Key Points
-
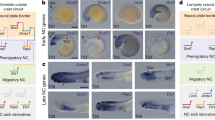
Craniofacial development is closely related to the segmentation of the hindbrain into rhombomeres. This segmentation is critical for the appropriate development of several cranial nerves as well as for defining the migration pathways of the neural crest cells towards the periphery.
-
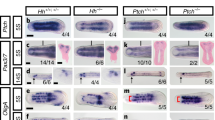
There is a molecular correlate of hindbrain segmentation, as the expression of two gene families are essential for establishing compartments and imposing segmental identity — the ephrin/Eph receptors and the Hox transcription factors. The expression of the Hox genes is not exclusive to the hindbrain itself, but it is also evident in the neural crest cells that populate the branchial arches, providing further evidence that hindbrain segmentation has direct effects on craniofacial patterning.
-
Early experiments grafting avian neural crest primordia to ectopic locations indicated that the morphogenetic programme and the Hox gene code of the neural crest cells is acquired before migration from the neural tube and is merely carried to the periphery. This idea has been also favoured by rhombomere-transplantation experiments to sites anterior to the otic vesicle.
-
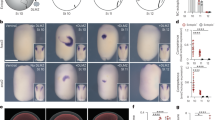
More recent experiments have shown that the morphogenetic programme and Hox gene expression are not irreversibly committed but can show plasticity. Chick rhombomere transplantations to locations caudal to the otic vesicle, as well as rhombomere rotations and ablations, have shown that environmental factors can influence the fate of migratory neural crest cells, although the evidence has not been entirely homogeneous.
-
Observations made in other species have also provided evidence for Hox gene expression plasticity. Grafting of small numbers of cells has revealed that the size of the cell community is an important determinant of the degree of plasticity such that smaller grafts or individual cells are more sensitive to environmental cues. Indeed, this dependence on community size could potentially help to explain the heterogeneity of the observations made in chick embryos.
-
Regulatory sequences that drive the region-specific expression of Hox genes have begun to be identified. Although more cases need to be investigated, it is possible that independent regulatory elements may be used for controlling their expression in mesenchymal versus neurogenic neural crest cell derivatives.
-
The plasticity observed at the single cell level induce by environmental cues may be important for the establishment of precise boundaries in the hindbrain. This phenomenon in combination with the ephrin/Eph receptor-mediated segregation may provide a mechanism for the generation of precise rhombomeric territories during development.
Abstract
Understanding the patterning mechanisms that control head development — particularly the neural crest and its contribution to bones, nerves and connective tissue — is an important problem, as craniofacial anomalies account for one-third of all human congenital defects. Classical models for craniofacial patterning argue that the morphogenic program and Hox gene identity of the neural crest is pre-patterned, carrying positional information acquired in the hindbrain to the peripheral nervous system and the branchial arches. Recently, however, plasticity of Hox gene expression has been observed in the hindbrain and cranial neural crest of chick, mouse and zebrafish embryos. Hence, craniofacial development is not dependent on neural crest pre-patterning, but is regulated by a more complex integration of cell and tissue interactions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fraser, S., Keynes, R. & Lumsden, A. Segmentation in the chick embryo hindbrain is defined by cell lineage restrictions. Nature 344, 431–435 (1990).
Guthrie, S. & Lumsden, A. Formation and regeneration of rhombomere boundaries in the developing chick hindbrain. Development 112, 221–229 (1991).
Guthrie, S., Prince, V. & Lumsden, A. Selective dispersal of avian rhombomere cells in orthotopic and heterotopic grafts. Development 118, 527–538 (1993).
Birgbauer, E. & Fraser, S. E. Violation of cell lineage restriction compartments in the chick hindbrain. Development 120 , 1347–1356 (1994).
Xu, Q., Mellitzer, G., Robinson, V. & Wilkinson, D. In vivo cell sorting in complementary segmental domains mediated by Eph receptors and ephrins. Nature 399, 267– 271 (1999).Together with paper below, implicates the Eph receptor/ephrin families in sorting mechanisms in the hindbrain, providing a key means of sharpening rhombomere boundaries.
Mellitzer, G., Xu, Q. & Wilkinson, D. Eph receptors and ephrins restrict cell intermingling and communciation. Nature 400, 77– 81 (1999).Important evidence that bidirectional signalling between two populations can function to restrict the mixing potential and lineage relationships of opposing cell populations. Relevant to how hindbrain rhombomere populations are separated.
Wilkinson, D. G., Bhatt, S., Cook, M., Boncinelli, E. & Krumlauf, R. Segmental expression of Hox-2 homeobox-containing genes in the developing mouse hindbrain. Nature 341 , 405–409 (1989).
Hunt, P. et al. A distinct Hox code for the branchial region of the head . Nature 353, 861–864 (1991).
Lumsden, A. & Krumlauf, R. Patterning the vertebrate neuraxis . Science 274, 1109–1115 (1996).
Keynes, R. & Krumlauf, R. Hox genes and regionalization of the nervous system. Annu. Rev. Neurosci. 17, 109–132 (1994).
Wintgate, R. & Lumsden, A. Persistence of rhombomeric organisation in the postsegmental avian hindbrain. Development 122 , 2143–2152 (1996).
Marín, F. & Puelles, L. Morphological fate of rhombomeres in quail/chick chimeras: a segmental analysis of hindbrain nuclei. Eur. J. Neurosci. 7, 1714– 1738 (1995).
Lumsden, A. & Keynes, R. Segmental patterns of neuronal development in the chick hindbrain. Nature 337, 424– 428 (1989).
Sechrist, J. & Bronner-Fraser, M. Birth and differentiation of reticular neurons in the chick hindbrain: Ontogeny of the first neuronal population. Neuron 7, 947– 963 (1991).
Clarke, J. D. & Lumsden, A. Segmental repetition of neuronal phenotype sets in the chick embryo hindbrain. Development 118, 151–162 (1993).
Marshall, H. et al. Retinoic acid alters hindbrain Hox code and induces transformation of rhombomeres 2/3 into a 4/5 identity. Nature 360, 737–741 (1992).
Carpenter, E. M., Goddard, J. M., Chisaka, O., Manley, N. R. & Capecchi, M. R. Loss of Hoxa-1 (Hox-1.6) function results in the reorganization of the murine hindbrain. Development 118, 1063–1075 (1993).
Gilland, E. & Baker, R. Conservation of neuroepithelial and mesodermal segments in the embryonic vertebrate head. Acta Anat. 148, 110–123 ( 1993).
Lumsden, A., Sprawson, N. & Graham, A. Segmental origin and migration of neural crest cells in the hindbrain region of the chick embryo. Development 113, 1281–1291 (1991).
Sechrist, J., Serbedzija, G. N., Scherson, T., Fraser, S. E. & Bronner-Fraser, M. Segmental migration of the hindbrain neural crest does not arise from its segmental generation. Development 118, 691–703 (1993).Important fate mapping data, which revealed that odd-numbered rhombomeres generate neural crest and that crest cells from these segments migrate in unusual routes generating contributions to several arches.
Serbedzija, G., Fraser, S. & Bronner-Fraser, M. Vital dye analysis of cranial neural crest cell migration in the mouse embryo. Development 116, 297 –307 (1992).
Couly, G. & LeDouarin, N. The fate map of cephalic neural primordium at the presomitic to the 3-somite stage in the avian embryo. Development 103, S101–S113 (1988).
Köntges, G. & Lumsden, A. Rhombencephalic neural crest segmentation is preserved throughout craniofacial ontogeny. Development 122, 3229–3242 (1996).Key fate-mapping data in avian embryos that shows the detailed register between neural crest and its contributions to branchial arch derivatives.
Graham, A., Heyman, I. & Lumsden, A. Even-numbered rhombomeres control the apoptotic elimination of neural crest cells from odd-numbered rhombomeres in the chick hindbrain . Development 119, 233– 245 (1993).
Graham, A., Francis-West, P., Brickell, P. & Lumsden, A. The signalling molecule BMP4 mediates apoptosis in the rhombencephalic neural crest. Nature 372, 684– 686 (1994).
Kulesa, P. & Fraser, S. Neural crest cell dynamics revealed by time-lapse video microscopy of whole chick explant cultures. Dev. Biol. 204, 327–344 (1998).An important study on the dynamic migration of cranial neural crest cells. In combination with an in ovo study, this paper uncovered extensive cell interactions between dispersed and migratory neural crest cells. It illustrated complex migratory pathways and the fact that cells are normally testing the environment. It helped to explain many of the community effects seen in chick grafts.
Trainor, P., Manzanares, M. & Krumlauf, R. in Results and Problems in Cell Differentiation (eds Goffinet, A. & Rackic, P.) 51–89 (Springer, Berlin, 2000).
Schneider-Maunoury, S. et al. Disruption of Krox-20 results in alteration of rhombomeres 3 and 5 in the developing hindbrain. Cell 75, 1199–1214 (1993).
Schneider-Maunoury, S., Seitanidou, T., Charnay, P. & Lumsden, A. Segmental and neuronal architecture of the hindbrain of Krox-20 mouse mutants. Development 124, 1215– 1226 (1997).
Swiatek, P. J. & Gridley, T. Perinatal lethality and defects in hindbrain development in mice homozygous for a targeted mutation of the zinc finger gene Krox-20. Genes Dev. 7, 2071–2084 (1993).
Mark, M. et al. Two rhombomeres are altered in Hoxa-1 mutant mice. Development 119, 319–338 (1993).
Dollé, P. et al. Local alterations of Krox-20 and Hox gene expression in the hindbrain suggest lack of rhombomeres 4 and 5 in homozygote null Hoxa-1 (Hox-1. 6) mutant embryos. Proc. Natl Acad. Sci. USA 90, 7666–7670 ( 1993).
Cordes, S. P. & Barsh, G. S. The mouse segmentation gene kr encodes a novel basic domain-leucine zipper transcription factor. Cell 79, 1025–1034 ( 1994).
Moens, C. B., Yan, Y.-L., Appel, B., Force, A. G. & Kimmel, C. B. valentino: a zebrafish gene required for normal hindbrain segmentation. Development 122, 3981–3990 (1996).
Moens, C. B., Cordes, S. P., Giorgianni, M. W., Barsh, G. S. & Kimmel, C. B. Equivalence in the genetic control of hindbrain segmentation in fish and mouse. Development 125, 381–391 (1998).
Manzanares, M. et al. The role of kreisler in segmentation during hindbrain development. Dev. Biol. 211, 220– 237 (1999).
Sham, M. H. et al. The zinc finger gene Krox-20 regulates Hoxb-2 (Hox2. 8) during hindbrain segmentation. Cell 72, 183–196 (1993).
Nonchev, S. et al. Segmental expression of Hoxa-2 in the hindbrain is directly regulated by Krox-20. Development 122 , 543–554 (1996).
Vesque, C. et al. Hoxb-2 transcriptional activation by Krox-20 in vertebrate hindbrain requires an evolutionary conserved cis-acting element in addition to the Krox-20 site. EMBO J. 15 , 5383–5896 (1996).
Manzanares, M. et al. Segmental regulation of Hoxb3 by kreisler. Nature 387, 191–195 ( 1997).
Manzanares, M. et al. Conserved and distinct roles of kreisler in regulation of the paralogous Hoxa3 and Hoxb3 genes. Development 126, 759–769 ( 1999).
Pöpperl, H. et al. Segmental expression of Hoxb1 is controlled by a highly conserved autoregulatory loop dependent upon exd/Pbx. Cell 81, 1031–1042 ( 1995).
Studer, M. et al. Genetic interaction between Hoxa1 and Hoxb1 reveal new roles in regulation of early hindbrain patterning. Development 125, 1025–1036 (1998).
Krumlauf, R. Hox genes in vertebrate development. Cell 78, 191–201 (1994).
McGinnis, W. & Krumlauf, R. Homeobox genes and axial patterning . Cell 68, 283–302 (1992).
Goddard, J., Rossel, M., Manley, N. & Capecchi, M. Mice with targeted disruption of Hoxb1 fail to form the motor nucleus of the VIIth nerve . Development 122, 3217– 3228 (1996).
Gavalas, A. et al. Hoxa1 and Hoxb1 synergize in patterning the hindbrain, cranial nerves and second pharyngeal arch. Development 125, 1123–1136 ( 1998).
Studer, M., Lumsden, A., Ariza-McNaughton, L., Bradley, A. & Krumlauf, R. Altered segmental identity and abnormal migration of motor neurons in mice lacking Hoxb-1. Nature 384, 630–635 ( 1996).
Zhang, M. et al. Ectopic Hoxa-1 induces rhombomere transformation in mouse hindbrain. Development 120, 2431– 2442 (1994).
Barrow, J. R., Stadler, H. S. & Capecchi, M. R. Roles of Hoxa1 and Hoxa2 in patterning the early hindbrain of the mouse. Development 127, 933–944 (2000).
Helmbacher, F. et al. Hoxa1 and Krox20 synergize to control the development of rhombomere 3. Development 125, 4739– 4748 (1998).
Xu, Q., Alldus, G., Holder, N. & Wilkinson, D. G. Expression of truncated Sek-1 receptor tyrosine kinase disrupts the segmental restriction of gene expression in the Xenopus and zebrafish hindbrain . Development 121, 4005– 4016 (1995).
Gale, N. W. et al. Eph receptors and ligands comprise two major specificity subclasses and are reciprocally compartmentalized during embryogenesis. Neuron 17, 9–19 ( 1996).
Gilardi-Hebenstreit, P. et al. An EPH-related receptor protein-tyrosine kinase gene segmentally-expressed in the developing mouse hindbrain. Oncogene 7, 2499–2506 (1992).
Becker, N. et al. Several receptor tyrosine kinase genes of the Eph family are segmentally expressed in the developing hindbrain. Mech. Dev. 47, 3–18 ( 1994).
Nieto, M. A., Gilardi-Hebenstreit, P., Charnay, P. & Wilkinson, D. A receptor protein tyrosine kinase implicated in the segmental patterning of the hindbrain and mesoderm. Development 116, 1137–1150 (1992).
Theil, T. et al. Segmental expression of the EphA4 (Sek-1) receptor tyrosine kinase in the hindbrain is under the direct transcriptional control of Krox20 . Development 125, 443– 452 (1998).
Chen, J. & Ruley, H. An enhancer element in the EphA2 (Eck) gene sufficient for rhombomere-specific expression is activated by Hoxa1 and Hoxb1. J. Biol. Chem. 273 , 24670–24675 (1998).
Taneja, R. et al. The expression pattern of the mouse receptor tyrosine kinase gene MDK1 is conserved through evolution and requires Hoxa-2 for rhombomere-specific expression in mouse embryos. Dev. Biol. 177, 397–412 (1996).
Smith, A., Robinson, V., Patel, K. & Wilkinson, D. G. The EphA4 and EphB1 receptor tyrosine kinases and ephrin-B2 ligand regulate targeted migration of branchial neural crest cells. Curr. Biol. 7, 561–570 (1997).
Krull, C. E. et al. Interactions of Eph-related receptors and ligands confer rostrocaudal pattern to trunk neural crest migration. Curr. Biol. 7, 571–580 (1997).
Hunt, P., Wilkinson, D. & Krumlauf, R. Patterning the vertebrate head: murine Hox 2 genes mark distinct subpopulations of premigratory and migrating neural crest . Development 112, 43–51 (1991).
Chisaka, O. & Capecchi, M. Regionally restricted developmental defects resulting from targeted disruption of the mouse homeobox gene hox1.5. Nature 350, 473– 479 (1991).
Gendron-Maguire, M., Mallo, M., Zhang, M. & Gridley, T. Hoxa-2 mutant mice exhibit homeotic transformation of skeletal elements derived from cranial neural crest. Cell 75, 1317– 1331 (1993).
Rijli, F. M. et al. A homeotic transformation is generated in the rostral branchial region of the head by disruption of Hoxa-2, which acts as a selector gene. Cell 75, 1333–1349 (1993).References 64 and 65 are two key papers that illustrate the important role of Hoxa2 in controlling mesenchymal arch derivatives and raise numerous issues with regard to the ground state of cranial neural crest.
Condie, B. & Capecchi, M. Mice homozygous for a targeted disruption of Hoxd-3(Hox-4.1) exhibit anterior transformations of the first and second cervical vertebrae, the atlas and axis. Development 119, 579–595 ( 1993).
Ramirez-Solis, R., Zheng, H., Whiting, J., Krumlauf, R. & Bradley, A. Hox-B4 (Hox-2.6) mutant mice show homeotic transformation of cervical vertebra and defects in the closure of the sternal rudiments. Cell 73, 279– 294 (1993).
Barrow, J. & Capecchi, M. Targeted disruption of the Hoxb2 locus in mice interferes with expression of Hoxb1 and Hoxb4. Development 122, 3817– 3828 (1996).
Le Douarin, N. The Neural Crest (Cambridge Univ. Press, Cambridge, 1983).
Noden, D. The role of the neural crest in patterning of avian cranial skeletal, connective, and muscle tissues. Dev. Biol. 96, 144– 165 (1983).A landmark paper that provided the first evidence that, on transposition to new locations, neural crest cells are capable of duplicating structures from their point of origin. It argued for a pre-patterning of the crest before it migrates from the neural tube.
Noden, D. Interactions and fates of avian craniofacial mesenchyme. Development 103, 121–140 ( 1988).
Noden, D. The control of avian cephalic neural crest cytodifferentiation. II. Neural tissues. Dev. Biol. 67, 313– 329 (1978).
Noden, D. The control of avian cephalic neural crest cytodifferentiation. I. Skeletal and connective tissues. Dev. Biol. 67, 296 –312 (1978).
Guthrie, S. et al. Neuroectodermal autonomy of Hox-2.9 expression revealed by rhombomere transpositions. Nature 356, 157–159 (1992).
Kuratani, S. C. & Eichele, G. Rhombomere transposition repatterns the segmental organization of cranial nerves and reveals cell-autonomous expression of a homeodomain protein. Development 117 , 105–117 (1993).
Prince, V. & Lumsden, A. Hoxa-2 expression in normal and transposed rhombomeres: independent regulation in the neural tube and neural crest. Development 120, 911– 923 (1994).
Simon, H., Hornbruch, A. & Lumsden, A. Independent assignment of antero-posterior and dorso-ventral positional values in the developing chick hindbrain. Curr. Biol. 5, 205–214 ( 1995).
Grapin-Botton, A., Bonnin, M.-A., Ariza-McNaughton, L., Krumlauf, R. & LeDouarin, N. M. Plasticity of transposed rhombomeres: Hox gene induction is correlated with phenotypic modifications. Development 121, 2707–2721 (1995).Important evidence that hindbrain rhombomeres show plasticity in Hox gene expression when grafted to new locations. Suggested that genetic programs of anterior–posterior patterning were not autonomous as previously thought.
Itasaki, N., Sharpe, J., Morrison, A. & Krumlauf, R. Reprogramming Hox expression in the vertebrate hindbrain: Influence of paraxial mesoderm and rhombomere transposition. Neuron 16, 487–500 (1996).Novel interspecies grafting between mouse and chick and within chick embryos to show that plasticity of rhombomeres in posterior grafts is dependent on signals from somites. Suggested that somites and paraxial mesoderm might have a normal role in patterning the CNS.
Grapin-Botton, A., Bonnin, M.-A. & Le Douarin, N. Hox gene induction in the neural tube depends on three parameters: competence, signal supply and paralogue group. Development 124, 849–859 (1997).
Gould, A., Itasaki, N. & Krumlauf, R. Initiation of rhombomeric Hoxb4 expression requires induction by somites and a retinoid pathway. Neuron 21, 39–51 (1998). An important regulatory study that linked the plasticity of Hox gene expression induced by somites with a normal role for signals from mesoderm in establishing normal domains of Hoxb4 gene expression.
Saldivar, J., Krull, C., Krumlauf, R., Ariza-McNaughton, L. & Bronner-Fraser, M. Rhombomere of origin determines autonomous versus environmentally regulated expression of Hoxa3 in the avian embryo . Development 122, 895– 904 (1996).Chick rotations experiments that revealed differential autonomy and plasticity of cranial neural crest depending on the rhombomere of origin. Showed that the dynamics of crest patterning are not uniform but vary with environmental influence.
Hunt, P., Clarke, J. D. W., Buxton, P., Ferretti, P. & Thorogood, P. Stability and plasticity of neural crest patterning and branchial arch Hox code after extensive cephalic crest rotation. Dev. Biol. 198, 82– 104 (1998).Important study showing that plasticity or autonomy can vary depending on the type of manipulation. It also showed that the apparent autonomy of Hox gene expression can vary with the stage of analysis as patterns can be reprogrammed at later times following the manipulations.
Yntema, C. & Hammond, W. Depletions and abnormalities in the cervical sympathetic system of the chick following exterpation of the neural crest. J. Exp. Zool. 100, 237– 263 (1945).
McKee, G. & Ferguson, M. The effects of mesencephalic neural crest cell extirpation on the development of chicken embryos. J. Anat. 139, 491–512 ( 1984).
Scherson, T., Serbedzija, G., Fraser, S. & Bronner-Fraser, M. Regulative capacity of the cranial neural tube and neural crest. Development 118, 1049–1061 (1993).
Hunt, P., Ferretti, P., Krumlauf, R. & Thorogood, P. Restoration of normal Hox code and branchial arch morphogenesis after extensive deletion of hindbrain neural crest. Dev. Biol. 168, 584–597 (1995).
Diaz, C. & Glover, J. Appropriate pattern formation following regulative regeneration in the hindbrain neural tube. Development 122, 3095–3105 ( 1996).
Couly, G. F., Grapin-Bottom, A., Coltey, P. & Le Douarin, N. M. The regeneration of the cephalic neural crest, a problem revisited: the regenerating cells originate from the contralateral or from the anterior and posterior neural folds. Development 122, 3393– 3407 (1996).
Saldivar, J. R., Sechrist, J. W., Krull, C. E., Ruffin, S. & Bronner-Fraser, M. Dorsal hindbrain ablation results in the rerouting of neural crest migration and the changes in gene expression, but normal hyoid development. Development 124, 2729–2739 (1997).
Noden, D. Patterning of avian craniofacial muscles. Dev. Biol. 116, 347–356 (1986).
Trainor, P. & Krumlauf, R. Plasticity in mouse neural crest cells reveals a new patterning role for cranial mesoderm. Nature Cell Biol. 2, 96–102 ( 2000).The application of grafting experiments, genetic markers and lineage tracing to mouse embryos was used for the first time as a means of investigating patterning in head development. It showed evidence for plasticity of rhombomeric and neural crest at the cellular level. First evidence that head mesoderm is important for regulation of crest patterning.
Golding, J., Trainor, P., Krumlauf, R. & Gassman, M. Defects in pathfinding by cranial neural crest cells in mice lacking the Neuregulin receptor ErbB4. Nature Cell Biol. 2, 103 –109 (2000).Important application of new grafting methods in cultured mouse embryos to evaluate the role of environmental signals on neural crest migration and plasticity in wild-type and ErbB4 mutant embryos.
Kulesa, P., Bronner-Fraser, M. & Fraser, S. In ovo time-lapse analysis after dorsal neural tube ablation shows rerouting of chick hindbrain neural crest. Development 127, 2843–2852 (2000).An important study revealing the dynamic movements of cranial neural crest cells in ovo . It further confirmed the extensive cell interactions and connections between dispersed and migratory neural crest cells, and showed the importance of environmental signalling in neural crest migration.
Couly, G., Grapin-Botton, A., Coltey, P., Ruhin, B. & Le Douarin, N. M. Determination of the identity of the derivatives of the cephalic neural crest: incompatibility between Hox gene expression and lower jaw development. Development 128, 3445–3459 ( 1998).Critical paper revealing autonomy of the genetic programme of Hoxa2 . Clearly linked correct patterns of Hoxa2 expression with the morphogenesis of jaw structures. It raised a number of issues with respect to the ground state of cranial neural crest.
Frasch, M., Chen, X. & Lufkin, T. Evolutionary-conserved enhancers direct region-specific expression of the murine Hoxa-1 and Hoxa-2 loci in both mice and Drosophila. Development 121, 957–974 (1995).
Maconochie, M. et al. Regulation of Hoxa2 in cranial neural crest cells involves members of the AP-2 family. Development 126, 1483–1494 (1999). Key molecular evidence that the neural crest and hindbrain patterns of Hoxa2 gene expression are independently regulated. Argues against a pre-patterning model.
Nonchev, S. et al. The conserved role of Krox-20 in directing Hox gene expression during vertebrate hindbrain segmentation. Proc. Natl Acad. Sci. USA 93, 9339–9345 (1996).
Maconochie, M. et al. Cross-regulation in the mouse HoxB complex: the expression of Hoxb2 in rhombomere 4 is regulated by Hoxb1. Genes Dev. 11, 1885–1896 (1997).
Chisaka, O., Musci, T. & Capecchi, M. Developmental defects of the ear, cranial nerves and hindbrain resulting from targeted disruption of the mouse homeobox gene Hox-1.6. Nature 355, 516– 520. (1992).
Lufkin, T., Dierich, A., LeMeur, M., Mark, M. & Chambon, P. Disruption of the Hox-1.6 homeobox gene results in defects in a region corresponding to its rostral domain of expression. Cell 66, 1105–1119 ( 1991).
Gavalas, A., Davenne, M., Lumsden, A., Chambon, P. & Rijli, F. Role of Hoxa-2 in axon pathfinding and rostral hindbrain patterning. Development 124, 3693 –3702 (1997).
Northcutt, R. & Gans, C. The genesis of neural crest and epidermal placodes: a reinterpretation of vertebrate origns. Q. Rev. Biol. 58, 1–28 (1983).
Gans, C. & Northcutt, R. Neural crest and the origin of vertebrates: a new head. Science 220, 268 –274 (1983).
Gans, C. Stages in the origin of vertebrates: analysis by means of scenarios. Biol. Rev. 64, 221–268 (1989).
Rijli, F., Gavalas, A. & Chambon, P. Segmentation and specification in the branchial region of the head: The role of Hox selector genes. Int. J. Dev. Biol. 42, 393–401 ( 1998).
Mallo, M. & Brandlin, I. Segmental identity can change independently in the hindbrain and rhombencephalic neural crest. Dev. Dynam. 210, 146–156 ( 1997).
Acknowledgements
We thank members of the lab for valuable discussions and T. Schilling for communicating results before publication. P.T. was supported by EMBO and HFSP postdoctoral fellowships and R.K. by Core MRC Programme support and an EEC Biotechnology Network grant.
Author information
Authors and Affiliations
Glossary
- PARAXIAL MESODERM
-
Region of the mesoderm adjacent to the notochord, which becomes segmented rostro-caudally to give rise to the somites early in development.
- NEURAL CREST
-
Group of cells that migrate from the neural tube to the periphery, where they give rise to a wide variety of cell types.
- MESENCHYME
-
Undifferentiated connective tissue present in the early embryo.
- HINDBRAIN
-
Division of the nervous system that consists of the pons, the cerebellum and the medulla. It is derived from the rhombencephalon, one of the three primary embryonic vesicles of the neural tube.
- HOX GENES
-
Transcription factors expressed in specific patterns, which are important for determining regional identity along the antero-posterior axis in the embryo. They are also known as homeobox genes.
- CRANIAL GANGLIA
-
Groups of neurons that carry somatosensory information from the head and neck to the central nervous system.
- BRANCHIOMOTOR NERVES
-
Cranial nerves that control muscles derived from the branchial arches. In humans, they are the trigeminal, the facial, the glossopharyngeal, the vagus and the spinal accessory nerves.
- BRANCHIAL ARCHES
-
Structures populated by neural crest cells found in the early embryo, which give rise to most facial structures in the adult.
- MOSAIC
-
Tissue containing two or more genetically distinct cell types.
- DOMINANT-NEGATIVE
-
A mutant protein capable of forming a heteromeric complex with the normal molecule, knocking out the activity of the entire complex.
- OTIC VESICLE
-
Ectodermal invagination that constitutes the primordium of the internal ear.
- SOMITES
-
Axial blocks of mesoderm along the vertebrate axis that differentiate into skin, bone and muscle.
- MESENCEPHALON
-
One of the three primary embryonic vesicles of the neural tube, which gives rise to the midbrain.
- RHOMBENCEPHALON
-
One of the three primary embryonic vesicles of the neural tube, which gives rise to the hindbrain.
- CELL AUTONOMY
-
A genetic trait in which only the genotype of the cell determines the corresponding phenotype. In contrast, a non-cell-autonomous phenotype is caused by the action of other cells on the cell of interest.
- NEUREGULIN
-
Protein implicated in neural and muscular development. Among other functions, it stimulates the proliferation of Schwann cells and the rate of synthesis of nicotinic acetylcholine receptors in muscle.
- HOMEOTIC TRANSFORMATION
-
The conversion of one body part into another due to mutations in a Hox gene.
- RETINOIC ACID
-
A morphogen and regulator of differentiation during embryogenesis.
Rights and permissions
About this article
Cite this article
Trainor, P., Krumlauf, R. Patterning the cranial neural crest: Hinbrain segmentation and hox gene plasticity. Nat Rev Neurosci 1, 116–124 (2000). https://doi.org/10.1038/35039056
Issue Date:
DOI: https://doi.org/10.1038/35039056
This article is cited by
-
Cellular and molecular mechanisms of the organogenesis and development, and function of the mammalian parathyroid gland
Cell and Tissue Research (2023)
-
Evolutionary divergence of a Hoxa2b hindbrain enhancer in syngnathids mimics results of functional assays
Development Genes and Evolution (2021)
-
The genetic architecture of human brainstem structures and their involvement in common brain disorders
Nature Communications (2020)