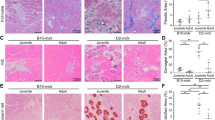
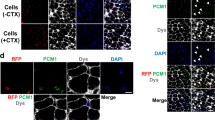
Abstract
Cell transplantation holds promise as a potential treatment for cardiac dysfunction. Our group has isolated populations of murine skeletal muscle-derived stem cells (MDSCs) that exhibit stem cell-like properties. Here, we investigated the fate of MDSCs after transplantation into the hearts of dystrophin-deficient mdx mice, which model Duchenne muscular dystrophy (DMD). Transplanted MDSCs generated large grafts consisting primarily of numerous dystrophin-positive myocytes and, to a lesser degree, dystrophin-negative nonmyocytes that expressed an endothelial phenotype. Most of the dystrophin-positive myocytes expressed a skeletal muscle phenotype and did not express a cardiac phenotype. However, some donor myocytes, located at the graft–host myocardium border, were observed to express cardiac-specific markers. More than half of these donor cells that exhibited a cardiac phenotype still maintained a skeletal muscle phenotype, demonstrating a hybrid state. Sex-mismatched donors and hosts revealed that many donor-derived cells that acquired a cardiac phenotype did so through fusion with host cardiomyocytes. Connexin43 gap junctions were not expressed by donor-derived myocytes in the graft. Scar tissue formation in the border region may inhibit the fusion and gap junction connections between donor and host cells. This study demonstrates that MDSC transplantation warrants further investigation as a potential therapy for cardiac dysfunction in DMD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Hoffman EP, Brown Jr RH, Kunkel LM . Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell 1987; 51: 919–928.
Towbin JA, Bowles NE . The failing heart. Nature 2002; 415: 227–233.
Badorff C et al. Enteroviral protease 2A cleaves dystrophin: evidence of cytoskeletal disruption in an acquired cardiomyopathy. Nat Med 1999; 5: 320–326.
Xiong D et al. Dystrophin deficiency markedly increases enterovirus-induced cardiomyopathy: A genetic predisposition to viral heart disease. Nat Med 2002; 8: 872–877.
Danialou G et al. Dystrophin-deficient cardiomyocytes are abnormally vulnerable to mechanical stress-induced contractile failure and injury. FASEB J 2001; 15: 1655–1657.
Kamogawa Y et al. Dystrophin-deficient myocardium is vulnerable to pressure overload in vivo. Cardiovasc Res 2001; 50: 509–515.
Yue Y et al. Microdystrophin gene therapy of cardiomyopathy restores dystrophin–glycoprotein complex and improves sarcolemma integrity in the mdx mouse heart. Circulation 2003; 108: 1626–1632.
Yue Y et al. Full-length dystrophin expression in half of the heart cells ameliorates beta-isoproterenol-induced cardiomyopathy in mdx mice. Hum Mol Genet 2004; 13: 1669–1675.
Gregorevic P et al. Systemic delivery of genes to striated muscles using adeno-associated viral vectors. Nat Med 2004; 10: 828–834.
Koh GY et al. Stable fetal cardiomyocyte grafts in the hearts of dystrophic mice and dogs. J Clin Invest 1995; 96: 2034–2042.
Sakai T, Ling Y, Payne TR, Huard J . The use of ex vivo gene transfer based on muscle-derived stem cells for cardiovascular medicine. Trends Cardiovasc Med 2002; 12: 115–120.
Hughes S . Cardiac stem cells. J Pathol 2002; 197: 468–478.
Matsuura K et al. Adult cardiac Sca-1 positive cells differentiate into beating cardiomyocytes. J Biol Chem 2004; 279: 11384–11391.
Beltrami AP et al. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 2003; 114: 763–776.
Martin CM et al. Persistent expression of the ATP-binding cassette transporter, Abcg2, identifies cardiac SP cells in the developing and adult heart. Dev Biol 2004; 265: 262–275.
Laugwitz KL et al. Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 2005; 433: 647–653.
Oh H et al. Cardiac progenitor cells from adult myocardium: homing, differentiation, and fusion after infarction. Proc Natl Acad Sci USA 2003; 100: 12313–12318.
Thompson RB et al. Comparison of intracardiac cell transplantation: autologous skeletal myoblasts versus bone marrow cells. Circulation 2003; 108 (Suppl 1): II264–II271.
Dorfman J et al. Myocardial tissue engineering with autologous myoblast implantation. J Thorac Cardiovasc Surg 1998; 116: 744–751.
Yoon PD, Kao RL, Magovern GJ . Myocardial regeneration. Transplanting satellite cells into damaged myocardium. Tex Heart Inst J 1995; 22: 119–125.
Reinecke H, Poppa V, Murry CE . Skeletal muscle stem cells do not transdifferentiate into cardiomyocytes after cardiac grafting. J Mol Cell Cardiol 2002; 34: 241–249.
Koh GY, Klug MG, Soonpaa MH, Field LJ . Differentiation and long-term survival of C2C12 myoblast grafts in heart. J Clin Invest 1993; 92: 1548–1554.
Scorsin M et al. Comparison of the effects of fetal cardiomyocyte and skeletal myoblast transplantation on postinfarction left ventricular function. J Thorac Cardiovasc Surg 2000; 119: 1169–1175.
Dib N et al. Endoventricular transplantation of allogenic skeletal myoblasts in a porcine model of myocardial infarction.J Endovasc Ther 2002; 9: 313–319.
Taylor DA et al. Regenerating functional myocardium: improved performance after skeletal myoblast transplantation. Nat Med 1998; 4: 929–933.
Murry CE, Wiseman RW, Schwartz SM, Hauschka SD . Skeletal myoblast transplantation for repair of myocardial necrosis. J Clin Invest 1996; 98: 2512–2523.
Chiu RC, Zibaitis A, Kao RL . Cellular cardiomyoplasty: myocardial regeneration with satellite cell implantation. Ann Thorac Surg 1995; 60: 12–18.
Atkins BZ et al. Cellular cardiomyoplasty improves diastolic properties of injured heart. J Surg Res 1999; 85: 234–242.
Atkins BZ et al. Myogenic cell transplantation improves in vivo regional performance in infarcted rabbit myocardium. J Heart Lung Transplant 1999; 18: 1173–1180.
Atkins BZ et al. Intracardiac transplantation of skeletal myoblasts yields two populations of striated cells in situ. Ann Thorac Surg 1999; 67: 124–129.
Hagege AA et al. Viability and differentiation of autologous skeletal myoblast grafts in ischaemic cardiomyopathy. Lancet 2003; 361: 491–492.
Herreros J et al. Autologous intramyocardial injection of cultured skeletal muscle-derived stem cells in patients with non-acute myocardial infarction. Eur Heart J 2003; 24: 2012–2020.
Menasche P et al. Myoblast transplantation for heart failure. Lancet 2001; 357: 279–280.
Menasche P et al. Autologous skeletal myoblast transplantation for severe postinfarction left ventricular dysfunction. J Am Coll Cardiol 2003; 41: 1078–1083.
Pagani FD et al. Autologous skeletal myoblasts transplanted to ischemia-damaged myocardium in humans. Histological analysis of cell survival and differentiation. J Am Coll Cardiol 2003; 41: 879–888.
Lee JY et al. Clonal isolation of muscle-derived cells capable of enhancing muscle regeneration and bone healing. J Cell Biol 2000; 150: 1085–1100.
Qu-Petersen Z et al. Identification of a novel population of muscle stem cells in mice: potential for muscle regeneration. J Cell Biol 2002; 157: 851–864.
Cao B et al. Muscle stem cells differentiate into haematopoietic lineages but retain myogenic potential. Nat Cell Biol 2003; 5:640–646.
Hoffman EP, Morgan JE, Watkins SC, Partridge TA . Somatic reversion/suppression of the mouse mdx phenotype in vivo. J Neurol Sci 1990; 99: 9–25.
Wang X et al. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature 2003; 422: 897–901.
Terada N et al. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature 2002; 416: 542–545.
Ying QL, Nichols J, Evans EP, Smith AG . Changing potency by spontaneous fusion. Nature 2002; 416: 545–548.
Alvarez-Dolado M et al. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature 2003; 425: 968–973.
Vassilopoulos G, Wang PR, Russell DW . Transplanted bone marrow regenerates liver by cell fusion. Nature 2003; 422: 901–904.
Nygren JM et al. Bone marrow-derived hematopoietic cells generate cardiomyocytes at a low frequency through cell fusion, but not transdifferentiation. Nat Med 2004; 10: 494–501.
Rubart M, Soonpaa MH, Nakajima H, Field LJ . Spontaneous and evoked intracellular calcium transients in donor-derived myocytes following intracardiac myoblast transplantation. J Clin Invest 2004; 114: 775–783.
Reinecke H, Minami E, Poppa V, Murry CE . Evidence for fusion between cardiac and skeletal muscle cells. Circ Res 2004; 94: e56–e60.
Pavlath GK, Rich K, Webster SG, Blau HM . Localization of muscle gene products in nuclear domains. Nature 1989; 337: 570–573.
Leobon B et al. Myoblasts transplanted into rat infarcted myocardium are functionally isolated from their host. Proc Natl Acad Sci USA 2003; 100: 7808–7811.
Qu Z et al. Development of approaches to improve cell survival in myoblast transfer therapy. J Cell Biol 1998; 142: 1257–1267.
Qu Z, Huard J . Matching host muscle and donor myoblasts for myosin heavy chain improves myoblast transfer therapy. Gene Therapy 2000; 7: 428–437.
van Deutekom JC et al. Implications of maturation for viral gene delivery to skeletal muscle. Neuromuscul Disord 1998; 8: 135–148.
Acknowledgements
This work was supported in part by grants to Dr Johnny Huard from the Muscular Dystrophy Association (USA), the NIH (1U54AR050733-01; R01-HL069368-02), the Pittsburgh Tissue Engineering Initiative (PTEI), the William F and Jean W Donaldson Chair and the Orris C Hirtzel and Beatrice Dewey Hirtzel Memorial Foundation at Children's Hospital of Pittsburgh, and the Henry J Mankin Endowed Chair at the University of Pittsburgh. This work was also supported by a predoctoral fellowship from the American Heart Association (0315349U, TR Payne). We thank Dr Yong Li, Nikhil Kekre, Michael Thompson, William H Foster, Marcelle Pellerin, Jing Zhou and Dr Zhuqing Qu for their technical assistance, Ryan Sauder for his excellent editorial assistance with the manuscript, Dr Ronald J Jankowski for his participation in helpful discussions, and Dr Bruno Péault for his critical comments on the manuscript. We would also like to thank Dr Terry Partridge for his gracious gift of the dystrophin antibody, Dr Roscoe Stanyon for his gracious gift of mouse chromosomal probes and primary PCR DNA products and for answering many questions pertaining to FISH, and Dr Paul Robbins for the nLacZ retrovirus.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Payne, T., Oshima, H., Sakai, T. et al. Regeneration of dystrophin-expressing myocytes in the mdx heart by skeletal muscle stem cells. Gene Ther 12, 1264–1274 (2005). https://doi.org/10.1038/sj.gt.3302521
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302521
Keywords
This article is cited by
-
Intercellular mitochondrial transfer as a means of tissue revitalization
Signal Transduction and Targeted Therapy (2021)
-
NF-κB inhibition reveals a novel role for HGF during skeletal muscle repair
Cell Death & Disease (2015)
-
Muscle-derived stem/progenitor cell dysfunction limits healthspan and lifespan in a murine progeria model
Nature Communications (2012)
-
Human Skeletal Muscle Cells With a Slow Adhesion Rate After Isolation and an Enhanced Stress Resistance Improve Function of Ischemic Hearts
Molecular Therapy (2012)
-
Regeneration of skeletal muscle
Cell and Tissue Research (2012)