Abstract
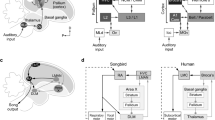
Hummingbirds have developed a wealth of intriguing features, such as backwards flight, ultraviolet vision, extremely high metabolic rates, nocturnal hibernation, high brain-to-body size ratio and a remarkable species–specific diversity of vocalizations1,2,3,4. Like humans, they have also developed the rare trait of vocal learning, this being the ability to acquire vocalizations through imitation rather than instinct5,6. Here we show, using behaviourally driven gene expression in freely ranging tropical animals, that the forebrain of hummingbirds contains seven discrete structures that are active during singing, providing the first anatomical and functional demonstration of vocal nuclei in hummingbirds. These structures are strikingly similar to seven forebrain regions that are involved in vocal learning and production in songbirds and parrots7,8,9,10,11,12,13—the only other avian orders known to be vocal learners5. This similarity is surprising, as songbirds, parrots and hummingbirds are thought to have evolved vocal learning and associated brain structures independently5,14, and it indicates that strong constraints may influence the evolution of forebrain vocal nuclei.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schuchmann, K.-L. in Handbook of the birds of the world: barn-owls to hummingbirds (eds del Hoyo, J., Elliott, A. & Sargatal, J.) 468– 680 (Lynx Ediciones, Barcelona, 1999).
Ventura, D. F. & Takase, E. Ultraviolet color discrimination in the hummingbird. Invest. Ophthalmol. Vis. Sci. 35 , 2168 (1994).
Rehkamper, G., Schuchmann, K. L., Schleicher, A. & Zilles, K. Encephalization in hummingbirds (Trochilidae). Brain Behav. Evol. 37, 85–91 ( 1991).
Vielliard, J. Catálogo sonográfico dos cantos e piados dos beija-flores do Brasil. Boletim do Museu de Biologia “Mello Leitão” Série Biologia 58, 1–20 (1983).
Nottebohm, F. The origins of vocal learning. Am. Nat. 106, 116–140 (1972).
Baptista, L. F. & Schuchmann, K. L. Song learning in the anna hummingbird (Calypte anna). Ethology 84, 15–26 (1990).
Nottebohm, F., Stokes, T. M. & Leonard, C. M. Central control of song in the canary, Serinus canarius. J. Comp. Neurol. 165, 457– 486 (1976).
Scharff, C. & Nottebohm, F. A comparative study of the behavioral deficits following lesions of various parts of the zebra finch song system: implications for vocal learning. J. Neurosci. 11, 2896–2913 (1991).
Yu, A. C. & Margoliash, D. Temporal hierarchical control of singing in birds. Science 273, 1871– 1875 (1996).
Hessler, N. A. & Doupe, A. J. Singing-related neural activity in a dorsal forebrain-basal ganglia circuit of adult zebra finches. J. Neurosci. 19, 10461– 10481 (1999).
Durand, S. E., Heaton, J. T., Amateau, S. K. & Brauth, S. E. Vocal control pathways through the anterior forebrain of a parrot (Melopsittacus undulatus). J. Comp. Neurol. 377, 179 –206 (1997).
Jarvis, E. D., Scharff, C., Grossman, M. R., Ramos, J. A. & Nottebohm, F. For whom the bird sings: context-dependent gene expression. Neuron 21, 775– 788 (1998).
Jarvis, E. D. & Mello, C. V. Molecular mapping of brain areas involved in parrot vocal communication. J. Comp. Neurol. 419, 1–31 (2000).
Brenowitz, E. A. Comparative approaches to the avian song system. J. Neurobiol. 33, 517–531 ( 1997).
Ruschi, A. Hummingbirds of State of Espirito Santo (Rios Editora, São Paulo, 1982).
Chaudhuri, A. Neural activity mapping with inducible transcription factors. Neuroreport 8, 5–9 ( 1997).
Mello, C. V. & Clayton, D. F. Song-induced ZENK gene expression in auditory pathways of songbird brain and its relation to the song control system. J. Neurosci. 14, 6652– 6666 (1994).
Jarvis, E. D., Schwabl, H., Ribeiro, S. & Mello, C. V. Brain gene regulation by territorial singing behavior in freely ranging songbirds. Neuroreport 8, 2073–2077 ( 1997).
Wild, J. M., Dongfeng, L. & Eagleton, C. Projections of the dorsomedial nucleus of the intercollicular complex (DM) in relation to respiratory-vocal nuclei in the brainstem of pigeon (Columbia livia) and zebra finch (Taeniopygia guttata). J. Comp. Neurol. 377, 392–413 (1997).
Nottebohm, F., Kelley, D. B. & Paton, J. A. Connections of vocal control nuclei in the canary telencephalon. J. Comp. Neurol. 207, 344 –357 (1982).
Lidsky, T. I., Manetto, C. & Schneider, J. S. A consideration of sensory factors involved in motor functions of the basal ganglia. Brain Res. 356, 133–146 (1985).
Guinee, L. H. & Payne, K. B. Rhyme-like repetitions in songs of humpback whales. Ethology 79, 295– 306 (1988).
Esser, K. H. Audio-vocal learning in a non-human mammal: the lesser spear-nosed bat Phyllostomus discolor. Neuroreport 5, 1718–1720 (1994).
Sibley, C. G. & Ahlquist, J. E. Phylogeny and Classification of Birds: A Study in Molecular Evolution (Yale Univ. Press, New Haven, 1990).
Kroodsma, D. E. & Konishi, M. A suboscine bird (eastern phoebe, Sayornis phoebe) develops normal song without auditory feedback. Anim. Behav. 42, 477– 487 (1991).
Feduccia, A. Explosive evolution in tertiary birds and mammals. Science 267, 637–638 (1995).
Koenig, C. Vocal patterns as interspecific isolating mechanisms in screech owls of the genus Otus (Aves: Strigidae) of southern South America. Stuttg. Beitr. Nat.kd. A Biol. 0(511), 1– 35 (1994).
Payne, R. B. in Current Ornithology (ed. Johnston, R. J.) 87–126 (Plenum, New York, 1986).
Miller, E. H. in Ecology and evolution of acoustic communication in birds (eds Kroodsma, D. E. & Miller, E. H.) 241–257 (Cornell Univ. Press, Ithaca, 1996).
Ellis, D. H., Swengel, S. R., Archibald, G. W. & Kepler, C. B. A sociogram for the cranes of the world. Behav. Processes 43, 125–151 (1998).
Acknowledgements
We thank the Museu de Biologia Mello Leitão (MBML) in Espírito Santo, Brazil, S. Mendes and D. Loss for providing a natural space and services that made this project possible. We also thank the MBML and A. Ruschi for permission to partially reproduce hummingbird illustrations by E. Demonte; P. Rousselot, K. S. Leon and A. Ferreira for help with recordings, sonograms and behavioural scoring; P. Delgado for histological assistance; S. Baumwell and L. Moore for help in manuscript preparation; S. Durand, R. Mooney and S. Nowicki for comments on the manuscript; L. Katz and N. Cant for use of microscope equipment; J. Ahlquist for discussions on avian evolution; C. Cunningham for assistance with phylogenetic analysis; and F. Nottebohm for his support. This project was approved by the Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis (IBAMA) and Conselho Nacional de Desenvolvimento Cientifico e Tecnológico (CNPq); funding was provided by the Kluge Trust Fund and Duke University start-up funds to E.D.J., an NIDCD grant to C.V.M., and personal funds by E.D.J. and C.V.M. We dedicate this paper to the memory of L. Baptista, a pioneer of vocal communication in hummingbirds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jarvis, E., Ribeiro, S., da Silva, M. et al. Behaviourally driven gene expression reveals song nuclei in hummingbird brain. Nature 406, 628–632 (2000). https://doi.org/10.1038/35020570
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35020570
This article is cited by
-
Analysis of vocal communication in the genus Falco
Scientific Reports (2023)
-
Absence of song suggests heterogeneity of vocal-production learning in hummingbirds
Journal of Ornithology (2023)
-
Call rate in Common Cuckoos does not predict body size and responses to conspecific playbacks
Journal of Ornithology (2021)
-
The hummingbird syrinx morphome: a detailed three-dimensional description of the black jacobin’s vocal organ
BMC Zoology (2020)
-
Molecular specializations of deep cortical layer analogs in songbirds
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.