Abstract
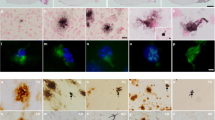
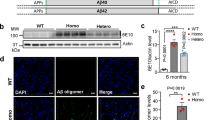
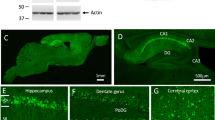
ALZHEIMER'S disease (AD) affects more than 30% of people over 80 years of age1,2. The aetiology and pathogenesis of this progressive dementia is poorly understood, but symptomatic disease is associated histopathologically with amyloid plaques, neurofibrillary tangles and neuronal loss primarily in the temporal lobe and neocortex of the brain. The core of the extracellular plaque is a derivative of the amyloid precursor protein (APP)3, referred to as β/A4 (refs 4–6), and contains the amino-acid residues 29–42 that are normally embedded in the membrane-spanning region of the precursor3. The cellular source of APP and the relationship of its deposition to the neuropathology of AD is unknown. To investigate the relationship between APP over-expression and amyloidogenesis, we have developed a vector to drive expression specifically in neurons of a C-terminal fragment of APP that contains the β/A4 region, and have used a transgenic mouse system7,8 to insert and express this construct. We report here that overexpression of this APP transgene in neurons is sufficient to produce extracellular dense-core amyloid plaques, neurofibrillary tangles and neuronal degeneration similar to that in the AD brain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Change history
19 March 1992
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1038/356265a0
References
Evans, D. A. et al. J. Am. Med. Ass. 262, 2551–2556 (1989).
Katzman, R. & Saitoh, T. FASEB J. 280, 278–286 (1991).
Kang, J. et al. Nature 325, 733–736 (1987).
Glenner, G. G. & Wong, C. W. Biochem. biophys. Res. Commun. 120, 885–890 (1984).
Joachim, C. L., Duffy, L. K., Morris, J. H. & Selkoe, D. J. Brain Res. 474, 100–111 (1988).
Higgins, G. A. Neurobiol. Aging 11, 61–69 (1990).
Gordon, J. W., Scangos, G. A., Plotkin, D. J., Barbosa, J. A. & Ruddle, F. H. Proc. natn. Acad. Sci. U.S.A. 77, 7380–7384 (1980).
Gordon, J. W. & Ruddle, F. H. Science 214, 1244–1246 (1981).
Gordon, J. W. et al. Cell 50, 445–452 (1987).
Kollias, G. et al. Proc. natn. Acad. Sci. U.S.A. 84, 1492–1496 (1987).
Yankner, B. A. et al. Science 245, 417–420 (1989).
Higgins, G. A. et al. Proc. natn. Acad. Sci. U.S.A. 85, 1297–1301 (1988).
Joachim, C. L., Morris, J. H. & Selkoe, D. J. Am. J. Path. 5, 309–319 (1989).
Yamaguchi, H., Nakazato, Y., Hirai, S., Shoji, M. & Harigaya, Y. Am. J. Path. 135, 593–597 (1989).
Giaccone, G. et al. Neurosci. Lett 97, 232–238 (1989).
Mann, D. M. A. Neurobiol. Aging 10, 397–399 (1989).
Motte, J. & Williams, R. S. Acta Neuropath 77, 535–546 (1989).
Rumble, B. et al. New Engl. J. Med. 320, 1446–1452 (1989).
Wisniewski, H. M., Weigel, J., Wang, K. C., Kumawa, M. & Lach, B. Can. J. Neurol. Sci. 16, 535–542 (1989).
Masters, C. L. et al. EMBO J. 4, 2757–2763 (1985).
Siman, R., Card, J. P., Nelson, R. B. & Davis, L. G. Neuron 3, 275–285 (1989).
Quon, D. et al. Nature 352, 239–241 (1991).
Wirak, D. O. et al. Science 253, 323–325 (1991).
Tsurushita, N., Maki, H. & Korn, L. J. Gene 62, 135–139 (1988).
Gunning, P., Leavitt, J., Muscat, G., Ng, S.-Y. & Kedes, L. Proc. natn. Acad. Sci. U.S.A. 84, 4831–4835 (1987).
Gordon, J. W. & Ruddle, F. H. Meth. Enzym. 101, 411–433 (1983).
Southern, E. M. J. molec. Biol. 98, 503–517 (1975).
Glisin, V., Crkvenkjakov, R. & Byus, C. Biochemistry 13, 2633–2637 (1974).
Bielschowsky, M. J. Psych. Neurol. 3, 169–188 (1904).
Higgins, G. A., Koh, S., Chen, K. S. & Gage, F. H. Neuron 3, 247–256 (1989).
Author information
Authors and Affiliations
Additional information
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.1038/356265a0
About this article
Cite this article
Kawabata, S., Higgins, G. & Gordon, J. RETRACTED ARTICLE: Amyloid plaques, neurofibrillary tangles and neuronal loss in brains of transgenic mice overexpressing a C-terminal fragment of human amyloid precursor protein. Nature 354, 476–478 (1991). https://doi.org/10.1038/354476a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/354476a0
This article is cited by
-
Association between ABCB1 polymorphisms and haplotypes and Alzheimer’s disease: a meta-analysis
Scientific Reports (2016)
-
Combined administration of D-galactose and aluminium induces Alzheimerlike lesions in brain
Neuroscience Bulletin (2011)
-
Alzheimer disease models and human neuropathology: similarities and differences
Acta Neuropathologica (2008)
-
Artificial chromosome-based transgenes in the study of genome function
Mammalian Genome (2006)
-
The Alzheimer's Aβ peptide induces neurodegeneration and apoptotic cell death in transgenic mice
Nature Genetics (1995)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.