Abstract
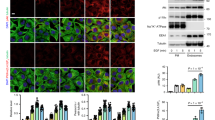
PROTEIN kinase C (PKC) transduces receptor-mediated signals by phosphorylating membrane-bound substrates which then act as effectors of specific cellular responses1. The myristoylated alanine-rich C kinase substrate (MARCKS) is a specific PKC substrate which has been implicated in macrophage activation, neurosecretion and growth factor-dependent mitogenesis2–5. Myristoylation of MARCKS is required for effective binding to the plasma membrane6 where it colocalizes with PKC7. Here we report that PKC-dependent phosphorylation displaces MARCKS from the membrane and that its subsequent dephosphorylation is accompanied by its reassociation with the membrane. This cycle of phosphorylation-dependent membrane attachment and detachment of a myristoylated protein represents a novel mechanism of reversible membrane targeting. As MARCKS is a calmodulin- and actin-binding protein (ref. 8, and J. Hartwig et al., manuscript submitted), the cycle of membrane attachment/detachment represents a mechanism through which PKC might reversibly regulate actin–membrane interaction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Nishizuka, Y. Nature 334, 661–665 (1988).
Aderem, A. A. et al. Nature 332, 362–364 (1988).
Wu, W. S., Walaas, S. I., Nairn, A. C. & Greengard, P. Proc. natn. Acad Sci. U.S.A. 79, 5249–5253 (1982).
Rozengurt, E., Rodriguez-Pena, M. & Smith, K. A. Proc. natn. Acad. Sci. U.S.A. 80, 7244–7248 (1983).
Blackshear, P. X., Wen, L., Glynn, B. P. & Witters, L. A. J. biol. Chem. 261, 1459–1469 (1986).
Graff, J. M., Gordon, J. I. & Blackshear, P. J. Science 246, 503–506 (1989).
Rosen, A., Keenan, K. F., Thelen, M., Nairn, A. C. & Aderem, A. A. J. exp. Med. 172, 1211–1215 (1990).
Graff, J. M., Young, T. M., Johnson, J. D. & Blackshear, P. J. J. biol. Chem. 264, 21818–21823 (1989).
Thelen, M., Rosen, A., Nairn, A. C. & Aderem, A. Proc. natn. Acad. Sci. U.S.A. 87, 5603–5607 (1990).
Dewald, B., Thelen, M. & Baggiolini, M. J. biol. Chem. 263, 16179–16184 (1988).
Bialojan, C. & Takai, A. Biochem. J. 256, 283–290 (1988).
Haystead, T. A. J. et al. Nature 337, 78–81 (1989).
Resh, M. D. Cell 58, 281–286 (1989).
Resh, M. D. & Ling, H. Nature 346, 84–86 (1990).
Veillette, A., Bookman, M. A., Horak, E. M. & Bolen, J. B. Cell 55, 301–308 (1988).
Borregaard, N., Heiple, J. M., Simons, E. R. & Clark, R. A. J. Cell Biol. 97, 52–61 (1983).
Rosen, A., Nairn, A. C., Greengard, P., Cohn, Z. A. & Aderem, A. A. J. biol. Chem. 264, 9118–9121 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thelen, M., Rosen, A., Nairn, A. et al. Regulation by phosphorylation of reversible association of a myristoylated protein kinase C substrate with the plasma membrane. Nature 351, 320–322 (1991). https://doi.org/10.1038/351320a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/351320a0
This article is cited by
-
Myristoylated, alanine-rich C-kinase substrate (MARCKS) regulates toll-like receptor 4 signaling in macrophages
Scientific Reports (2023)
-
Electrostatic switch mechanisms of membrane protein trafficking and regulation
Biophysical Reviews (2023)
-
Pathophysiological roles of myristoylated alanine-rich C-kinase substrate (MARCKS) in hematological malignancies
Biomarker Research (2021)
-
Pharmacological rescue of cognitive function in a mouse model of chemobrain
Molecular Neurodegeneration (2021)
-
An electrostatic switching mechanism to control the lipid transfer activity of Osh6p
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.