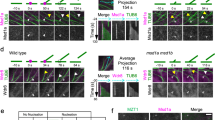
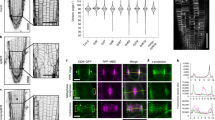
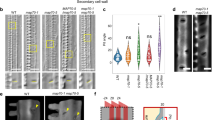
Abstract
Microtubules orchestrate cell division and morphogenesis, but how they disassemble and reappear at different subcellular locations is unknown. Microtubule organizing centres are thought to have an important role, but in higher plants microtubules assemble in ordered configurations even though microtubule organizing centres are inconspicuous or absent. Plant cells generate highly organized microtubule arrays that coordinate mitosis, cytokinesis and expansion. Inhibiting microtubule assembly prevents chromosome separation1, blocks cell division2 and impairs growth polarity3. Microtubules are essential for the formation of cell walls, through an array of plasma-membrane-associated cortical microtubules whose control mechanisms are unknown. Using a genetic strategy to identify microtubule organizing factors in Arabidopsis thaliana, we isolated temperature-sensitive mutant alleles of the MICROTUBULE ORGANIZATION 1 (MOR1) gene. Here we show that MOR1 is the plant version of an ancient family of microtubule-associated proteins4. Point mutations that substitute single amino-acid residues in an amino-terminal HEAT repeat impart reversible temperature-dependent cortical microtubule disruption, showing that MOR1 is essential for cortical microtubule organization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Baskin, T. I. & Cande, W. Z. The structure and function of the mitotic spindle in flowering plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 41, 277– 315 (1990).
Mayer, U., Herzog, U., Berger, F., Inze, D. & Jurgens, G. Mutations in the pilz group genes disrupt the microtubule cytoskeleton and uncouple cell cycle progression from cell division in Arabidopsis embryo and endosperm. Eur. J. Cell Biol. 78, 100– 108 (1999).
Baskin, T. I., Wilson, J. E., Cork, A. & Williamson, R. E. Morphology and microtubule organization in Arabidopsis roots exposed to oryzalin or taxol. Plant Cell Physiol. 35, 935– 942 (1994).
Tournebize, R. et al. Control of microtubule dynamics by the antagonistic activities of XMAP215 and XKCM1 in Xenopus egg extracts. Nature Cell Biol. 2, 13– 19 (2000).
Sugimoto, K., Williamson, R. E. & Wasteneys, G. O. New techniques enable comparative analysis of microtubule orientation, wall texture and growth rate in intact roots of Arabidopsis thaliana. Plant Physiol. 124, 1493– 1506 (2000).
Wasteneys, G. O. The cytoskeleton and growth polarity. Curr. Opin. Plant Biol. 3, 503– 511 (2000).
Charrasse, S. et al. The TOGp protein is a new human microtubule-associated protein homologous to the Xenopus XMAP215. J. Cell Sci. 111, 1371– 1383 (1998).
Cullen, C. F., Deak, P., Glover, D. M. & Ohkura, H. Mini spindles: A gene encoding a conserved microtubule-associated protein required for the integrity of the mitotic spindle in Drosophila. J. Cell Biol. 146, 1005– 1018 (1999).
Graf, R., Daunderer, C. & Schliwa, M. Dictyostelium DdCP224 is a microtubule-associated protein and a permanent centrosomal resident involved in centrosome duplication. J. Cell Sci. 113, 1747– 1758 (2000).
Wang, P. J. & Huffaker, T. C. Stu2p: A microtubule-binding protein that is an essential component of the yeast spindle pole body. J. Cell Biol. 139, 1271– 1280 (1997).
Nabeshima, K. et al. p93dis1, which is required for sister chromatid separation, is a novel microtubule and spindle pole body-associating protein phosphorylated at the Cdc2 target sites. Genes Dev. 9, 1572– 1585 (1995).
Matthews, L. R., Carter, P., Thierrymieg, D. & Kemphues, K. Zyg-9, a Caenorhabditis elegans protein required for microtubule organization and function, is a component of meiotic and mitotic spindle poles. J. Cell Biol. 141, 1159– 1168 (1998).
Andrade, M. A., Ponting, C. P., Gibson, T. J. & Bork, P. Homology-based method for identification of protein repeats using statistical significance estimates. J. Mol. Biol. 298, 521– 537 (2000).
Andrade, M. A. & Bork, P. Heat repeats in the Huntingtons disease protein. Nature Genet. 11, 115– 116 (1995).
Spittle, C., Charrasse, S., Larroque, C. & Cassimeris, L. The interaction of TOGp with microtubules and tubulin. J. Biol. Chem. 275, 20748– 20753 (2000).
Kunz, J., Schneider, U., Howald, I., Schmidt, A. & Hall, M. N. HEAT repeats mediate plasma membrane localization of Tor2p in yeast. J. Biol. Chem. 275, 37011– 37020 (2000).
Ueda, K., Matsuyama, T. & Hashimoto, T. Visualization of microtubules in living cells of transgenic Arabidopsis thaliana. Protoplasma 206, 201– 206 (1999).
Ueda, K. & Matsuyama, T. Rearrangement of cortical microtubules from transverse to oblique or longitudinal in living cells of transgenic Arabidopsis thaliana. Protoplasma 213, 28– 38 (2000).
Gard, D. L. & Kirchner, M. W. A microtubule-associated protein from Xenopus eggs that specifically promotes assembly at the plus-end. J. Cell Biol. 105, 2203– 2215 (1987).
Charrasse, S., Lorca, T., Doree, M. & Larroque, C. The Xenopus XMAP215 and its human homologue TOG proteins interact with cyclin B1 to target p34cdc2 to microtubules during mitosis. Exp. Cell Res. 254, 249– 256 (2000).
Vasquez, R. J., Gard, D. L. & Cassimeris, L. Phosphorylation by CDK1 regulates XMAP215 function in vitro. Cell. Motil. Cytoskeleton 43, 310– 321 (1999).
Andersen, S. S. Xenopus interphase and mitotic microtubule-associated proteins differentially suppress microtubule dynamics in vitro. Cell. Motil. Cytoskeleton 41, 202– 213 (1998).
Kennelly, P. J. & Krebs, E. G. Consensus sequences as substrate specificity determinants for protein kinases and protein phosphatases. J. Biol. Chem. 266, 15555– 15558 (1991).
Wick, S. Plant microtubules meet their MAPs and mimics. Nature Cell Biol. 2, E204– E206 (2000).
Lloyd, C. W. & Hussey, P. J. Microtubule-associated proteins in plants—why we need a MAP. Nature Rev. Mol. Cell Biol. 2, 40– 47 (2001).
Smertenko, A. et al. A new class of microtubule-associated proteins in plants. Nature Cell Biol. 2, 750– 753 (2000).
Wasteneys, G. O., Willingale-Theune, J. & Menzel, D. Freeze shattering: a simple and effective method for permeabilizing higher plant cell walls. J. Microsc. 188, 51– 61 (1997).
Konieczny, A. & Ausubel, F. M. A procedure for mapping Arabidopsis mutations using co-dominant ecotype-specific PCR-based markers. Plant J. 4, 403– 410 (1993).
Hall, T. A. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 41, 95– 98 (1999).
Acknowledgements
R. E. Williamson provided support from the inception of this project and supplied EMS-mutagenized seed stock. The Arabidopsis Biological Resource Centre, Ohio State University, supplied BACs. We thank T. Hashimoto and K. Ueda for the GFP::TUA6 line, M. Luo and A. Chaudhury for the binary clone used in complementation, H. Kazama for helpful advice, R. Heady and A. Knox for microscopy assistance, and J. B. Gibson, B. E. S. Gunning and A. R. Hardham for comments on the manuscript. An Australian Research Council QEII Fellowship to G.O.W. supported this work. Coauthor contributions include: mutant identification and genetic analysis (G.O.W. and N.G.H.), gene mapping (O.V., K.-J.W., A.T.W. & G.O.W.), cloning and sequence analysis (A.T.W., K.-J.W., M.C.R. & G.O.W.), microtubule dynamics (G.O.W.) and morphological analysis (K.S., N.G.H. & G.O.W.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary gene maps
AtMOR1 nucleotide and deduced amino acid sequences. Splice junctions (><) are shown and exons are numbered from the start codon (underlined). mor1 mutation sites are indicated (Ñ). This RT-PCR-derived sequence data (GenBank AF367246) is slightly different to that predicted (GenBank AC006068.3) due to inaccurate prediction of some splice sites by the latter.
Rights and permissions
About this article
Cite this article
Whittington, A., Vugrek, O., Wei, K. et al. MOR1 is essential for organizing cortical microtubules in plants. Nature 411, 610–613 (2001). https://doi.org/10.1038/35079128
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35079128
This article is cited by
-
Cortical microtubule dynamics during reaction wood formation ensures context-appropriate cellulose microfibril angle in woody trees
Trees (2024)
-
Anatomical observation and transcriptome analysis of branch-twisted mutations in Chinese jujube
BMC Genomics (2023)
-
Rice kinesin-related protein STD1 and microtubule-associated protein MAP65-5 cooperatively control microtubule bundling
Planta (2023)
-
CLASP balances two competing cell division plane cues during leaf development
Nature Plants (2022)
-
Mutation in Arabidopsis MOR1 gene impairs endocytosis in stamen filament cells and results in anther indehiscence
Plant Growth Regulation (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.