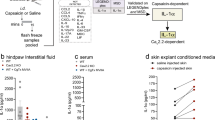
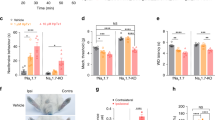
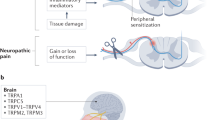
Abstract
The vanilloid receptor-1 (VR1) is a ligand-gated, non-selective cation channel expressed predominantly by sensory neurons. VR1 responds to noxious stimuli including capsaicin, the pungent component of chilli peppers, heat and extracellular acidification, and it is able to integrate simultaneous exposure to these stimuli1,2. These findings and research linking capsaicin with nociceptive behaviours (that is, responses to painful stimuli in animals3 have led to VR1 being considered as important for pain sensation. Here we have disrupted the mouse VR1 gene using standard gene targeting techniques. Small diameter dorsal root ganglion neurons isolated from VR1-null mice lacked many of the capsaicin-, acid- and heat-gated responses that have been previously well characterized in small diameter dorsal root ganglion neurons from various species. Furthermore, although the VR1-null mice appeared normal in a wide range of behavioural tests, including responses to acute noxious thermal stimuli, their ability to develop carrageenan-induced thermal hyperalgesia was completely absent. We conclude that VR1 is required for inflammatory sensitization to noxious thermal stimuli but also that alternative mechanisms are sufficient for normal sensation of noxious heat.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Caterina, M. J. et al. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389, 816– 824 (1997).
Tominaga, M. et al. The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron 21, 531– 543 (1998).
Szallasi, A. & Blumberg, P. M. Vanilloid (Capsaicin) receptors and mechanisms. Pharmacol. Rev. 51, 159– 211 (1999).
Bevan, S. et al. Capsazepine: A competitive antagonist of the sensory neurone excitant capsaicin. Br. J. Pharmacol. 107, 544–552 (1992).
Wood, J. N. et al. Capsaicin-induced ion fluxes in dorsal root ganglion cells in culture. J. Neurosci. 8, 3208– 3220 (1988).
Bevan, S. & Yeats, J. Protons activate a cation conductance in a sub-population of rat dorsal root ganglion neurones. J. Physiol. 433, 145–161 ( 1991).
Waldmann, R. et al. H+-gated cation channels. Ann. NY Acad. Sci. 868, 67–76 ( 1999).
Bevan, S. & Winter, J. Nerve growth factor (NGF) differentially regulates the chemosensitivity of adult rat cultured sensory neurones. J. Neurosci. 15, 4918–4926 (1995).
Ueno, S., Tsuda, M., Iwanaga, T. & Inoue, K. Cell type-specific ATP-activated responses in rat dorsal root ganglion neurones. Br. J. Pharmacol. 126, 429–436 (1999).
Zygmunt, P. M. et al. Vanilloid receptors on sensory nerves mediate the vasodilatory action of anandamide. Nature 400, 452– 457 (1999).
Cesare, P. & McNaughton, P. A novel heat-activated current in nociceptive neurons and its sensitization by bradykinin. Proc. Natl Acad. Sci. USA 93, 15435–15439 (1996).
Nagy, I. & Rang, H. P. Similarities and differences between the responses of rat sensory neurons to noxious heat and capsaicin. J. Neurosci. 19, 10647–10655 (1999).
Nagy, J. I. & Kooy, D. van der. Effects of neonatal capsaicin treatment on nociceptive thresholds in the rat. J. Neurosci. 3, 1145–1150 (1983).
Caterina, M. J., Rosen, T. A., Tominaga, M., Brake, A. J. & Julius, D. A capsaicin-receptor homologue with a high threshold for noxious heat. Nature 398, 436–441 (1999).
Hargreaves, K., Dubner, R., Brown, F., Flores, C. & Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 32, 77– 88 (1988).
Kwak, J. Y., Jung, J. Y., Hwang, S. W., Lee, W. T. & Oh, U. A capsaicin-receptor antagonist, capsazepine, reduces inflammation-induced hyperalgesic responses in the rat: evidence for an endogenous capsaicin-like substance. Neuroscience 86, 619–626 (1998).
Sasamura, T., Sasaki, M., Tohda, C. & Kuraishi, Y. Existence of capsaicin-sensitive glutamatergic terminals in rat hypothalamus. Neuroreport 9, 2045–2048 (1998).
Rogers, D. C. et al. ‘SHIRPA’—a comprehensive behavioural and functional analysis of mouse phenotype. Mamm. Genome 8, 711–713 (1997).
Yagi, T. et al. A novel selection for homologous recombinants using diptheria toxin A frgament gene. Anal. Biochem. 214, 77–86 (1993).
Torres, R. M. & Kuhn, R. Laboratory Protocols for Conditional Gene Targeting. (Oxford Univ. Press, Oxford, 1997).
Hooper, M., Hardy, K., Handyside, A., Hunter, S. & Monk, M. HPRT-deficient (Lesch-Nyhan) mouse embryos derived from germline colonization by cultured cells. Nature 326 , 292–295 (1987).
Rogers, D. C. et al. Use of SHIRPA and discriminant analysis to characterise marked differences in the behavioural phenotype of six inbred mouse strains. Behav. Brain Res. 105, 207–217 (1999).
Miliken, G. A. & Johnson, D. E. in Analysis of Messy Data 29–45 (Chapman & Hall, London, 1992).
Acknowledgements
The authors would like to acknowledge P. Hayes, J. Nation, S. Pickering and C. David for technical assistance, and S. Rastan, F. Walsh, M. Geppert and D. Simmons for valuable critique.
Author information
Authors and Affiliations
Supplementary information
41586_2000_BF35012076_MOESM1_ESM.doc
Methods for Targeting of VR1 gene and generation of mutant mice & table 1: Summary of SHIRPA primary screening results. (DOC 35 kb)
Rights and permissions
About this article
Cite this article
Davis, J., Gray, J., Gunthorpe, M. et al. Vanilloid receptor-1 is essential for inflammatory thermal hyperalgesia . Nature 405, 183–187 (2000). https://doi.org/10.1038/35012076
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35012076
This article is cited by
-
Structure-guided peptide engineering of a positive allosteric modulator targeting the outer pore of TRPV1 for long-lasting analgesia
Nature Communications (2023)
-
TRP (transient receptor potential) ion channel family: structures, biological functions and therapeutic interventions for diseases
Signal Transduction and Targeted Therapy (2023)
-
The role of protein kinases in diabetic neuropathic pain: an update review
Journal of Diabetes & Metabolic Disorders (2023)
-
N-Oleoyl dopamine induces IL-10 via central nervous system TRPV1 and improves endotoxemia and sepsis outcomes
Journal of Neuroinflammation (2022)
-
The industrial solvent 1,4-dioxane causes hyperalgesia by targeting capsaicin receptor TRPV1
BMC Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.