Abstract
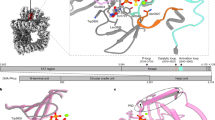
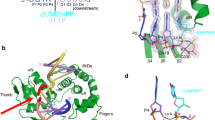
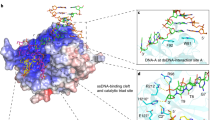
THE success of cisplatin in cancer chemotherapy derives from its ability to crosslink DNA and alter the structure. Most cisplatiná¤-DNA adducts are intrastrand d(GpG) and d(ApG) crosslinks1, which unwind and bend the duplex to facilitate the binding of proteins that contain one or more high-mobility-group (HMG) domains2. When HMG-domain proteins such as HMG1, IXR (intrastrand-crosslink recognition) protein from yeast, or human upstream-binding factor (hUBF) bind cisplatin intrastrand crosslinks, they can be diverted from their natural binding sites on the genome and shield the adducts from excision repair3-5. These activities sensitize cells to cisplatin and contribute to its cytotoxic properties. Crystallographic information about the structure of cisplatiná¤-DNA adducts has been limited to short single-stranded deoxyoligonucleotides such ascis[Pt(NH3)2{d(pGpG)}]6-8. Here we describe the X-ray structure at 2.6 Å resolution of a double-stranded DNA dodecamer containing this adduct. Our information provides, to our knowledge, the first crystallographic look at a platinated DNA duplex and should help the design of new platinum and other metal crosslinking antitumour drug candidates. Moreover, the structure reveals a unique fusion of A- and B-type DNA segments that could be of more general importance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fichtinger-Schepman, A. M. J., van der Veer, J. L., den Hartog, J. H. J., Lohman, P. H. M. & Reedijk, J. Biochemistry 24, 707–713 (1985).
Whitehead, J. P. & Lippard, S. J. in Metal Ions in Biological Systems (eds Sigel, H. & Sigel, A.) (Marcel Dekker, New York, in the press).
Brown, S. J., Kellett, P. J. & Lippard, S. J. Science 261, 603–605 (1993).
Treiber, D. K., Zhai, X., Jantzen, H.-M. & Essigmann, J. M. Proc. natn. Acad. Sci. U.S.A. 91, 5672–5676 (1994).
Huang, J. C., Zamble, D. B., Reardon, J. T., Lippard, S. J. & Sancar, A. Proc. natn. Acad. Sci. U.S.A. 91, 10394–10398 (1994).
Sherman, S. E., Gibson, D., Wang, A. H.-J. & Lippard, S. J. Science 230, 412–417 (1985).
Admiraal, G., van der Veer, J. L., de Graaff, R. A. G., den Hartog, J. H. J. & Reedijk, J. J. Am. chem. Soc. 109, 592–594 (1987).
Sherman, S. E., Gibson, D., Wang, A. H. J. & Lippard, S. J. J. Am. chem. Soc. 110, 7368–7381 (1988).
Rice, J. A., Crothers, D. M., Pinto, A. L. & Lippard, S. J. Proc. natn. Acad. Sci. U.S.A. 85, 4158–4161 (1988).
Bellon, S. F. & Lippard, S. J. Biophys. Chem. 35, 179–188 (1990).
Bellon, S. F., Coleman, J. H. & Lippard, S. J. Biochemistry 30, 8026–8035 (1991).
Wang, A. H.-J. & Teng, M.-K. J. Cryst. Growth 90, 295–310 (1988).
Wang, A. H.-J., Fujii, S., van Boom, J. H. & Rich, A. Proc. natn. Acad. Sci. U.S.A. 79, 3968–3972 (1982).
den Hartog, J. H. J. et al. J. biomolec. struct. Dyn. 2, 1137–1155 (1985).
Sherman, S. E. & Lippard, S. J. Chem. Rev. 87, 1153–1181 (1987).
Herman, F. et al. Eur. J. Biochem. 194, 119–133 (1990).
Saenger, W. in Principles of Nucleic Acid Structure 226 (Springer, New York, 1984).
Frederick, C. A. et al. Eur. J. Biochem. 181, 295–307 (1989).
Chow, C. S., Whitehead, J. P. & Lippard, S. J. Biochemistry 33, 15124–15130 (1994).
King, C.-Y. & Weiss, M. A. Proc. natn. Acad. Sci. U.S.A. 90, 11990–11994 (1993).
Werner, M. H., Huth, J. R., Gronenborn, A. M. & Clore, G. M. Cell 81, 705–714 (1995).
Bruhn, S. L., Toney, J. H. & Lippard, S. J. Prog. Inorg. Chem. 38, 477–516 (1990).
Collaborative Computational Project, Number 4 Acta crystallogr. D50, 760–763 (1994).
Jones, T. A., Zou, J.-Y., Cowan, S. W. & Kjeldgaard, M. Acta. crystallogr. A47, 110–119 (1991).
Brünger, A. T. X-PLOR Version 3.1. A System for X-ray Crystallography and NMR (Yale University Press, New Haven, 1992).
Brünger, A. T. Nature 355, 472–475 (1992).
Lavery, R. & Sklenar, H. J. biomolec. struct. Dyn. 6, 63–91 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takahara, P., Rosenzweig, A., Frederick, C. et al. Crystal structure of double-stranded DNA containing the major adduct of the anticancer drug cisplatin. Nature 377, 649–652 (1995). https://doi.org/10.1038/377649a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/377649a0
This article is cited by
-
Rational design of platinum(II) complexes with orthogonally oriented triazolyl ligand with emission enhancement characteristics for cancer chemotherapy in vivo
Science China Chemistry (2023)
-
Phenols, antioxidant and anticancer properties of Tagetes minuta, Euphorbia granulata and Galinsoga parviflora: in vitro and in silico evaluation
Journal of Umm Al-Qura University for Applied Sciences (2023)
-
Genomic aberrations after short-term exposure to colibactin-producing E. coli transform primary colon epithelial cells
Nature Communications (2021)
-
Blood-triggered generation of platinum nanoparticle functions as an anti-cancer agent
Nature Communications (2020)
-
Pharmacological Properties of Pt(II) and Pt(IV) Complexes with 2,2′-Dipyridylamine; the Comparative In Vitro Thereof
Cell Biochemistry and Biophysics (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.