Abstract
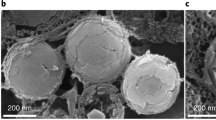
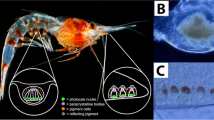
Photosensitivity in most echinoderms has been attributed to ‘diffuse’ dermal receptors1,2,3. Here we report that certain single calcite crystals used by brittlestars for skeletal construction4,5 are also a component of specialized photosensory organs, conceivably with the function of a compound eye. The analysis of arm ossicles in Ophiocoma6 showed that in light-sensitive species, the periphery of the labyrinthic calcitic skeleton extends into a regular array of spherical microstructures that have a characteristic double-lens design. These structures are absent in light-indifferent species. Photolithographic experiments in which a photoresist film was illuminated through the lens array showed selective exposure of the photoresist under the lens centres. These results provide experimental evidence that the microlenses are optical elements that guide and focus the light inside the tissue. The estimated focal distance (4–7 µm below the lenses) coincides with the location of nerve bundles—the presumed primary photoreceptors. The lens array is designed to minimize spherical aberration and birefringence and to detect light from a particular direction. The optical performance is further optimized by phototropic chromatophores that regulate the dose of illumination reaching the receptors. These structures represent an example of a multifunctional biomaterial that fulfills both mechanical and optical functions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hyman, L. H. The Invertebrates Vol. 4 (McGraw-Hill, New York, 1955).
Millott, N. The photosensitivity of echinoids. Adv. Mar. Biol. 13, 1–52 (1975).
Yoshida, M., Takasu, N. & Tamotsu, S. in Photoreception and Vision in Invertebrates; (ed. Ali, M. A.) 743–771 (Plenum, New York, 1984).
Lowenstam, H. A. & Weiner, S. On Biomineralization 123–134 (Oxford Univ. Press, Oxford, 1989).
Wainwright, S. A., Biggs, W. D., Currey, J. D. & Gosline, J. M. Mechanical Design in Organisms (John Wiley, New York, 1976).
Hendler, G. & Byrne, M. Fine structure of the dorsal arm plate of Ophiocoma wendti (Echinodermata, Ophiuroidea). Zoomorphology 107, 261–272 (1987).
Hendler, G. Brittlestar color-change and phototaxis (Echinodermata: Ophiuroidea: Ophiocomidae). Mar. Ecol. 5, 379–401 (1984).
Cowles, R. P. Stimuli produced by light and by contact with solid walls as factors in the behavior of ophiuroids. J. Exp. Zool. 9, 387–416 (1910).
Donnay, G. & Pawson, D. L. X-ray diffraction studies of echinoderm plates. Science 166, 1147–1150 (1969).
Ameye, L., Hermann, R., Wilt, F. & Dubois, P. Ultrastructural localization of proteins involved in sea urchin biomineralization. J. Histochem. Cytochem. 47, 1189–1200 (1999).
Clarkson, E. N. K. & Levi-Setti, R. Trilobite eyes and the optics of Des Cartes and Huygens. Nature 254, 663–667 (1975).
Gal, J., Horvath, G., Clarkson, E. N. K. & Haiman, O. Image formation by bifocal lenses in a trilobite eye? Vision Res. 40, 843–853 (2000).
Towe, K. M. Trilobite eyes: calcified lenses in vivo. Science 179, 1007–1010 (1973).
Döderlein, L. Ueber ‘Krystallkörper’ bei Seesternen. Denkschr. Med. Nat. Ges. Jena 8, 491–494 (1898).
Smith, A. B. in Special Papers in Palaeontology 25, 1–81 (Palaeontology Association, London, 1980).
Xia, Y. N. & Whitesides, G. M. Soft lithography. Annu. Rev. Mater. Sci. 28, 153–184 (1998).
Aizenberg, J., Rogers, J. A., Paul, K. E. & Whitesides, G. M. Imaging the irradiance distribution in the optical near field. Appl. Phys. Lett. 71, 3773–3775 (1997).
Flint, H. T. Geometrical Optics (Methuen, London, 1936).
Cobb, J. L. S. & Moore, A. Comparative studies on receptor structure in the brittlestar Ophiura ophiura. J. Neurocytol. 15, 97–108 (1986).
Johnsen, S. Identification and localization of a possible rhodopsin in the echinoderms Asterias forbesi (Asteroidea) and Ophioderma brevispinum (Ophiuroidea). Biol. Bull. 193, 97–105 (1997).
Cobb, J. L. S. & Hendler, G. Neurophysiological characterization of the photoreceptor system in brittlestars. Comp. Biochem. Physiol. 97A, 329–333 (1990).
Stubbs, T. R. in Echinoderms (ed. Lawrence, J. M.) 403–408 (Proc. Int. Echinoderms Conf., Tampa Bay, 1982).
Land, M. F. in Comparative Physiology and Evolution in Invertebrates B: Invertebrate Visual Centers and Behavior I (ed. Autrum, H.) 471–592 (Springer, Berlin, 1981).
Mann, S. & Ozin, G. A. Synthesis of inorganic materials with complex form. Nature 382, 313–318 (1996).
Belcher, A. M., Hansma, P. K., Stucky, G. D. & Morse, D. E. First steps in harnessing the potential of biomineralization. Acta Mater. 46, 733–736 (1998).
Acknowledgements
We thank P. Wiltzius and M. Megens for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aizenberg, J., Tkachenko, A., Weiner, S. et al. Calcitic microlenses as part of the photoreceptor system in brittlestars. Nature 412, 819–822 (2001). https://doi.org/10.1038/35090573
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35090573
This article is cited by
-
Light-driven nucleation, growth, and patterning of biorelevant crystals using resonant near-infrared laser heating
Nature Communications (2023)
-
Nonclassical mechanisms to irreversibly suppress β-hematin crystal growth
Communications Biology (2023)
-
Strain-tunable optical microlens arrays with deformable wrinkles for spatially coordinated image projection on a security substrate
Microsystems & Nanoengineering (2022)
-
Facile fabrication of self-doped N/O-rich hierarchical porous carbon from wild shrimp carapace with stud-like calcium carbonate arrays for supercapacitors
Biomass Conversion and Biorefinery (2022)
-
Effect of Diameter, Length, and Chirality on the Properties of Silicon Nanotubes
Silicon (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.