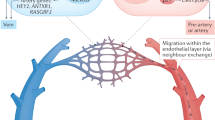
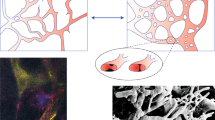
Abstract
A recent explosion in newly discovered vascular growth factors has coincided with exploitation of powerful new genetic approaches for studying vascular development. An emerging rule is that all of these factors must be used in perfect harmony to form functional vessels. These new findings also demand re-evaluation of therapeutic efforts aimed at regulating blood vessel growth in ischaemia, cancer and other pathological settings.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ferrara, N. Vascular endothelial growth factor: molecular and biological aspects. Curr. Top. Microbiol. Immunol. 237, 1– 30 (1999).
Dvorak, H. F., Nagy, J. A., Feng, D., Brown, L. F. & Dvorak, A. M. Vascular permeability factor/vascular endothelial growth factor and the significance of microvascular hyperpermeability in angiogenesis . Curr. Top. Microbiol. Immunol. 237, 97 –132 (1999).
Eriksson, U. & Alitalo, K. Structure, expression and receptor-binding properties of novel vascular endothelial growth factors. Curr. Top. Microbiol. Immunol. 237, 41–57 (1999).
Risau, W. Mechanisms of angiogenesis. Nature 386, 671–674 (1997).
Kenyon, B. M. et al. A model of angiogenesis in the mouse cornea. Invest. Ophthalmol. Vis. Sci. 37, 1625–1632 (1996).
Ribatti, D., Vacca, A., Roncali, L. & Dammacco, F. The chick embryo chorioallantoic membrane as a model for in vivo research on angiogenesis. Int. J. Dev. Biol. 40, 1189–1197 (1996).
Gale, N. W. & Yancopoulos, G. D. Growth factors acting via endothelial cell-specific receptor tyrosine kinases: VEGFs, angiopoietins, and ephrins in vascular development. Genes Dev. 13, 1055–1066 (1999).
Carmeliet, P. Mechanisms of angiogenesis and arteriogenesis. Nature Med. 6, 389–395 (2000).
Hellstrom, M., Kaln, M., Lindahl, P., Abramsson, A. & Betsholtz, C. Role of PDGF-B and PDGFR-beta in recruitment of vascular smooth muscle cells and pericytes during embryonic blood vessel formation in the mouse. Development 126, 3047– 3055 (1999).
Hirschi, K. K., Rohovsky, S. A., Beck, L. H., Smith, S. R. & D'Amore, P. A. Endothelial cells modulate the proliferation of mural cell precursors via platelet-derived growth factor-BB and heterotypic cell contact. Circ. Res. 84, 298–305 (1999).
Suri, C. et al. Requisite role of Angiopoietin-1, a ligand for the Tie2 receptor, during embryonic angiogenesis. Cell 87, 1171–1180 (1996).
Maisonpierre, P. C. et al. Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science 277, 55– 60 (1997).
Holash, J., Wiegand, S. J. & Yancopoulos, G. D. New model of tumor angiogenesis: dynamic balance between vessel regression and growth mediated by angiopoietins and VEGF. Oncogene 18, 5356–5362 ( 1999).
Holash, J. et al. Vessel cooption, regression, and growth in tumors mediated by angiopoietins and VEGF. Science 284, 1994–1998 (1999).
Soker, S., Takashima, S., Miao, H., Neufeld, G. & Klagsbrun, M. Neuropilin-1 is expressed by endothelial and tumor cells as an isoform-specific receptor for vascular endothelial growth factor. Cell 92, 735–745 ( 1998).
Shalaby, F. et al. Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature 376, 62–66 (1995).
Fong, G. H., Rossant, J., Gertenstein, M. & Breitman, M. L. Role of the Flt-1 receptor tyrosine kinase in regulating assembly of vascular endothelium. Nature 376, 66– 70 (1995).
Hiratsuka, S., Minowa, O., Kuno, J., Noda, T. & Shibuya, M. Flt-1 lacking the tyrosine kinase domain is sufficient for normal development and angiogenesis in mice. Proc. Natl Acad. Sci. USA 95, 9349–9354 ( 1998).
Taipale, J. et al. Vascular endothelial growth factor receptor-3. Curr. Top. Microbiol. Immunol. 237, 85– 96 (1999).
Persico, M. G., Vincenti, V. & DiPalma, T. Structure, expression and receptor-binding properties of placenta growth factor (PlGF). Curr. Top. Microbiol. Immunol. 237, 31–40 ( 1999).
Olofsson, B., Jeltsch, M., Eriksson, U. & Alitalo, K. Current biology of VEGF-B and VEGF-C. Curr. Opin. Biotechnol. 10, 528–535 (1999).
Bellomo, D. et al. Mice lacking the vascular endothelial growth factor-B gene (Vegfb) have smaller hearts, dysfunctional coronary vasculature, and impaired recovery from cardiac ischemia. Circ. Res. 86, E29–E35 (2000).
Carmeliet, P. et al. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature 380, 435–439 (1996).
Ferrara, N. et al. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 380, 439– 442 (1996).
Carmeliet, P. et al. Impaired myocardial angiogenesis and ischemic cardiomyopathy in mice lacking the vascular endothelial growth factor isoforms VEGF164 and VEGF188. Nature Med. 5, 495– 502 (1999).
Miquerol, L., Langille, B. L. & Nagy, A. Embryonic development is disrupted by modest increases in vascular endothelial growth factor gene expression. Development 127, 3941–3946 ( 2000).
Gerber, H. P. et al. VEGF is required for growth and survival in neonatal mice . Development 126, 1149– 1159 (1999).
Ferrara, N. et al. Vascular endothelial growth factor is essential for corpus luteum angiogenesis. Nature Med. 4, 336– 340 (1998).
Gerber, H. P. et al. VEGF couples hypertrophic cartilage remodeling, ossification and angiogenesis during endochondral bone formation. Nature Med. 5, 623–628 ( 1999).
Stone, J. et al. Development of retinal vasculature is mediated by hypoxia-induced vascular endothelial growth factor (VEGF) expression by neuroglia. J. Neurosci. 15, 4738–4747 (1995).
Alon, T. et al. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity . Nature Med. 1, 1024–1028 (1995).
Benjamin, L. E., Hemo, I. & Keshet, E. A plasticity window for blood vessel remodelling is defined by pericyte coverage of the preformed endothelial network and is regulated by PDGF-B and VEGF. Development 125, 1591 –1598 (1998).
Stone, J. et al. Roles of vascular endothelial growth factor and astrocyte degeneration in the genesis of retinopathy of prematurity. Invest. Ophthalmol. Vis. Sci. 37, 290–299 ( 1996).
Pierce, E. A., Foley, E. D. & Smith, L. E. Regulation of vascular endothelial growth factor by oxygen in a model of retinopathy of prematurity. Arch. Ophthalmol . 114, 1219–1228 ( 1996). [Published erratum appears in Arch. Ophthalmol. 115, 427 (1997).]
Aiello, L. P. et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N. Engl. J. Med. 331, 1480–1487 ( 1994).
Adamis, A. P. et al. Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am. J. Ophthalmol. 118, 445–450 (1994).
Springer, M. L., Chen, A. S., Kraft, P. E., Bednarski, M. & Blau, H. M. VEGF gene delivery to muscle: potential role for vasculogenesis in adults. Mol. Cell 2, 549–558 (1998).
Detmar, M. et al. Increased microvascular density and enhanced leukocyte rolling and adhesion in the skin of VEGF transgenic mice. J. Invest. Dermatol. 111, 1–6 (1998 ).
Larcher, F., Murillas, R., Bolontrade, M., Conti, C. J. & Jorcano, J. L. VEGF/VPF overexpression in skin of transgenic mice induces angiogenesis, vascular hyperpermeability and accelerated tumor development. Oncogene 17, 303– 311 (1998).
Thurston, G. et al. Leakage-resistant blood vessels in mice transgenically overexpressing angiopoietin-1. Science 286, 2511– 2514 (1999).
Pettersson, A. et al. Heterogeneity of the angiogenic response induced in different normal adult tissues by vascular permeability factor/vascular endothelial growth factor. Lab. Invest. 80, 99– 115 (2000).
Thurston, G. et al. Angiopoietin-1 protects the adult vasculature against plasma leakage. Nature Med. 6, 460– 463 (2000).
Korhonen, J. et al. Enhanced expression of the tie receptor tyrosine kinase in endothelial cells during neovascularization. Blood 80, 2548–2555 (1992).
Maisonpierre, P. C., Goldfarb, M., Yancopoulos, G. D. & Gao, G. Distinct rat genes with related profiles of expression define a TIE receptor tyrosine kinase family. Oncogene 8, 1631 –1637. (1993).
Sato, T. N., Qin, Y., Kozak, C. A. & Audus, K. L. tie-1 and tie-2 define another class of putative receptor tyrosine kinase genes expressed in early embryonic vascular system. Proc. Natl Acad. Sci. USA 90, 9355–9358 (1993).
Dumont, D. J., Gradwohl, G. J., Fong, G.-H., Auerbach, R. & Breitman, M. L. The endothelial-specific receptor tyrosine kinase, tek, is a member of a new subfamily of receptors. Oncogene 8, 1293–1301 ( 1993).
Iwama, A. et al. Molecular cloning and characterization of mouse Tie and Tek receptor tyrosine kinase genes and their expression in hematopoietic stem cells. Biochem. Biophys. Res. Commun. 195, 301–309 (1993).
Davis, S. et al. Isolation of angiopoietin-1, a ligand for the TIE2 receptor, by secretion-trap expression cloning. Cell 87, 1161–1169 (1996).
Valenzuela, D. et al. Angiopoietins 3 and 4: diverging gene counterparts in mouse and man. Proc. Natl Acad. Sci. USA 96, 1904 –1909 (1999).
Dumont, D. J. et al. Dominant-negative and targeted null mutations in the endothelial receptor tyrosine kinase, tek, reveal a critical role in vasculogenesis of the embryo. Genes Dev. 8, 1897– 1909 (1994).
Sato, T. N. et al. Distinct roles of the receptor tyrosine kinases Tie-1 and Tie-2 in blood vessel formation. Nature 376, 70–74 (1995).
Suri, C. et al. Angiopoietin-1 promotes increased vascularization in vivo. Science 282, 468–471 ( 1998).
Goede, V., Schmidt, T., Kimmina, S., Kozian, D. & Augustin, H. G. Analysis of blood vessel maturation processes during cyclic ovarian angiogenesis. Lab. Invest. 78, 1385–1394 (1998).
Stratmann, A., Risau, W. & Plate, K. H. Cell type-specific expression of Angiopoietin-1 and Angiopoietin-2 suggests a role in glioblastoma angiogenesis. Am. J. Pathol. 153, 1459–1466 (1998).
Zagzag, D. et al. In Situ expression of angiopoietins in astrocytomas identifies angiopoietin-2 as an early marker of tumor angiogenesis. Exp. Neurol. 159, 391–400 ( 1999).
Flanagan, J. G. & Vanderhaeghen, P. The ephrins and Eph receptors in neural development. Annu. Rev. Neurosci. 21, 309–345 (1998).
Davis, S. et al. Ligands for the EPH-related receptor tyrosine kinases that require membrane attachment or clustering for activity. Science 266, 816–819 (1994).
Wang, H. U., Chen, Z. & Anderson, D. J. Molecular distinction and angiogenic interaction between embryonic arteries and veins revealed by ephrin-B2 and its receptor Eph-B4. Cell 93, 741–753 (1998).
Adams, R. H. et al. Roles for ephrinB ligands and EphB receptors in cardiovascular development: demarcation of arterial/venous domains, vascular morphogenesis, and sprouting angiogenesis. Genes Dev. 13, 295–306 (1999).
Gerety, S. S., Wang, H. U., Chen, Z. F. & Anderson, D. J. Symmetrical mutant phenotypes of the receptor EphB4 and its specific transmembrane ligand ephrin-B2 in cardiovascular development. Mol. Cell 4, 403–414 (1999).
Folkman, J. Tumor angiogenesis: therapeutic implications. N. Engl. J. Med. 285, 1182–1186 ( 1971).
Folkman, J. What is the evidence that tumors are angiogenesis dependent? J. Natl. Cancer Instit. 82, 4–6 (1990).
Hanahan, D. & Folkman, J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86, 353–364 (1996).
Wesseling, P., van der Laak, J. A., de Leeuw, H., Ruiter, D. J. & Burger, P. C. Quantitative immunohistological analysis of the microvasculature in untreated human glioblastome multiforme . J Neurosurg. 81, 902– 909 (1994).
Holmgren, L., O'Reilly, M. S. & Folkman, J. Dormancy of micrometastases: balanced proliferation and apoptosis in the presence of angiogenesis suppression. Nature Med. 1, 149–153 ( 1995).
Pezzella, F. et al. Non-small-cell lung carcinoma tumor growth without morphological evidence of neo-angiogenesis. Am. J. Pathol. 151, 1417–1423 (1997).
Kim, K. J. et al. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 362, 841–844 (1993).
Ferrara, N. & Alitalo, K. Clinical applications of angiogenic growth factors and their inhibitors. Nature Med. 5, 1359–1364 (1999).
Henry, T. D. et al. Results of intracoronary recombinant human vascular endothelial growth factor (RhVEGF) administration trial. J. Am. Coll. Cardiol. 31, 65A (1998).
Chiron Coorporation. Chiron announces preliminary findings from phase II clinical trial of FGF-2. 〈http://www.prnewswire.com/micro/CHIR 〉 Company Press Release 12 March 2000.
O'Reilly, M. S. et al. Angiostatin: a novel angiogenesis inhibitor that mediates the suppression of metastases by a Lewis Lung Carcinoma. Cell 79, 315–328 (1994).
O'Reilly, M. S. et al. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88, 277–285 (1997).
O'Reilly, M. S., Pirie-Shepherd, S., Lane, W. S. & Folkman, J. Antiangiogenic activity of the cleaved conformation of the serpin antithrombin . Science 285, 1926–1928 (1999).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yancopoulos, G., Davis, S., Gale, N. et al. Vascular-specific growth factors and blood vessel formation. Nature 407, 242–248 (2000). https://doi.org/10.1038/35025215
Issue Date:
DOI: https://doi.org/10.1038/35025215
This article is cited by
-
Effects of needle puncturing on re-vascularization and follicle survival in xenotransplanted human ovarian tissue
Reproductive Biology and Endocrinology (2023)
-
The long non-coding RNA NEAT1 promotes the progression of human ovarian cancer through targeting miR-214-3p and regulating angiogenesis
Journal of Ovarian Research (2023)
-
Suppression of angiopoietin-like 4 reprograms endothelial cell metabolism and inhibits angiogenesis
Nature Communications (2023)
-
FNIP1 abrogation promotes functional revascularization of ischemic skeletal muscle by driving macrophage recruitment
Nature Communications (2023)
-
Development of pro-angiogenic wound dressings from 2-deoxy-D-ribose (2dDR)-loaded decellularized plant leaves
Journal of Materials Science (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.