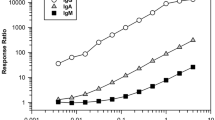
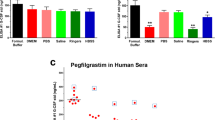
Abstract
Several regulatory challenges are encountered when proceeding towards human clinical trials with gene therapy products, one of these relating to the validity of the analytical methods. Many of the analytical methods used in preclinical studies and/or for analysing the gene therapy products are not yet recognized as a standardized pharmacopoeial methods. Thus, their validity should be demonstrated for the regulatory authorities by thorough assay validations, the extent being dependent on the product developmental stage. We hereby provide an overview on the critical parameters that have to be taken into consideration during the optimization and validation of bio-analytical methods such as PCR, enzyme-linked immunospot assay, Microarray and Limulus Amebocyte Lysate analyses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
The United States Pharmacopeial Convention Inc. Cell and Gene Therapy Products. US Pharmacopeia: Rockville, 2004, pp 2550–2578.
The European Agency for the Evaluation of Medicinal Products, Human Medicines Evaluation Unit. Note for Guidance on Validation of Analytical Methods: Definitions and Terminology. London, 1994, CPMP/ICH/381/95.
The European Agency for the Evaluation of Medicinal Products, Human Medicines Evaluation Unit. Note for Guidance on Validation of Analytical Procedures: Methodology. London, 1996, CPMP/ICH/281/95.
Food and Drug Administration, Center for Drug Evaluation and Research (CDER). Center for Veterinary Medicine (CVM). Guidance for Industry: Bioanalytical Method Validation. US Department of Health and Human Services: Rockville, 2001.
The European Agency for the Evaluation of Medicinal Products, Human Medicines Evaluation Unit. Note for Guidance on Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products. London, 1999, CPMP/ICH/365/96.
The fitness for purpose of analytical methods. Eurachem Guidance Document No. 1/WELAC Guidance Document No. WGD 2: guidance on the interpretation of the EN 45000 series of standards and ISO/IEC Guide 25, 1993.
Mullis KB, Faloona FA . Specific synthesis of DNA in vitro via polymerase-catalyzed chain reaction. Methods Enzymol 1987; 155: 335–350.
Saiki KR et al. Primer-directed enzymatic amplification of DNA with thermostabile DNA polymerase. Science 1988; 239: 487–491.
Neumaier M, Braun A, Wagener C . Fundamentals of quality assessment of molecular amplification methods in clinical diagnostics. Clin Chem 1998; 44: 12–26.
Sedgwick JD, Holt PG . A solid-phase immunoenzymatic technique for the enumeration of specific antibody-secreting cells. J Immunol Methods 1983; 57: 301–309.
Czerkinsky CC et al. A solid-phase enzyme-linked immunospot (ELISPOT) assay for enumeration of specific antibody-secreting cells. J Immunol Methods 1983; 65: 109–121.
Olive M, Eisenlohr LC, Flomenberg P . Quantitative analysis of adenovirus-specific CD4+ T-cell responses from healthy adults. Viral Immunol 2001; 14: 403–413.
Smith JG et al. Development and validation of a gamma interferon ELISPOT assay for quantitation of cellular immune responses to varicella-zoster virus. Clin Diagn Lab Immunol 2001; 8: 871–879.
Woodberry T et al. Prime boost vaccination strategies: CD8 T cell numbers, protection and Th1 bias. J Immunol 2003; 170: 2599–2604.
Engvall E, Perlmann P . Enzyme-linked immunosorbent assay, Elisa. 3. Quantitation of specific antibodies by enzyme-labeled anti-immunoglobulin in antigen-coated tubes. J Immunol 1972; 109: 129–135.
Alwine JC, Kemp DJ, Stark GR . Method for detection of specific RNAs in agarose gels by transfer to diazobenzyloxymethyl-paper and hybridization with DNA probes. Proc Natl Acad Sci USA 1977; 74: 5350–5354.
Towbin H, Staehelin T, Gordon J . Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 1979; 76: 4350–4354.
Schena M et al. Parallel human genome analysis: microarray-based expression monitoring of 1000 genes. Proc Natl Acad Sci USA 1996; 93: 10614–10619.
Howard KA et al. Formulation of a microparticle carrier for oral polyplex-based DNA vaccines. Biochim Biophys Acta 2004; 1674: 149–157.
Ohlfest JR, Lobitz PD, Perkinson SG, Largaespada DA . Integration and long-term expression in xenografted human glioblastoma call using a plasmid-based trasposon system. Mol Ther 2004; 10: 260–268.
Min KA, Lee SK, Kim CK . Improved gene expression pattern using Epstein–Barr virus (EBV)-based plasmid and cationic emulsion. Biomaterials 2005; 26: 1063–1070.
Barrett JC, Kawasaki ES . Microarrays: the use of oligonucleotide and cDNA for the analysis of gene expression. Drug Discov Today 2003; 8: 134–141.
Brazma A et al. Minimum information about a microarray experiment (MIAME) – toward standards for microarray data. Nat Genet 2001; 29: 365–371.
Council of Europe. Pyrogens. European Pharmacopoeia: Strasbourg, 2004, p 152.
The United States Pharmacopeial Convention Inc. Pyrogen Test. US Pharmacopeia: Rockville, 2004, pp 2289–2290.
The United States Pharmacopeial Convention Inc. Bacterial Endotoxins Test. US Pharmacopeia: Rockville, 2004, pp 2264–2267.
Council of Europe. Bacterial Endotoxins. European Pharmacopoeia: Strasbourg, 2004, pp 161–168.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tuomela, M., Stanescu, I. & Krohn, K. Validation overview of bio-analytical methods. Gene Ther 12 (Suppl 1), S131–S138 (2005). https://doi.org/10.1038/sj.gt.3302627
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302627
Keywords
This article is cited by
-
A non-human primate in vitro functional assay for the early evaluation of TB vaccine candidates
npj Vaccines (2021)
-
An optimized IFN-γ ELISpot assay for the sensitive and standardized monitoring of CMV protein-reactive effector cells of cell-mediated immunity
BMC Immunology (2017)
-
Library of molecular associations: curating the complex molecular basis of liver diseases
BMC Genomics (2010)
-
Results and harmonization guidelines from two large-scale international Elispot proficiency panels conducted by the Cancer Vaccine Consortium (CVC/SVI)
Cancer Immunology, Immunotherapy (2008)
-
Phase I trial of DNA-hsp65 immunotherapy for advanced squamous cell carcinoma of the head and neck
Cancer Gene Therapy (2008)