Abstract
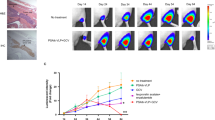
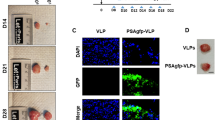
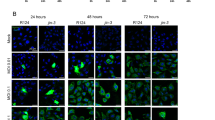
Gene-directed enzyme prodrug therapy (GDEPT) based on the Escherichia coli enzyme, purine nucleoside phosphorylase (PNP), provides a novel strategy for treating slowly growing tumors like prostate cancer (CaP). PNP converts systemically administered prodrug, fludarabine phosphate, to a toxic metabolite, 2-fluoroadenine, that kills PNP-expressing and nearby cells by inhibiting DNA, RNA and protein synthesis. Reporter gene expression directed by a hybrid prostate-directed promoter and enhancer, PSMEPb, was assayed after plasmid transfection or viral transduction of prostate and non-CaP cell lines. Androgen-sensitive (AS) LNCaP-LN3 and androgen-independent (AI) PC3 human CaP xenografts in nude mice were injected intratumorally with an ovine atadenovirus vector, OAdV623, that carries the PNP gene under PSMEPb, formulated with cationic lipid for enhanced infectivity. Fludarabine phosphate was then given intraperitoneally for 5 days at 75 mg/m2/day. PNP expression was evaluated by enzymic conversion of its substrate using reverse phase HPLC. OAdV623 showed excellent in vitro transcriptional specificity for CaP cells. In vivo, expression of PNP persisted for >6 days after OAdV623 injection and a single treatment provided 100% increase in tumor doubling time and >50% inhibition of tumor growth for both LNCaP-LN3 and PC3 lines, with increased tumor necrosis and apoptosis and decreased tumor cell proliferation. OAdV623 significantly suppressed the growth of AS and AI human CaP xenografts in mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Landis SH, Murray T, Bolden S, Wingo PA . Cancer statistics, 1998. CA Cancer. J Clin 1998; 48: 6–29.
Denis L, Murphy GP . Overview of phase III trials on combined androgen treatment in patients with metastatic prostate cancer. Cancer (Phila.) 1993; 72: 3888–3895.
Eastham JA et al. Prostate cancer gene therapy: herpes simplex virus thymidine kinase gene transduction followed by ganciclovir in mouse and human prostate cancer models. Hum Gene Ther 1996; 7: 515–523.
Martiniello R et al. In vivo gene therapy for prostate cancer: preclinical evaluation of two different enzyme-prodrug systems delivered by identical adenovirus vectors. Hum Gene Ther 1998; 9: 1617–1626.
Hermann JR et al. In situ gene therapy for adenocarcinoma of the prostate: a phase I clinical trial. Hum Gene Ther 1999; 10: 1239–1249.
Freytag SO et al. Phase I study of replication-competent adenovirus-mediated double suicide gene therapy for the treatment of locally recurrent prostate cancer. Cancer Res 2002; 62: 4968–4976.
Watt F et al. A tissue-specific enhancer of the prostate-specific membrane antigen gene, FOLH1. Genomics 2001; 73: 243–254.
Hofmann C et al. Ovine adenovirus vectors overcome preexisting humoral immunity against human adenoviruses in vivo. J Virol 1999; 73: 6930–6936.
Khatri A, Xu ZZ, Both GW . Gene expression by atypical recombinant ovine adenovirus vectors during abortive infection of human and animal cells in vitro. Virology 1997; 239: 226–237.
Xu ZZ et al. An ovine adenovirus vector lacks transforming ability in cells that are transformed by AD5 E1A/B sequences. Virology 2000; 270: 162–172.
Lockett LJ, Both GW . Complementation of a defective human adenovirus by an otherwise incompatible ovine adenovirus recombinant carrying a functional E1A gene. Virology 2002; 294: 333–341.
Both G . Xenogenic adenoviruses. In: Curiel D, Douglas J (eds). Adenoviral Vectors for Gene Therapy. Academic Press: San Diego, 2002, pp 447–479.
Voeks D et al. Transduction of biopsy samples: a way to bridge gene therapy between animals and humans. Biotechniques 2001; 31: 46–49.
Parker WB et al. Metabolism and metabolic actions of 6-methylpurine and 2-fluoroadenine in human cells. Biochem Pharmacol 1998; 55: 1673–1681.
Berges RR et al. Implication of cell kinetic changes during progression of human prostatic cancer. Clin Cancer Res 1995; 1: 473–480.
Sorscher EJ et al. Tumor cell bystander killing in colonic carcinoma utilizing the Escherichia coli DeoD gene to generate toxic purines. Gene Therapy 1994; 1: 233–238.
Voeks D et al. Gene therapy for prostate cancer delivered by ovine adenovirus and mediated by purine nucleoside phosphorylase and fludarabine in mouse models. Gene Therapy 2002; 9: 759–768.
Gavrieli Y, Sherman Y, Ben Sasson SA . Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 1992; 119: 493–501.
Gerdes J et al. Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J Immunol 1984; 133: 1710–1715.
Martiniello-Wilks R et al. Transcription-targeted gene therapy for androgen-independent prostate cancer. Cancer Gene Ther 2002; 9: 443–452.
Parker WB et al. In vivo gene therapy of cancer with E. coli purine nucleoside phosphorylase. Hum Gene Ther 1997; 8: 1637–1644.
Lockett LJ, Molloy PL, Russell PJ, Both GW . Relative efficiency of tumor cell killing in vitro by two enzyme-prodrug systems delivered by identical adenovirus vectors. Clin Cancer Res 1997; 3: 2075–2080.
Wu L et al. Chimeric PSA enhancers exhibit augmented activity in prostate cancer gene therapy vectors. Gene Therapy 2000; 8: 1416–1426.
Shirakawa T et al. Tissue-specific promoters in gene therapy for the treatment of prostate cancer. Mol Urol 2000; 4: 73–82.
Pramudji C et al. In situ prostate cancer gene therapy using a novel adenoviral vector regulated by the caveolin-1 promoter. Clin Cancer Res 2001; 7: 4272–4279.
Fasbender A et al. Complexes of adenovirus with polycationic polymers and cationic lipids increase the efficiency of gene transfer in vitro and in vivo. J Biol Chem 1997; 272: 6479–6489.
Snyder FF, Lukey T . Kinetic considerations for the regulation of adenosine and deoxyadenosine metabolism in mouse and human tissues based on a thymocyte model. Biochem Biophys Acta 1982; 696: 299–307.
Carson DA, Kaye J, Wasson DB . Differences in deoxyadenosine metabolism in human and mouse lymphocytes. J Immunol 1980; 124: 8–12.
Bramson JL, Hitt M, Gauldie J, Graham FI . Pre-existing immunity to adenovirus does not prevent tumor regression following intratumoral admiistration of a vector expressing IL-12 but inhibits virus dissemination. Gene Therapy 1997; 4: 1069–1076.
Benihoud K, Yeh P, Perricaudet M . Adenovirus vectors for gene delivery. Curr Opin Biotechnol 1999; 10: 440–447.
Eastham JA, Grafton W, Martin CM, Williams BJ . Suppression of primary tumor growth and the progression to metastasis with p53 adenovirus in human prostate cancer. J Urol 2000; 164: 814–819.
Friedland D et al. A phase II trial of docetaxel (Taxotere) in hormone-refractory prostate cancer: correlation of antintratumoralor effect to phosphorylation of Bcl-2. Semin Oncol 1999; 26: 19–23.
Krohne TU et al. Mechanisms of cell death induced by suicide genes encoding purine nucleoside phosphorylase and thymidine kinase in human hepatocellular carcinoma cells in vitro. Hepatology 2001; 34: 11–518.
Carroll AG, Voeller HJ, Sugars L, Gelmann EP . p53 oncogene mutations in three human prostate cancer cell lines. Prostate 1993; 23: 123–134.
Downing SR, Jackson P, Russell PJ . Mutations within the tumor suppressor gene p53 are not confined to a late event in prostate cancer progression: a review of the evidence. Urol Oncol 2001; 6: 103–110.
Pettaway CA et al. Selection of highly metastatic variants of different human prostate carcinomas using orthotopic implantation in nude mice. Clin Cancer Res 1996; 2: 1627–1636.
Löser P et al. Construction, rescue, and characterization of vectors derived from ovine atadenovirus. J Virol 2003; 77: 11941–11951.
Vrati S et al. Construction and transfection of ovine adenovirus genomic clones to rescue modified viruses. Virology 1996; 220: 200–203.
Xu ZZ, Hyatt A, Boyle DB, Both GW . Construction of ovine adenovirus recombinants by gene insertion or deletion of related terminal region sequences. Virology 1997; 230: 62–71.
Boyle DB et al. Characterisation of Australian ovine adenovirus isolates. Vet Microbiol 1994; 41: 281–291.
Cameron FH et al. A transfection compound series based on a versatile Tris linkage. Biochim Biophys Acta 1999; 1417: 37–50.
Russell PJ et al. Bladder cancer xenografts: a model of tumor cell heterogeneity. Cancer Res 1986; 46: 2035–2040.
Acknowledgements
We thank Mayne Pharma Pty Ltd for funding the project, Kylie Smith and Emma Bennetts for technical assistance in the early phase of the project and Mila Sajinovic and Hnin Pwint for excellent animal husbandry.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wang, X., Martiniello-Wilks, R., Shaw, J. et al. Preclinical evaluation of a prostate-targeted gene-directed enzyme prodrug therapy delivered by ovine atadenovirus. Gene Ther 11, 1559–1567 (2004). https://doi.org/10.1038/sj.gt.3302308
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302308
Keywords
This article is cited by
-
Armed and targeted measles virus for chemovirotherapy of pancreatic cancer
Cancer Gene Therapy (2011)
-
Enhanced efficiency of prodrug activation therapy by tumor-selective replicating retrovirus vectors armed with the Escherichia coli purine nucleoside phosphorylase gene
Cancer Gene Therapy (2010)
-
Development of gene therapy in association with clinically used cytotoxic deoxynucleoside analogues
Cancer Gene Therapy (2009)