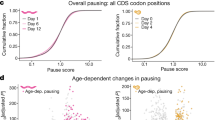
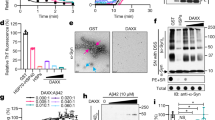
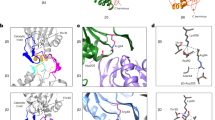
Abstract
THE N-end rule, a code that relates the metabolic stability of a protein to the identity of its ammo-terminal residue1, is universal in that different versions of the N-end rule operate in mammals2–5, yeast1,6,7 and bacteria (unpublished data). The N-end rule-based degradation signal comprises a destabilizing amino-terminal residue and a specific internal lysine residue1,6,7. We now show that, in a multisubunit protein, these two determinants can be located on different subunits and still target the protein for destruction. Moreover, in this case (trans recognition) only the subunit that bears the lysine determinant is actually degraded. Thus an oligomeric protein can contain both short-lived and long-lived subunits. These insights have functional and practical implications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bachmair, A., Finley, D. & Varshavsky, A. Science 234, 179–186 (1986).
Varshavsky, A. et al. in Ubiquitin (ed. Rechsteiner, M.) 287–324 (Plenum, New York, 1988).
Townsend, A. et al. J. exp. Med. 168, 1211–1224 (1988).
Reiss, Y., Kaim, D. & Hershko, A. J. biol. Chem. 263, 2693–2698 (1988).
Gonda, D. K. et al. J. biol. Chem. 264, 16700–16712 (1989).
Bachmair, A. & Varshavsky, A. Cell 56, 1019–1032 (1989).
Chau, V. et al. Science 243, 1576–1583 (1989).
Hershko, A. J. biol. Chem. 263, 15237–15240 (1988).
Zipser, D. J. molec. Biol. 7, 113–121 (1963).
Shifrin, S. & Steers, E. Jr Biochim. biophys. Acta 133, 463–471 (1967).
Givol, D., Craven, G. R., Steers, E. & Anfinsen, C. B. Biochim. biophys. Acta 113, 120–125 (1966).
Schimke, R. T. Adv. Enzymol. 37, 135–187 (1973).
Goldberg, A. K. & Dice, J. F. A. Rev. Biochem. 43, 835–869 (1974).
Hare, J. F. & Hodges, R. J. biol. Chem. 257, 3575–3580 (1982).
Evans, T. et al. Cell 33, 389–396 (1983).
Murray, A. W., Solomon, M. J. & Kirschner, M. W. Nature 339, 280–286 (1989).
Abel, T. & Maniatis, T. Nature 341, 24–25 (1989).
Goutte, C. & Johnson, A. D. Cell 52, 875–882 (1988).
Ingham, P. W. Nature 335, 25–34 (1988).
Scott, M. P. & Carroll, S. B. Cell 51, 689–698 (1987).
Hochstrasser, M. & Varshavsky, A. Cell 61, 697–708 (1990).
Ausubel, F. M. et al. Current Protocols in Molecular Biology (Wiley, New York, 1987).
Ullman, A. & Monod, J. Biochem. biophys. Res. Commun. 35, 35–42 (1969).
Guarente, L. Meth. Enzym. 101, 181–183 (1983).
Ferber, S. & Ciechanover, A. Nature 326, 808–811 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Johnson , E., Gonda, D. & Varshavsky, A. Cis-trans recognition and subunit-specific degradation of short-lived proteins. Nature 346, 287–291 (1990). https://doi.org/10.1038/346287a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/346287a0
This article is cited by
-
Phenotypes on demand via switchable target protein degradation in multicellular organisms
Nature Communications (2016)
-
Regulated protein turnover: snapshots of the proteasome in action
Nature Reviews Molecular Cell Biology (2014)
-
Defining the geometry of the two-component proteasome degron
Nature Chemical Biology (2011)
-
Substrate selection by the proteasome during degradation of protein complexes
Nature Chemical Biology (2009)
-
Proteasomes and their kin: proteases in the machine age
Nature Reviews Molecular Cell Biology (2004)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.