Abstract
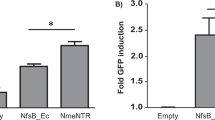
Escherichia coli nitroreductase (NTR) converts the prodrug CB1954 (5-(aziridin-1-yl)-2,4-dinitrobenzamide) into a bifunctional alkylating agent that causes DNA crosslinks. In this study, the ability of NTR to enhance the combined effects of CB1954 and radiation has been tested in vitro and in vivo. Stably transduced ovarian cancer cells (SKOV3-NTR) that are sensitive to CB1954 (IC50=0.35 μM) demonstrated enhanced cytotoxicity when treated with CB1954 and single-fraction irradiation. The NTR–CB1954 system mediated a bystander effect in combination with radiation on transfer of conditioned medium from SKOV3-NTR, but not SKOV3, cells to SW480 target cells. The ability of CB1954 to enhance radiation-induced cytotoxicity in SKOV3-NTR (but not SKOV3) cells was also demonstrated by fluorescence-activated cell sorting (FACS) with dual staining for propidium iodide/fluorescein diacetate, 4′,6-diamidino-2-phenylindole dichloride staining of apoptotic cells and measurement of double-stranded DNA breaks by FACS and confocal microscopy for γH2AX foci. Adenoviral delivery of NTR, under constitutive cytomegalovirus or tissue-specific CTP1 promoters, increased the in vitro cytotoxicity of CB1954 plus radiation in MTT and clonogenic assays. Finally, adenoviral delivery of NTR plus CB1954 enhanced the effect of fractionated radiotherapy (12 Gy in four fractions) in SW480 xenograft tumours in nude mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Harrington KJ, Melcher AA, Bateman AR, Ahmed A, Vile RG . Cancer gene therapy: part 2. Candidate transgenes and their clinical development. Clin Oncol (R Coll Radiol) 2002; 14: 148–169.
Relph K, Harrington K, Pandha H . Recent developments and current status of gene therapy using viral vectors in the United Kingdom. BMJ 2004; 329: 839–842.
Harrington KJ, Nutting CM . Interactions between ionising radiation and drugs in head and neck cancer—how can we maximize the therapeutic index? Curr Opin Investig Drugs 2002; 3: 807–811.
Cobb LM, Connors TA, Elson LA, Khan AH, Mitchley BC, Ross WC et al. 2,4-dinitro-5-ethyleneiminobenzamide (CB1954): a potent and selective inhibitor of growth of the Walker carcinoma 256. Biochem Pharmacol 1969; 18: 1519–1527.
Boland MP, Knox RJ, Roberts JJ . The differences in kinetics of rat and human DT diaphorase result in a differential sensitivity of derived cell lines to CB1954 (5-(aziridin-1-yl)-2,4-dinitrobenzamide). Biochem Pharmacol 1991; 41: 867–875.
Roberts JJ, Friedlos F, Knox RJ . CB 1954 (2,4-dinitro-5-aziridinyl benzamide) becomes a DNA interstrand crosslinking agent in Walker tumour cells. Biochem Biophys Res Commun 1986; 140: 1073–1078.
Anlezark GM, Melton RG, Sherwood RF, Coles B, Friedlos F, Knox RJ . The bioactivation of 5-(aziridin-1-yl)-2,4-dinitrobenzamide (CB 1954)—I. Purification and properties of a nitroreductase enzyme from Escherichia coli—a potential for antibody-directed enzyme prodrug therapy (ADEPT). Biochem Pharmacol 1992; 44: 2289–2295.
Walling JM, Stratford IJ, Adams GE . Radiosensitization by the 2,4-dinitro-5-aziridinyl benzamide CB 1954: a structure/activity study. Int J Radiat Biol Relat Stud Phys Chem Med 1987; 52: 31–41.
Hennequin C, Favaudon V . Biological basis for chemo–radiotherapy interactions. Eur J Cancer 2002; 38: 223–230.
Lipinski KS, Djeha AH, Ismail T, Mountain A, Young LS, Wrighton CJ . High-level, beta-catenin/TCF-dependent transgene expression in secondary colorectal cancer tissue. Mol Ther 2001; 4: 365–371.
Valerie K, Brust D, Farnsworth J, Amir C, Taher MM, Hershey C et al. Improved radiosensitization of rat glioma cells with adenovirus-expressed mutant herpes simplex virus-thymidine kinase in combination with acyclovir. Cancer Gene Ther 2000; 7: 879–884.
Anello R, Cohen S, Atkinson G, Hall SJ . Adenovirus-mediated cytosine deaminase gene transduction and 5-fluorocytosine therapy sensitises mouse prostate cancer cells to irradiation. J Urol 2000; 164: 2173–2177.
Kievit E, Nyati MK, Ng E, Stegman LD, Parsels J, Ross BD et al. Yeast cytosine deaminase improves radiosensitization and bystander effect by 5-fluorocytosine of human colorectal cancer xenografts. Cancer Res 2000; 60: 6649–6655.
Stackhouse MA, Pederson LC, Grizzle WE, Curiel DT, Gebert J, Haack K et al. Fractionated radiation therapy in combination with adenoviral delivery of the cytosine deaminase gene and 5-fluorocytosine enhances cytotoxic and antitumor effects in human colorectal and cholangiocarcinoma models. Gene Ther 2000; 7: 1019–1026.
Vlachaki MT, Chhikara M, Aguilar L, Zhu X, Chiu KJ, Woo S et al. Enhanced therapeutic effect of multiple injections of HSV-TK+GCV gene therapy in combination with ionizing radiation in a mouse mammary tumor model. Int J Radiat Oncol Biol Phys 2001; 51: 1008–1017.
Rosenberg E, Hawkins W, Holmes M, Amir C, Schmidt-Ullrich RK, Lin PS et al. Radiosensitization of human glioma cells in vitro and in vivo with acyclovir and mutant HSV-TK75 expressed from adenovirus. Int J Radiat Oncol Biol Phys 2002; 52: 831–836.
Freytag SO, Paielli D, Wing M, Rogulski K, Brown S, Kolozsvary A et al. Efficacy and toxicity of replication-competent adenovirus-mediated double suicide gene therapy in combination with radiation therapy in an orthotopic mouse prostate cancer model. Int J Radiat Oncol Biol Phys 2002; 54: 873–885.
Freytag SO, Khil M, Stricker H, Peabody J, Menon M, DePeralta-Venturina M et al. Phase I study of replication-competent adenovirus-mediated double suicide gene therapy for the treatment of locally recurrent prostate cancer. Cancer Res 2002; 62: 4968–4976.
Kambara H, Tamiya T, Ono Y, Ohtsuka S, Terada K, Adachi Y et al. Combined radiation and gene therapy for brain tumors with adenovirus-mediated transfer of cytosine deaminase and uracil phosphoribosyltransferase genes. Cancer Gene Ther 2002; 9: 840–845.
Chung SM, Advani SJ, Bradley JD, Kataoka Y, Vashistha K, Yan SY et al. The use of a genetically engineered herpes simplex virus (R7020) with ionizing radiation for experimental hepatoma. Gene Ther 2002; 9: 75–80.
Chung-Faye G, Palmer D, Anderson D, Clark J, Downes M, Baddeley J et al. Virus-directed, enzyme prodrug therapy with nitroimidazole reductase: a phase I and pharmacokinetic study of its prodrug, CB1954. Clin Cancer Res 2001; 7: 2662–2668.
Palmer DH, Mautner V, Mirza D, Oliff S, Gerritsen W, van der Sijp JR et al. Virus-directed enzyme prodrug therapy: intratumoral administration of a replication-deficient adenovirus encoding nitroreductase to patients with resectable liver cancer. J Clin Oncol 2004; 22: 1546–1552.
Schepelmann S, Hallenbeck P, Ogilvie LM, Hedley D, Friedlos F, Martin J et al. Systemic gene-directed enzyme prodrug therapy of hepatocellular carcinoma using a targeted adenovirus armed with carboxypeptidase G2. Cancer Res 2005; 65: 5003–5008.
Mayer A, Francis RJ, Sharma SK, Tolner B, Springer CJ, Martin J et al. A phase I study of single administration of antibody-directed enzyme prodrug therapy with the recombinant anti-carcinoembryonic antigen antibody fusion protein MFECP1 and a bis-iodo phenol mustard prodrug. Clin Cancer Res 2006; 12: 6509–6516.
Harrington KJ, Rowlinson-Busza G, Syrigos KN, Peters AM, Uster PS, Vile RG et al. Pegylated liposome encapsulated doxorubicin and cisplatin enhance the effect of radiotherapy in a tumor xenograft model. Clin Cancer Res 2000; 6: 4939–4949.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
White, C., Menghistu, T., Twigger, K. et al. Escherichia coli nitroreductase plus CB1954 enhances the effect of radiotherapy in vitro and in vivo. Gene Ther 15, 424–433 (2008). https://doi.org/10.1038/sj.gt.3303081
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3303081
Keywords
This article is cited by
-
Asymmetric ligand binding in homodimeric Enterobacter cloacae nitroreductase yields the Michaelis complex for nitroaromatic substrates
Journal of Molecular Modeling (2020)
-
Sensitive Fibre-Based Thermoluminescence Detectors for High Resolution In-Vivo Dosimetry
Scientific Reports (2015)
-
Adenovirus-mediated hypoxia-targeted gene therapy using HSV thymidine kinase and bacterial nitroreductase prodrug-activating genes in vitro and in vivo
Cancer Gene Therapy (2011)