseases of the Liver, 9th edn., Vol. 2. Lippincott, Williams and Wilkins: Philadelphia, 2003, pp 1585–1614.
Sherlock S, Dooley J (eds). Malignant liver tumors. In: Diseases of the Liver and Biliary System, 11th edn. Blackwell Science: Malden, MA, 2003, pp 537–562.
Rosai J . Rosai and Ackerman's Surgical Pathology. Mosby: Philadelphia, 9th edn., Vol. 1. 2004, pp 1001–1002.
Ferrell LD . Benign and malignant tumors of the liver. In: Odze RD, Goldblum JR, Crawford JM (eds). Surgical Pathology of the GI Tract, Liver, Biliary Tract and Pancreas. Saunders: Philadelphia, 2004, pp 1012.
Nagorney DM, Adson MA, Weiland LH, et al. Fibrolamellar hepatoma. Am J Surg 1985;149:113–119.
Haas JE, Muczynski KA, Krailo M, et al. Histopathology and prognosis in childhood hepatoblastoma and hepatocarcinoma. Cancer 1989;64:1082–1095.
Katzenstein HM, Krailo MD, Malogolowkin MH, et al. Fibrolamellar hepatocellular carcinoma in children and adolescents. Cancer 2003;97:2006–2012.
Hamilton SR, Aaltonen LA (eds). Tumors of the liver and intrahepatic bile ducts. In: Pathology and Genetics of the Digestive System. IARC Press: Lyon, 2000, pp 159–172.
Chedid A, Ryan LM, Dayal Y, et al. Morphology and other prognostic factors of hepatocellular carcinoma. Arch Pathol Lab Med 1999;123:524–528.
Quaglia A, Bhattacharjya S, Dhillon AP . Limitations of the histopathological diagnosis and prognostic assessment of hepatocellular carcinoma. Histopathology 2001;38:167–174.
Monto A, Wright TL . The epidemiology and prevention of hepatocellular carcinoma. Semin Oncol 2001;28:441–449.
Vecchio FM, Federico F, Dina MA . Copper and hepatocellular carcinoma. Digestion 1986;35:109–114.
Lefkowitch JH, Muschel R, Price JB, et al. Copper and copper-binding protein in fibrolamellar liver cell carcinoma. Cancer 1983;51:97–100.
Guigui B, Mavier P, Lescs MC, et al. Copper and copper-binding protein in liver tumors. Cancer 1988;61:1155–1158.
Garcia de Davila MT, Gonzalez-Crussi F, Mangkornkanok M . Fibrolamellar carcinoma of the liver in a child: ultrastructural and immunohistologic aspects. Pediatr Pathol 1987;7:319–331.
Wang JH, Dhillon AP, Sankey EA, et al. ‘Neuroendocrine’ differentiation in primary neoplasms of the liver. J Pathol 1991;163:61–67.
Caballero T, Aneiros J, Lopez-Caballero J, et al. Fibrolamellar hepatocellular carcinoma. An immunohistochemical and ultrastructural study. Histopathology 1985;9:445–456.
Zhao M, Laissue JA, Zimmermann A . ‘Neuroendocrine’ differentiation in hepatocellular carcinomas (HCCs): immunohistochemical reactivity is related to distinct tumor cell types, but not to tumor grade. Histol Histopathol 1993;8:617–626.
Huang XF, Wang CM, Dai XW, et al. Expressions of chromogranin A and cathepsin D in human primary hepatocellular carcinoma. World J Gastroenterol 2000;6:693–698.
Orsatti G, Greenberg PD, Rolfes DB, et al. DNA ploidy of fibrolamellar hepatocellular carcinoma by image analysis. Hum Pathol 1994;25:936–939.
Wilkens L, Bredt M, Flemming P, et al. Cytogenetic aberrations in primary and recurrent fibrolamellar hepatocellular carcinoma detected by comparative genomic hybridization. Am J Clin Pathol 2000;114:867–874.
Okada K, Kim YI, Nakashima K, et al. Fibrolamellar hepatocellular carcinoma coexistent with a hepatocellular carcinoma of common type: report of a case. Surg Today 1993;23:626–631.
Acknowledgements
We thank the UCSF Liver Center (P30 DK26743) for providing assistance with sample processing for immunohistochemistry.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kakar, S., Burgart, L., Batts, K. et al. Clinicopathologic features and survival in fibrolamellar carcinoma: comparison with conventional hepatocellular carcinoma with and without cirrhosis. Mod Pathol 18, 1417–1423 (2005). https://doi.org/10.1038/modpathol.3800449
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800449
Keywords
This article is cited by
-
Fibrolamellar Hepatocellular Carcinoma: A Population-Based Observational Study
Digestive Diseases and Sciences (2021)
-
Clinicopathological Features of Two Extremely Rare Hepatocellular Carcinoma Variants: a Brief Review of Fibrolamellar and Scirrhous Hepatocellular Carcinoma
Journal of Gastrointestinal Cancer (2020)
-
Cytoreductive Surgery and HIPEC for Recurrent Fibrolamellar Hepatocellular Carcinoma with Peritoneal Carcinomatosis
Journal of Gastrointestinal Cancer (2020)
-
HCCs and HCAs in Non-cirrhotic Patients: What You See May Not Be Enough
Digestive Diseases and Sciences (2019)
-
Analyzing cytogenetic chromosomal aberrations on fibrolamellar hepatocellular carcinoma detected by single-nucleotide polymorphs array
Neural Computing and Applications (2019)