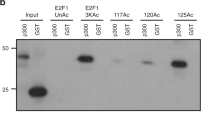
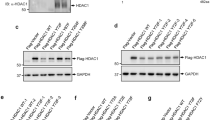
Abstract
The retinoblastoma tumour-suppressor protein Rb1 inhibits cell proliferation by repressing a subset of genes that are controlled by the E2F family of transcription factors2 and which are involved in progression from the G1 to the S phase of the cell cycle. Rb, which is recruited to target promoters by E2F1 (ref. 3), represses transcription by masking the E2F1 transactivation domain4 and by inhibiting surrounding enhancer elements5,6,7,8, an active repression that could be crucial for the proper control of progression through the cell cycle9. Some transcriptional regulators act by acetylating or deacetylating the tails protruding from the core histones10, thereby modulating the local structure of chromatin: for example, some transcriptional repressors function through the recruitment of histone deacetylases11. We show here that the histone deacetylase HDAC1 physically interacts and cooperates with Rb. In HDAC1, the sequence involved is an LXCXE motif, similar to that used by viral transforming proteins to contact Rb. Our results strongly suggest that the Rb/HDAC1 complex is a key element in the control of cell proliferation and differentiation and that it is a likely target for transforming viruses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Weinberg, R. A. The retinoblastoma gene and gene product. Cancer Surv. 12, 43–57 (1992).
Nevins, J. R. E2F: a link between the Rb tumor suppressor protein and viral oncoproteins. Science 258, 424–429 (1992).
Helin, K. et al. AcDNA encoding a pRB-binding protein with properties of the transcription factor E2F. Cell 70, 337–350 (1992).
Flemington, E. K., Speck, S. H. & Kaelin, W. G. J E2F-1-mediated transactivation is inhibited by complex formation with the retinoblastoma susceptibiity gene product. Proc. Natl Acad. Sci. USA 90, 6914–6918 (1993).
Weintraub, S. J., Prater, C. A. & Dean, D. C. Retinoblastoma protein switches the E2F site from positive to negative element. Nature 358, 259–261 (1992).
Bremner, R. et al. Direct transcriptional repression by pRB and its reversal by specific cyclins. Mol. Cell. Biol. 1555, 3256–3265 (1995).
Zacksenhaus, E., Jiang, Z., Phillips, R. A. & Gallie, B. L. Dual mechanisms of repression of E2F1 activity by the retinoblastoma gene product. EMBO J. 15, 5917–5927 (1996).
Chow, K. N., Starostik, P. & Dean, D. C. The Rb family contains a conserved cyclin-dependent-kinase-regulated transcriptional repressor motif. Mol. Cell. Biol. 16, 7173–7181 (1996).
Weinberg, R. A. E2F and cell proliferation: a world turned upside down. Cell 85, 457–459 (1996).
Turner, B. M. Histone acetylation and control of gene expression. J. Cell Sci. 99, 13–20 (1991).
Pazin, M. J. & Kadonaga, J. T. What's up and down with histone deacetylation and transcription? Cell 89, 325–328 (1997).
Yoshida, M., Kijima, M., Akita, M. & Beppu, T. Potent and specific inhibition of mammalian histone deacetylase both in vivo and in vitro by trichostatin A. J. Biol. Chem. 265, 17174–17179 (1990).
Weintraub, S. J. et al. Mechanism of active transcriptional repression by the retinoblastoma protein. Nature 375, 812–815 (1995).
Chow, K. N. & Dean, D. C. Domains A and B in the Rb pocket interact to form a transcriptional repressor motif. Mol. Cell. Biol. 16, 4862–4868 (1996).
Yang, W. M., Inouye, C., Zeng, Y., Bearss, D. & Seto, E. Transcriptional repression by YY1 is mediated by interaction with a mammalian homolog of the yeast global regulator RPD3. Proc. Natl Acad. Sci. USA 93, 12845–12850 (1996).
Taunton, J., Hassig, C. A. & Schreiber, S. L. Amammalian histone deacetylase related to the yeast transcriptional regulator Rpd3p. Science 272, 408–411 (1996).
Taya, Y. RB kinases and RB-binding proteins: new points of view. Trends Biochem. Sci. 22, 14–17 (1997).
Fattaey, A. R., Harlow, E. & Helin, K. Independent regions of adenovirus E1A are required for binding to and dissociation of E2F-protein complexes. Mol. Cell. Biol. 13, 7267–7277 (1993).
Trouche, D. et al. Hbrm is a downstream target for E2F1 repression by RB. Proc. Natl Acad. Sci. USA 94, 11295–11300 (1997).
Wolffe, A. P. & Pruss, D. Targeting chromatin disruption: Transcription regulators that acetylate histones. Cell 84, 817–819 (1996).
Alland, L. et al. Role for N-CoR and histone deacetylase in Sin3-mediated transcriptional repression. Nature 387, 49–55 (1997).
Zhang, Y., Iratni, R., Erdjument-Bromage, H., Tempst, P. & Reinberg, D. Histone deacetylases and SAP18, a novel polypeptide, are components of a human Sin3 complex. Cell 89, 357–364 (1997).
Laherty, C. D. et al. Histone deacetylases associated with the mSin3 corepressor mediate made transcriptional repression. Cell 89, 349–356 (1997).
Heinzel, T. et al. Acomplex containing N-CoR, mSin3 and histone deacetylase mediates transcriptional repression. Nature 387, 43–48 (1997).
Nagy, L. et al. Nuclear receptor repression mediated by a complex containing SMRT, mSin3A, and histone deacetylase. Cell 89, 373–380 (1997).
Hassig, C. A., Fleischer, T. C., Billin, A. N., Schreiber, S. L. & Ayer, D. E. Histone deacetylase activity is required for full transcriptional repression by mSin3A. Cell 89, 341–347 (1997).
Kaelin, W. G., Pallas, D. C., DeCaprio, J. A., Kaye, F. & Livingston, D. M. Identification of cellular proteins that can interact specifically with the T/E1A-binding region of the retinoblastoma gene product. Cell 64, 521–532 (1991).
Bandara, L. R. et al. DP-1: a cell cycle-regulated and phosphorylated component of transcription factor DRTF1/E2F which is functionally important for recognition by pRb and the adenovirus E4 orf 6/7 protein. EMBO J. 13, 3104–3114 (1994).
Groisman, R. et al. Physical interaction between the mitogen-responsive serum response factor and myogenic bHLH proteins. J. Biol. Chem. 271, 5258–5264 (1996).
Dignam, J. D., Lebowitz, R. M. & Roeder, R. G. Accurate transcription initiation by RNA polymerase II in a soluble. Nucleic Acids Res. 11, 1474–1486 (1983).
Acknowledgements
This paper is dedicated to the memory of Jacques G. Harel who originated this work. We thank N. La Thangue, W. Kaelin, T. Kouzarides, C. Hassig, E. Lam, S. A. Leibovitch, R. Watson, S.Schreiber and S.Khochbin for materials, and C. Jaulin, G. Almouzni, S. Kochbin and L. Pritchard for discussion. This work was supported by grants from the Ligue Nationale contre le Cancer, the Comité des Yvelines and theComité du Val de Marne, from the Association pour la Recherche sur le Cancer and from the Groupement des Entreprises Francaises dans la Lutte contre le Cancer. L.M.J. held a fellowship from the Société Française du Cancer; R.G. held a fellowship from the Association Française contre les Myopathies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Magnaghi-Jaulin, L., Groisman, R., Naguibneva, I. et al. Retinoblastoma protein represses transcription by recruiting a histone deacetylase. Nature 391, 601–605 (1998). https://doi.org/10.1038/35410
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35410
This article is cited by
-
Dysregulation of histone deacetylases in ocular diseases
Archives of Pharmacal Research (2024)
-
Acetyl-methyllysine marks chromatin at active transcription start sites
Nature (2023)
-
Targeting the E2F1/Rb/HDAC1 axis with the small molecule HR488B effectively inhibits colorectal cancer growth
Cell Death & Disease (2023)
-
Gcn5 and Rpd3 have a limited role in the regulation of cell cycle transcripts during the G1 and S phases in Saccharomyces cerevisiae
Scientific Reports (2019)
-
Dysregulation of histone deacetylases in carcinogenesis and tumor progression: a possible link to apoptosis and autophagy
Cellular and Molecular Life Sciences (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.