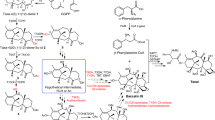
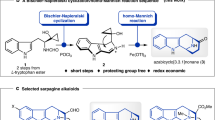
Abstract
TAXOL1–4, a substance originally isolated from the Pacific yew tree (Taxus brevifolia) more than two decades ago, has recently been approved for the clinical treatment of cancer patients. Hailed as having provided one of the most significant advances in cancer therapy5, this molecule exerts its anticancer activity by inhibiting mitosis through enhancement of the polymerization of tubulin and consequent stabilization of microtubules6. The scarcity of taxol and the ecological impact of harvesting it have prompted extensive searches for alternative sources including semisynthesis, cellular culture production and chemical synthesis2,3. The latter has been attempted for almost two decades, but these attempts have been thwarted by the magnitude of the synthetic challenge. Here we report the total synthesis of taxol by a convergent strategy, which opens a chemical pathway for the production of both the natural product itself and a variety of designed taxoids.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wani, M. C., Taylor, H. L., Wall, M. E., Coggon, P. & McPhail, A. T. J. Am. chem. Soc. 93, 2325–2327 (1971).
Nicolaou, K. C., Dai, W.-M. & Guy, R. K Angew. Chem. int. Edn engl. 33, 15–44 (1994).
Guenard, D., Gueritte-Voegelein, F. & Poitier, P. Acct Chem. Res. 26, 160–167 (1993).
Rowinksy, E. K., Cazenave, L. A. & Donehower, R. C. J. natn. Cancer Inst. 82, 1247–1259 (1990).
Paclitaxel (Taxol) Investigations Workshop Semin. Oncol. 20 (4, Suppl. 3), 1–60 (1993).
Schiff, P. B., Fant, J. & Horwitz, S. B. Nature 277, 665–667 (1979).
Nicolaou, K. C., Hwang, C.-K., Sorensen, E. J. & Claiborne, C. F. J. chem. Soc., chem. Commun. 1117–1118 (1992).
Chamberlin, A. R. & Bloom, S. H. Org. React. 39, 1–83 (1990).
McMurry, J. E. Chem. Rev. 89, 1513–1524 (1989).
Nicolaou, K. C., Nantermet, P. G., Ueno, H. & Guy, R. K. J. chem. Soc., chem. Commun. 295–296 (1994).
Ojima, I. et al. Tetrahedron 48, 6985–7012 (1992).
Nicolaou, K. C., Liu, J. J., Hwang, C.-K., Dai, W.-M. & Guy, R. K. J. chem. Soc., chem. Commun. 1118–1120 (1992).
Nicolaou, K. C., Yang, Z., Sorensen, E. J. & Nakada, M. J. chem. Soc., chem. Commun. 1024–1026 (1993).
Griffith, W. P., Ley, S. V. Aldrichimica Acta. 23, 13–19 (1990).
Sharpless, K. B. & Verhoeven, T. R. Aldrichimica Acta, 12, 63–74 (1979).
Magee, T. V., Bornmann, W. G., Isaacs, R. C. A. & Danishefsky, S. J. J. org. Chem. 57, 3274–3276 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nicolaou, K., Yang, Z., Liu, J. et al. Total synthesis of taxol. Nature 367, 630–634 (1994). https://doi.org/10.1038/367630a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/367630a0
This article is cited by
-
Alternaria alternata F3, a Novel Taxol-Producing Endophytic Fungus Isolated from the Fruits of Taxus cuspidata: Isolation, Characterization, Taxol Yield Improvement, and Antitumor Activity
Applied Biochemistry and Biotechnology (2023)
-
Construction of an Escherichia coli cell factory to synthesize taxadien-5α-ol, the key precursor of anti-cancer drug paclitaxel
Bioresources and Bioprocessing (2022)
-
Endophytic fungus Pseudodidymocyrtis lobariellae KL27 promotes taxol biosynthesis and accumulation in Taxus chinensis
BMC Plant Biology (2022)
-
Biosynthesis of paclitaxel using synthetic biology
Phytochemistry Reviews (2022)
-
Switching cell fate by the actin–auxin oscillator in Taxus: cellular aspects of plant cell fermentation
Plant Cell Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.