Abstract
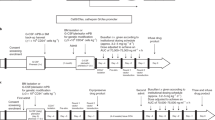
Hematopoietic stem cell gene therapy has not provided clinical success in disorders such as chronic granulomatous disease (CGD), where genetically corrected cells do not show a selective advantage in vivo. To facilitate selective expansion of transduced cells, we have developed a fusion receptor system that confers drug-induced proliferation. Here, a ‘selective amplifier gene (SAG)’ encodes a chimeric receptor (GcRER) that generates a mitotic signal in response to estrogen. We evaluated the in vivo efficacy of SAG-mediated cell expansion in a mouse disease model of X-linked CGD (X-CGD) that is deficient in the NADPH oxidase gp91phox subunit. Bone marrow cells from X-CGD mice were transduced with a bicistronic retrovirus encoding GcRER and gp91phox, and transplanted to lethally irradiated X-CGD recipients. Estrogen was administered to a cohort of the transplants, and neutrophil superoxide production was monitored. A significant increase in oxidase-positive cells was observed in the estrogen-treated mice, and repeated estrogen administration maintained the elevation of transduced cells for 20 weeks. In addition, oxidase-positive neutrophils were increased in the X-CGD transplants given the first estrogen even at 9 months post-transplantation. These results showed that the SAG system would enhance the therapeutic effects by boosting genetically modified, functionally corrected cells in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Curnutte JT, Dinauer MC . Genetic disorders of phagocyte killing. In: Stamatoyannopoulos G, Majerus PW, Perlmutter RM, Varmus H (eds). The Molecular Basis of Blood Diseases. W.B. Saunders: Philadelphia, 2001, pp 539–563.
Winkelstein JA et al. Chronic granulomatous disease: report on a national registry of 368 patients. Medicine 2000; 79: 155–169.
Weening RS, Kabel P, Pijman P, Roos D . Continuous therapy with sulfamethoxazole-trimethoprim in patients with chronic granulomatous disease. J Pediatr 1983; 103: 127–130.
The International Chronic Granulomatous Disease Cooperative Study Group. A controlled trial of interferon gamma to prevent infection in chronic granulomatous disease. N Engl J Med 1991; 324: 509–516.
Seger RA et al. Treatment of chronic granulomatous disease with myeloablative conditioning and an unmodified hemopoietic allograft: a survey of the European experience, 1985–2000. Blood 2002; 100: 4344–4350.
Horwitz ME et al. Treatment of chronic granulomatous disease with nonmyeloablative conditioning and a T-cell-depleted hematopoietic allograft. N Engl J Med 2001; 344: 881–888.
Kume A, Dinauer MC . Gene therapy for chronic granulomatous disease. J Lab Clin Med 2000; 135: 122–128.
Kamani N et al. Marrow transplantation in chronic granulomatous disease: an update, with 6-year follow-up. J Pediatr 1988; 113: 697–700.
Woodman RC et al. A new X-linked variant of chronic granulomatous disease characterized by the existence of a normal clone of respiratory burst-competent phagocytic cells. Blood 1995; 85: 231–241.
Buescher ES, Alling DW, Gallin JI . Use of an X-linked human neutrophil marker to estimate timing of lyonization and size of the dividing stem cell pool. J Clin Invest 1985; 76: 1581–1584.
Pollock JD et al. Mouse model of X-linked chronic granulomatous disease, an inherited defect in phagocyte superoxide production. Nat Genet 1995; 9: 202–209.
Jackson SH, Gallin JI, Holland SM . The p47phox mouse knock-out model of chronic granulomatous disease. J Exp Med 1995; 182: 751–758.
Björgvinsdóttir H et al. Retroviral-mediated gene transfer of gp91phox into bone marrow cells rescues defect in host defense against Aspergillus fumigatus in murine X-linked chronic granulomatous disease. Blood 1997; 89: 41–48.
Mardiney III M et al. Enhanced host defense after gene transfer in the murine p47phox-deficient model of chronic granulomatous disease. Blood 1997; 89: 2268–2275.
Malech HL et al. Prolonged production of NADPH oxidase-corrected granulocytes after gene therapy of chronic granulomatous disease. Proc Natl Acad Sci USA 1997; 94: 12133–12138.
Emery DW et al. Hematopoietic stem cell gene therapy. Int J Hematol 2002; 75: 228–236.
Moscow JA et al. Engraftment of MDR1 and NeoR gene-transduced hematopoietic cells after breast cancer chemotherapy. Blood 1999; 94: 52–61.
Abonour R et al. Efficient retrovirus-mediated transfer of the multidrug resistance 1 gene into autologous human long-term repopulating hematopoietic stem cells. Nat Med 2000; 6: 652–658.
Neff T, Blau CA . Pharmacologically regulated cell therapy. Blood 2001; 97: 2535–2540.
Kume A et al. Selective expansion of transduced cells for hematopoietic stem cell gene therapy. Int J Hematol 2002; 76: 299–304.
Ito K et al. Development of a novel selective amplifier gene for controllable expansion of transduced hematopoietic cells. Blood 1997; 90: 3884–3892.
Matsuda KM et al. Development of a modified selective amplifier gene for hematopoietic stem cell gene therapy. Gene Therapy 1999; 6: 1038–1044.
Xu R et al. A selective amplifier gene for tamoxifen-inducible expansion of hematopoietic cells. J Gene Med 1999; 1: 236–244.
Hanazono Y et al. In vivo selective expansion of gene-modified hematopoietic cells in a nonhuman primate model. Gene Therapy 2002; 9: 1055–1064.
Kume A et al. In vivo expansion of transduced murine hematopoietic cells with a selective amplifier gene. J Gene Med 2003; 5: 175–181.
Hawley RG, Lieu FHL, Fong AZC, Hawley TS . Versatile retroviral vectors for potential use in gene therapy. Gene Therapy 1994; 1: 136–138.
Dranoff G et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci USA 1993; 90: 3539–3543.
Duke GM, Hoffman MA, Palmenberg AC . Sequence and structural elements that contribute to efficient encephalomyocarditis virus RNA translation. J Virol 1992; 66: 1602–1609.
Pear WS, Nolan GP, Scott ML, Baltimore D . Production of high-titer helper-free retroviruses by transient transfection. Proc Natl Acad Sci USA 1993; 90: 8392–8396.
Onodera M et al. A simple and reliable method for screening retroviral producer clones without selectable markers. Hum Gene Ther 1997; 8: 1189–1194.
Zhen L et al. Gene targeting of X chromosome-linked chronic granulomatous disease locus in a human myeloid leukemia cell line and rescue by expression of recombinant gp91phox. Proc Natl Acad Sci USA 1993; 90: 9832–9836.
Nakamura M et al. Monoclonal antibody 7D5 raised to cytochrome b558 of human neutrophils: immunocytochemical detection of the antigen in peripheral phagocytes of normal subjects, patients with chronic granulomatous disease, and their carrier mothers. Blood 1987; 69: 1404–1408.
Hanenberg H et al. Colocalization of retrovirus and target cells on specific fibronectin fragments increases genetic transduction of mammalian cells. Nat Med 1996; 2: 876–882.
Vowells SJ et al. Flow cytometric analysis of the granulocyte respiratory burst: a comparison study of fluorescent probes. J Immunol Methods 1995; 178: 89–97.
Cavazzana-Calvo M et al. Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science 2000; 288: 669–672.
Hacein-Bey-Abina S et al. Sustained correction of X-linked severe combined immunodeficiency by ex vivo gene therapy. N Engl J Med 2002; 346: 1185–1193.
Kume A et al. Selective growth advantage of wild-type lymphocytes in X-linked SCID recipients. Bone Marrow Transplant 2002; 30: 113–118.
Hacein-Bey-Abina S et al. A serious adverse event after successful gene therapy for X-linked severe combined immunodeficiency. N Engl J Med 2003; 348: 255–256.
Kohn DB, Sadelain M, Glorioso JC . Occurrence of leukaemia following gene therapy of X-linked SCID. Nat Rev Cancer 2003; 3: 477–488.
Jin L et al. In vivo selection using a cell-growth switch. Nat Genet 2000; 26: 64–66.
Neff T et al. Pharmacologically regulated in vivo selection in a large animal. Blood 2002; 100: 2026–2031.
Kume A et al. Lymphoid reconstitution in X-linked severe combined immunodeficient mice by retrovirus-mediated gene transfer. Proc Jpn Acad 2002; 78: 211–216.
Kume A et al. Long-term tracking of murine hematopoietic cells transduced with a bicistronic retrovirus containing CD24 and EGFP genes. Gene Therapy 2000; 7: 1193–1199.
Yasueda H et al. High-level direct expression of semi-synthetic human interleukin-6 in Escherichia coli and production of N-terminus Met-free product. Bio/Technology 1990; 8: 1036–1040.
Sekhsaria S et al. Peripheral blood progenitors as a target for genetic correction of p47phox-deficient chronic granulomatous disease. Proc Natl Acad Sci USA 1993; 90: 7446–7450.
Acknowledgements
We thank Dr MC Dinauer for X-CGD mice, and Dr M Nakamura for 7D5 monoclonal antibody. We are also grateful to Amgen for SCF, Ajinomoto for IL-6, Chugai Pharmaceuticals for Epo and G-CSF, and Takara Bio for RetroNectin. This work was supported in part by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan, and the Ministry of Health, Labor and Welfare of Japan.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hara, T., Kume, A., Hanazono, Y. et al. Expansion of genetically corrected neutrophils in chronic granulomatous disease mice by cotransferring a therapeutic gene and a selective amplifier gene. Gene Ther 11, 1370–1377 (2004). https://doi.org/10.1038/sj.gt.3302317
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302317