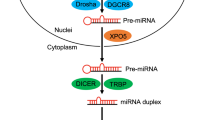
Abstract
MicroRNAs (miRNAs) are endogenous 19–25 nucleotide RNAs that have recently emerged as a novel class of important gene-regulatory molecules involved in many critical developmental and cellular functions. miRNAs have been implicated in the pathogenesis of several human diseases, such as neurodegenerative disorders, cancer, and more recently in viral and metabolic diseases. Unraveling the roles of miRNAs in cellular processes linked to human diseases will lead to novel opportunities for the regulation of protein function and will help to evaluate their potential for therapeutic intervention. Approaches to interfere with miRNA function in vitro and in vivo based on synthetic anti-miRNA oligonucleotides (AMOs) are discussed in this review.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ambros V . The functions of animal microRNAs. Nature 2004; 431: 350–355.
Bartel DP . MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004; 116: 281–297.
Filipowicz W, Jaskiewicz L, Kolb FA, Pillai RS . Post-transcriptional gene silencing by siRNAs and miRNAs. Curr Opin Struct Biol 2005; 15: 331–341.
Kim VN . MicroRNA biogenesis: coordinated cropping and dicing. Nat Rev Mol Cell Biol 2005; 6: 376–385.
Lee Y, Jeon K, Lee JT, Kim S, Kim VN . MicroRNA maturation: stepwise processing and subcellular localization. EMBO J 2002; 21: 4663–4670.
Cai X, Hagedorn CH, Cullen BR . Human microRNAs are processed from capped, polyadenylated transcripts that can also function as mRNAs. RNA 2004; 10: 1957–1966.
Lee Y, Kim M, Han J, Yeom KH, Lee S, Baek SH et al. MicroRNA genes are transcribed by RNA polymerase II. EMBO J 2004; 23: 4051–4060.
Lee Y, Ahn C, Han J, Choi H, Kim J, Yim J et al. The nuclear RNase III Drosha initiates microRNA processing. Nature 2003; 425: 415–419.
Yi R, Qin Y, Macara IG, Cullen BR . Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs. Genes Dev 2003; 17: 3011–3016.
Lund E, Guttinger S, Calado A, Dahlberg JE, Kutay U . Nuclear export of microRNA precursors. Science 2004; 303: 95–98.
Bernstein E, Caudy AA, Hammond SM, Hannon GJ . Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature 2001; 409: 363–366.
Hutvagner G, McLachlan J, Pasquinelli AE, Balint E, Tuschl T, Zamore PD . A cellular function for the RNA-interference enzyme Dicer in the maturation of the let-7 small temporal RNA. Science 2001; 293: 834–838.
Hutvagner G, Zamore PD . A microRNA in a multiple-turnover RNAi enzyme complex. Science 2002; 297: 2056–2060.
Martinez J, Patkaniowska A, Urlaub H, Luhrmann R, Tuschl T . Single-stranded antisense siRNAs guide target RNA cleavage in RNAi. Cell 2002; 110: 563–574.
Tang G . siRNA and miRNA: an insight into RISCs. Trends Biochem Sci 2005; 30: 106–114.
Yekta S, Shih IH, Bartel DP . MicroRNA-directed cleavage of HOXB8 mRNA. Science 2004; 304: 594–596.
Griffiths-Jones S . The microRNA Registry. Nucleic Acids Res 2004; 32: D109–D111.
Brennecke J, Hipfner DR, Stark A, Russell RB, Cohen SM . bantam encodes a developmentally regulated microRNA that controls cell proliferation and regulates the proapoptotic gene hid in Drosophila. Cell 2003; 113: 25–36.
Chen CZ, Li L, Lodish HF, Bartel DP . MicroRNAs modulate hematopoietic lineage differentiation. Science 2004; 303: 83–86.
Esau C, Kang X, Peralta E, Hanson E, Marcusson EG, Ravichandran LV et al. MicroRNA-143 regulates adipocyte differentiation. J Biol Chem 2004; 279: 52361–52365.
Poy MN, Eliasson L, Krutzfeldt J, Kuwajima S, Ma X, Macdonald PE et al. A pancreatic islet-specific microRNA regulates insulin secretion. Nature 2004; 432: 226–230.
Gregory RI, Shiekhattar R . MicroRNA biogenesis and cancer. Cancer Res 2005; 65: 3509–3512.
Dostie J, Mourelatos Z, Yang M, Sharma A, Dreyfuss G . Numerous microRNPs in neuronal cells containing novel microRNAs. Rna 2003; 9: 180–186.
Pfeffer S, Zavolan M, Grasser FA, Chien M, Russo JJ, Ju J et al. Identification of virus-encoded microRNAs. Science 2004; 304: 734–736.
McManus MT . MicroRNAs and cancer. Semin Cancer Biol 2003; 13: 253–258.
Gong H, Liu CM, Liu DP, Liang CC . The role of small RNAs in human diseases: potential troublemaker and therapeutic tools. Med Res Rev 2005; 25: 361–381.
Calin GA, Sevignani C, Dumitru CD, Hyslop T, Noch E, Yendamuri S et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci USA 2004; 101: 2999–3004.
Calin GA, Dumitru CD, Shimizu M, Bichi R, Zupo S, Noch E et al. Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci USA 2002; 99: 15524–15529.
Stilgenbauer S, Nickolenko J, Wilhelm J, Wolf S, Weitz S, Dohner K et al. Expressed sequences as candidates for a novel tumor suppressor gene at band 13q14 in B-cell chronic lymphocytic leukemia and mantle cell lymphoma. Oncogene 1998; 16: 1891–1897.
Migliazza A, Cayanis E, Bosch-Albareda F, Komatsu H, Martinotti S, Toniato E et al. Molecular pathogenesis of B-cell chronic lymphocytic leukemia: analysis of 13q14 chromosomal deletions. Curr Top Microbiol Immunol 2000; 252: 275–284.
Michael MZ, O'Connor SM, van Holst Pellekaan NG, Young GP, James RJ . Reduced accumulation of specific microRNAs in colorectal neoplasia. Mol Cancer Res 2003; 1: 882–891.
Johnson SM, Grosshans H, Shingara J, Byrom M, Jarvis R, Cheng A et al. RAS is regulated by the let-7 microRNA family. Cell 2005; 120: 635–647.
Takamizawa J, Konishi H, Yanagisawa K, Tomida S, Osada H, Endoh H et al. Reduced expression of the let-7 microRNAs in human lung cancers in association with shortened postoperative survival. Cancer Res 2004; 64: 3753–3756.
Metzler M, Wilda M, Busch K, Viehmann S, Borkhardt A . High expression of precursor microRNA-155/BIC RNA in children with Burkitt lymphoma. Genes Chromosomes Cancer 2004; 39: 167–169.
Tam W, Hughes SH, Hayward WS, Besmer P . Avian bic, a gene isolated from a common retroviral site in avian leukosis virus-induced lymphomas that encodes a noncoding RNA, cooperates with c-myc in lymphomagenesis and erythroleukemogenesis. J Virol 2002; 76: 4275–4286.
O'Donnell KA, Wentzel EA, Zeller KI, Dang CV, Mendell JT . c-Myc-regulated microRNAs modulate E2F1 expression. Nature 2005; 435: 839–843.
He L, Thomson JM, Hemann MT, Hernando-Monge E, Mu D, Goodson S et al. A microRNA polycistron as a potential human oncogene. Nature 2005; 435: 828–833.
Cai X, Lu S, Zhang Z, Gonzalez CM, Damania B, Cullen BR . Kaposi's sarcoma-associated herpesvirus expresses an array of viral microRNAs in latently infected cells. Proc Natl Acad Sci USA 2005; 102: 5570–5575.
Pfeffer S, Sewer A, Lagos-Quintana M, Sheridan R, Sander C, Grasser FA et al. Identification of microRNAs of the herpesvirus family. Nat Methods 2005; 2: 269–276.
Omoto S, Fujii YR . Regulation of human immunodeficiency virus 1 transcription by nef microRNA. J Gen Virol 2005; 86: 751–755.
Sullivan CS, Grundhoff AT, Tevethia S, Pipas JM, Ganem D . SV40-encoded microRNAs regulate viral gene expression and reduce susceptibility to cytotoxic T cells. Nature 2005; 435: 682–686.
Omoto S, Ito M, Tsutsumi Y, Ichikawa Y, Okuyama H, Brisibe EA et al. HIV-1 nef suppression by virally encoded microRNA. Retrovirology 2004; 1: 44.
Couturier JP, Root-Bernstein RS . HIV may produce inhibitory microRNAs (miRNAs) that block production of CD28, CD4 and some interleukins. J Theor Biol 2005; 235: 169–184.
Meister G, Tuschl T . Mechanisms of gene silencing by double-stranded RNA. Nature 2004; 431: 343–349.
Lecellier CH, Dunoyer P, Arar K, Lehmann-Che J, Eyquem S, Himber C et al. A cellular microRNA mediates antiviral defense in human cells. Science 2005; 308: 557–560.
Lu S, Cullen BR . Adenovirus VA1 noncoding RNA can inhibit small interfering RNA and MicroRNA biogenesis. J Virol 2004; 78: 12868–12876.
Famulok M, Mayer G . Intramers and aptamers: applications in protein-function analyses and potential for drug screening. Chembiochem 2005; 6: 19–26.
Jin P, Alisch RS, Warren ST . RNA and microRNAs in fragile X mental retardation. Nat Cell Biol 2004; 6: 1048–1053.
Caudy AA, Myers M, Hannon GJ, Hammond SM . Fragile X-related protein and VIG associate with the RNA interference machinery. Genes Dev 2002; 16: 2491–2496.
Ishizuka A, Siomi MC, Siomi H . A Drosophila fragile X protein interacts with components of RNAi and ribosomal proteins. Genes Dev 2002; 16: 2497–2508.
Jin P, Zarnescu DC, Ceman S, Nakamoto M, Mowrey J, Jongens TA et al. Biochemical and genetic interaction between the fragile X mental retardation protein and the microRNA pathway. Nat Neurosci 2004; 7: 113–117.
Mourelatos Z, Dostie J, Paushkin S, Sharma A, Charroux B, Abel L et al. miRNPs: a novel class of ribonucleoproteins containing numerous microRNAs. Genes Dev 2002; 16: 720–728.
Gregory RI, Yan KP, Amuthan G, Chendrimada T, Doratotaj B, Cooch N et al. The Microprocessor complex mediates the genesis of microRNAs. Nature 2004; 432: 235–240.
Landthaler M, Yalcin A, Tuschl T . The human DiGeorge syndrome critical region gene 8 and its D. melanogaster homolog are required for miRNA biogenesis. Curr Biol 2004; 14: 2162–2167.
Iwai N, Naraba H . Polymorphisms in human pre-miRNAs. Biochem Biophys Res Commun 2005; 331: 1439–1444.
Zeng Y, Yi R, Cullen BR . MicroRNAs and small interfering RNAs can inhibit mRNA expression by similar mechanisms. Proc Natl Acad Sci USA 2003; 100: 9779–9784.
Ganju P, Hall J . Potential applications of siRNA for pain therapy. Expert Opin Biol Ther 2004; 4: 531–542.
Lee YS, Kim HK, Chung S, Kim KS, Dutta A . Depletion of human micro-RNA miR-125b reveals that it is critical for the proliferation of differentiated cells but not for the down-regulation of putative targets during differentiation. J Biol Chem 2005; 280: 16635–16641.
Boutla A, Delidakis C, Tabler M . Developmental defects by antisense-mediated inactivation of micro-RNAs 2 and 13 in Drosophila and the identification of putative target genes. Nucleic Acids Res 2003; 31: 4973–4980.
Hutvagner G, Simard MJ, Mello CC, Zamore PD . Sequence-specific inhibition of small RNA function. PLoS Biol 2004; 2: E98.
Devi GR, Beer TM, Corless CL, Arora V, Weller DL, Iversen PL . In vivo bioavailability and pharmacokinetics of a c-MYC antisense phosphorodiamidate morpholino oligomer, AVI-4126, in solid tumors. Clin Cancer Res 2005; 11: 3930–3938.
Henry SP, Geary RS, Yu R, Levin AA . Drug properties of second-generation antisense oligonucleotides: how do they measure up to their predecessors? Curr Opin Invest Drugs 2001; 2: 1444–1449.
Friedman KJ, Kole J, Cohn JA, Knowles MR, Silverman LM, Kole R . Correction of aberrant splicing of the cystic fibrosis transmembrane conductance regulator (CFTR) gene by antisense oligonucleotides. J Biol Chem 1999; 274: 36193–36199.
Mani S, Goel S, Nesterova M, Martin RM, Grindel JM, Rothenberg ML et al. Clinical studies in patients with solid tumors using a second-generation antisense oligonucleotide (GEM 231) targeted against protein kinase A type I. Ann N Y Acad Sci 2003; 1002: 252–262.
Cheng AM, Byrom MW, Shelton J, Ford LP . Antisense inhibition of human miRNAs and indications for an involvement of miRNA in cell growth and apoptosis. Nucleic Acids Res 2005; 33: 1290–1297.
Martin P . A New Access to 2-O-Alkylated Ribonucleosides and Properties of 2-O-Alkylated Oligoribonucleotides. Helvetica Chim Acta 1995; 78: 486–504.
Karras JG, McKay RA, Dean NM, Monia BP . Deletion of individual exons and induction of soluble murine interleukin-5 receptor-alpha chain expression through antisense oligonucleotide-mediated redirection of pre-mRNA splicing. Mol Pharmacol 2000; 58: 380–387.
Baker BF, Lot SS, Condon TP, Cheng-Flournoy S, Lesnik EA, Sasmor HM et al. 2′-O-(2-Methoxy)ethyl-modified anti-intercellular adhesion molecule 1 (ICAM-1) oligonucleotides selectively increase the ICAM-1 mRNA level and inhibit formation of the ICAM-1 translation initiation complex in human umbilical vein endothelial cells. J Biol Chem 1997; 272: 11994–12000.
Gleave ME, Monia BP . Antisense therapy for cancer. Nat Rev Cancer 2005; 5: 468–479.
Vester B, Wengel J . LNA (locked nucleic acid): high-affinity targeting of complementary RNA and DNA. Biochemistry 2004; 43: 13233–13241.
Childs JL, Disney MD, Turner DH . Oligonucleotide directed misfolding of RNA inhibits Candida albicans group I intron splicing. Proc Natl Acad Sci USA 2002; 99: 11091–11096.
Elayadi AN, Braasch DA, Corey DR . Implications of high-affinity hybridization by locked nucleic acid oligomers for inhibition of human telomerase. Biochemistry 2002; 41: 9973–9981.
Mouritzen P, Nielsen AT, Pfundheller HM, Choleva Y, Kongsbak L, Moller S . Single nucleotide polymorphism genotyping using locked nucleic acid (LNA). Expert Rev Mol Diagn 2003; 3: 27–38.
Fluiter K, ten Asbroek AL, de Wissel MB, Jakobs ME, Wissenbach M, Olsson H et al. In vivo tumor growth inhibition and biodistribution studies of locked nucleic acid (LNA) antisense oligonucleotides. Nucleic Acids Res 2003; 31: 953–962.
Valoczi A, Hornyik C, Varga N, Burgyan J, Kauppinen S, Havelda Z . Sensitive and specific detection of microRNAs by northern blot analysis using LNA-modified oligonucleotide probes. Nucleic Acids Res 2004; 32: e175.
Chan JA, Krichevsky AM, Kosik KS . MicroRNA-21 is an antiapoptotic factor in human glioblastoma cells. Cancer Res 2005; 65: 6029–6033.
Lim LP, Lau NC, Garrett-Engele P, Grimson A, Schelter JM, Castle J et al. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature 2005; 433: 769–773.
Robins H, Li Y, Padgett RW . Incorporating structure to predict microRNA targets. Proc Natl Acad Sci USA 2005; 102: 4006–4009.
John B, Enright AJ, Aravin A, Tuschl T, Sander C, Marks DS . Human MicroRNA targets. PLoS Biol 2004; 2: e363.
Krek A, Grun D, Poy MN, Wolf R, Rosenberg L, Epstein EJ et al. Combinatorial microRNA target predictions. Nat Genet 2005; 37: 495–500.
Lewis BP, Burge CB, Bartel DP . Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 2005; 120: 15–20.
Esquela-Kerscher A, Slack FJ . The age of high-throughput microRNA profiling. Nat Methods 2004; 1: 106–107.
Lu J, Getz G, Miska EA, Alvarez-Saavedra E, Lamb J, Peck D et al. MicroRNA expression profiles classify human cancers. Nature 2005; 435: 834–838.
Beal J . Silence is golden: can RNA interference therapeutics deliver? Drug Discov Today 2005; 10: 169–172.
de Fougerolles A, Manoharan M, Meyers R, Vornlocher HP . RNA interference in vivo: toward synthetic small inhibitory RNA-based therapeutics. Methods Enzymol 2005; 392: 278–296.
Acknowledgements
We would like to thank Iwan Beuvink for critically reading this manuscript and for valuable comments. We apologize to our colleagues whose outstanding contributions to the growing miRNA field were not cited as primary references only through space constraints.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weiler, J., Hunziker, J. & Hall, J. Anti-miRNA oligonucleotides (AMOs): ammunition to target miRNAs implicated in human disease?. Gene Ther 13, 496–502 (2006). https://doi.org/10.1038/sj.gt.3302654
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302654
Keywords
This article is cited by
-
MicroRNAs as the critical regulators of cisplatin resistance in gastric tumor cells
Genes and Environment (2021)
-
Biogenesis and function of extracellular miRNAs
ExRNA (2019)
-
Inhibition of microRNA-711 limits angiopoietin-1 and Akt changes, tissue damage, and motor dysfunction after contusive spinal cord injury in mice
Cell Death & Disease (2019)
-
MicroRNAs and Long Non-coding RNAs in Genetic Diseases
Molecular Diagnosis & Therapy (2019)
-
Emerging ways to treat breast cancer: will promises be met?
Cellular Oncology (2018)