Abstract
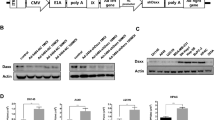
Successful cancer therapy using replicating viral vectors relies on the spread of virus from infected to uninfected cells. To date, there has been limited clinical success in the use of replicating adenoviruses. In animal models, established xenograft tumors are rarely eliminated despite the persistence of high viral titers within the tumor. Hypoxia is a prevalent characteristic of solid tumors, whereas adenovirus naturally infects tissues exposed to ambient oxygen concentrations. Here, we report that hypoxia (1% oxygen) reduces adenoviral replication in H1299 and A549 lung cancer cells, BxPC-3 pancreatic cancer cells, LNCaP prostate cancer cells and HCT116 colon cancer cells. However, hypoxia does not reduce cell viability or restrict S-phase entry. Importantly, the production of E1a and fiber proteins under hypoxic conditions was substantially decreased at 24 and 48 h compared to room air controls. In contrast, Northern analysis showed similar levels of E1a mRNA in room air and hypoxic conditions. In conclusion, a level of hypoxia similar to that found within solid tumors reduces the replication of adenoviral vectors by reduction of viral protein expression without a reduction in mRNA levels. To further improve oncolytic therapy using a replicating adenovirus, it is important to understand the mechanism through which hypoxia and the virus interact to control expression of viral and cellular proteins, and consequently to develop means to overcome decreased viral production in hypoxic conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Heise C, Kirn DH . Replication-selective adenoviruses as oncolytic agents. J Clin Invest 2000; 105: 847–851.
Hawkins LK, Lemoine NR, Kirn D . Oncolytic biotherapy: a novel therapeutic platform. Lancet Oncol 2002; 3: 17–26.
Assessment of adenoviral vector safety and toxicity: report of the National Institutes of Health Recombinant DNA Advisory Committee. Hum Gene Ther 2002; 13: 3–13.
Harrison D et al. Wild-type adenovirus decreases tumor xenograft growth, but despite viral persistence complete tumor responses are rarely achieved – deletion of the viral E1b-19-kD gene increases the viral oncolytic effect. Hum Gene Ther 2001; 12: 1323–1332.
Doronin K et al. Tumor-specific, replication-competent adenovirus vectors overexpressing the adenovirus death protein. J Virol 2000; 74: 6147–6155.
Sauthoff H et al. Intratumoral spread of wild-type adenovirus is limited after local injection of human xenograft tumors: virus persists and spreads systemically at late time points. Hum Gene Ther 2003; 14: 425–433.
Roelvink PW et al. The coxsackievirus-adenovirus receptor protein can function as a cellular attachment protein for adenovirus serotypes from subgroups A, C, D, E, and F. J Virol 1998; 72: 7909–7915.
Kim M et al. The therapeutic efficacy of adenoviral vectors for cancer gene therapy is limited by a low level of primary adenovirus receptors on tumour cells. Eur J Cancer 2002; 38: 1917–1926.
Rauen KA et al. Expression of the coxsackie adenovirus receptor in normal prostate and in primary and metastatic prostate carcinoma: potential relevance to gene therapy. Cancer Res 2002; 62: 3812–3818.
Hockel M, Vaupel P . Tumor hypoxia: definitions and current clinical, biologic, and molecular aspects. J Natl Cancer Inst 2001; 93: 266–276.
Dunn JF et al. Can NMR diffusion-weighted imaging provide quantitative information on tumor interstital pO2? Adv Exp Med Biol 1997; 411: 209–214.
Brown JM, Wilson WR . Exploiting tumour hypoxia in cancer treatment. Nat Rev Cancer 2004; 4: 437–447.
Wouters BG et al. Hypoxia as a target for combined modality treatments. Eur J Cancer 2002; 38: 240–257.
Metzen E, Ratcliffe PJ . HIF hydroxylation and cellular oxygen sensing. Biol Chem 2004; 385: 223–230.
Horwitz MS . Adenoviridae and their replication. In: Fields BN, Knipe DM (eds). Virology. Lippincott Williams & Wilkins: New York, 2001, pp 2301–2326.
Goda N et al. Hypoxia-inducible factor 1alpha is essential for cell cycle arrest during hypoxia. Mol Cell Biol 2003; 23: 359–369.
Gardner LB et al. Hypoxia inhibits G1/S transition through regulation of p27 expression. J Biol Chem 2001; 276: 7919–7926.
Gardner LB, Li F, Yang X, Dang CV . Anoxic fibroblasts activate a replication checkpoint that is bypassed by E1a. Mol Cell Biol 2003; 23: 9032–9045.
Goodrum FD, Ornelles DA . Roles for the E4 orf6, orf3, and E1B 55-kiloDalton proteins in cell cycle-independent adenovirus replication. J Virol 1999; 73: 7474–7488.
Goodrum FD, Ornelles DA . The early region 1B 55-kiloDalton oncoprotein of adenovirus relieves growth restrictions imposed on viral replication by the cell cycle. J Virol 1997; 71: 548–561.
Kraggerud SM, Sandvik JA, Pettersen EO . Regulation of protein synthesis in human cells exposed to extreme hypoxia. Anticancer Res 1995; 15: 683–686.
Koumenis C et al. Regulation of protein synthesis by hypoxia via activation of the endoplasmic reticulum kinase PERK and phosphorylation of the translation initiation factor eIF2alpha. Mol Cell Biol 2002; 22: 7405–7416.
Arsham AM, Howell JJ, Simon MC . A novel hypoxia-inducible factor-independent hypoxic response regulating mammalian target of rapamycin and its targets. J Biol Chem 2003; 278: 29655–29660.
Kirn D . Replication-selective oncolytic adenoviruses: virotherapy aimed at genetic targets in cancer. Oncogene 2000; 19: 6660–6669.
Curiel DT, Gerritsen WR, Krul MR . Progress in cancer gene therapy. Cancer Gene Ther 2000; 7: 1197–1199.
Howe JA et al. Retinoblastoma growth suppressor and a 300-kDa protein appear to regulate cellular DNA synthesis. Proc Natl Acad Sci USA 1990; 87: 5883–5887.
Schneider RJ, Mohr I . Translation initiation and viral tricks. Trends Biochem Sci 2003; 28: 130–136.
Cuesta R, Xi Q, Schneider RJ . Structural basis for competitive inhibition of eIF4G-Mnk1 interaction by the adenovirus 100-kiloDalton protein. J Virol 2004; 78: 7707–7716.
Bett AJ, Krougliak V, Graham FL . DNA sequence of the deletion/insertion in early region 3 of Ad5 dl309. Virus Res 1995; 39: 75–82.
Graham FL, Prevec L . Methods for construction of adenovirus vectors. Mol Biotechnol 1995; 3: 207–220.
Mittereder N, March KL, Trapnell BC . Evaluation of the concentration and bioactivity of adenovirus vectors for gene therapy. J Virol 1996; 70: 7498–7509.
Sauthoff H, Heitner S, Rom WN, Hay JG . Deletion of the adenoviral E1b-19 kD gene enhances tumor cell killing of a replicating adenoviral vector. Hum Gene Ther 2000; 11: 379–388.
Acknowledgements
This work was supported by NIH-NCI R01CA89086, NIH-NCI R01CA102053, NIH-NCRR grant MO1RR-00096, Stony Wold-Herbert Fund and Goldring Clinical Scholars Fund.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pipiya, T., Sauthoff, H., Huang, Y. et al. Hypoxia reduces adenoviral replication in cancer cells by downregulation of viral protein expression. Gene Ther 12, 911–917 (2005). https://doi.org/10.1038/sj.gt.3302459
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302459
Keywords
This article is cited by
-
Oxygen: viral friend or foe?
Virology Journal (2020)
-
Oncolytic viruses for cancer immunotherapy
Journal of Hematology & Oncology (2020)
-
Hepatocellular carcinoma-targeting oncolytic adenovirus overcomes hypoxic tumor microenvironment and effectively disperses through both central and peripheral tumor regions
Scientific Reports (2018)
-
A hypoxia- and telomerase-responsive oncolytic adenovirus expressing secretable trimeric TRAIL triggers tumour-specific apoptosis and promotes viral dispersion in TRAIL-resistant glioblastoma
Scientific Reports (2018)
-
Prospects for combined use of oncolytic viruses and CAR T-cells
Journal for ImmunoTherapy of Cancer (2017)