Abstract
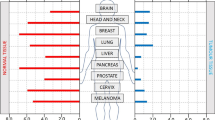
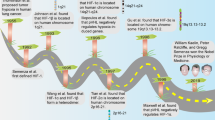
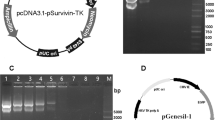
Hypoxia is a common physiological feature of tumours. It activates a signalling cascade that culminates in the stabilization of the HIF-1 transcription factor and activation of genes that possess a hypoxia response element (HRE). We have used an optimized hypoxia responsive promoter (OBHRE) to investigate hypoxia-targeted gene expression in vivo in the context of an adenovirus vector. The OBHRE promoter showed limited activity in the liver or spleen such that expression was 1000-fold lower than that driven by the strong CMV/IE promoter. However, in the context of the tumour microenvironment, the OBHRE promoter achieved expression levels comparable to that of the CMV/IE promoter. Next, we showed that an adenovirus expressing the human cytochrome P450 (CYP2B6) regulated by the OBHRE promoter delays tumour growth in response to the prodrug cyclophosphamide (CPA). Finally, we exploited the hepatotropism of adenovirus to investigate whether the OBHRE promoter could mitigate the hepatotoxicity of a recombinant adenovirus expressing thymidine kinase (TK) in the context of the prodrug ganciclovir (GCV). High-dose Ad.CMVTK/GCV treatment caused significant liver necrosis whereas the same dose of Ad.HRETK was well tolerated. These in vivo data demonstrate that hypoxia-targeted gene expression via the OBHRE promoter can be used to increase the therapeutic window of cytotoxic cancer gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Shirakawa T et al. Tissue-specific promoters in gene therapy for the treatment of prostate cancer. Mol Urol 2000; 4: 73–82.
He P et al. The targeted expression of interleukin-2 in human hepatocellular carcinoma cells. J Exp Clin Cancer Res 2000; 19: 183–187.
Vaupel PW . Oxygenation of solid tumours In: Teicher BA (ed). Drug Resistance in Oncology. Marcel Dekker: New York, 1993, pp 53–85.
Semenza GL . HIF-1 and human disease: one highly involved factor. Genes Dev 2000; 14: 1983–1991.
Maxwell PH, Pugh CW, Ratcliffe PJ . Inducible operation of the erythropoietin 3′ enhancer in multiple cell lines: evidence for a widespread oxygen-sensing mechanism. Proc Natl Acad Sci USA 1993; 90: 2423–2427.
Wang GL, Semenza GL . General involvement of hypoxia-inducible factor 1 in transcriptional response to hypoxia. Proc Natl Acad Sci USA 1993; 90: 4304–4308.
Talks KL et al. The expression and distribution of the hypoxia-inducible factors HIF-1alpha and HIF-2alpha in normal human tissues, cancers, and tumor-associated macrophages. Am J Pathol 2000; 157: 411–421.
Zhong H et al. Overexpression of hypoxia-inducible factor 1alpha in common human cancers and their metastases. Cancer Res 1999; 59: 5830–5835.
Semenza GL . Regulation of mammalian O2 homeostasis by hypoxia-inducible factor 1. Annu Rev Cell Dev Biol 1999; 15: 551–578.
Aebersold DM et al. Expression of hypoxia-inducible factor-1alpha: a novel predictive and prognostic parameter in the radiotherapy of oropharyngeal cancer. Cancer Res 2001; 61: 2911–2916.
Birner P et al. Expression of hypoxia-inducible factor 1alpha in epithelial ovarian tumors: its impact on prognosis and on response to chemotherapy. Clin Cancer Res 2001; 7: 1661–1668.
Birner P et al. Expression of hypoxia-inducible factor-1 alpha in oligodendrogliomas: its impact on prognosis and on neoangiogenesis. Cancer 2001; 92: 165–171.
Binley K et al. An adenoviral vector regulated by hypoxia for the treatment of ischaemic disease and cancer. Gene Ther 1999; 6: 1721–1727.
Boast K et al. Characterisation of physiologically regulated vectors for the treatment of ischemic disease. Human Gene Therapy 1999; 10: 21972208.
Griffiths L et al. The macrophage – a novel system to deliver gene therapy to pathological hypoxia. Gene Ther 2000; 7: 255–262.
Greco O, Dachs GU . Gene directed enzyme/prodrug therapy of cancer: historical appraisal and future prospectives. J Cell Physiol 2001; 187: 22–36.
Brand K et al. Liver-associated toxicity of the HSV-tk/GCV approach and adenoviral vectors. Cancer Gene Ther 1997; 4: 9–16.
Franz WM, Rothmann T, Frey N, Katus HA . Analysis of tissue-specific gene delivery by recombinant adenoviruses containing cardiac-specific promoters. Cardiovasc Res 1997; 35: 560–566.
Shi Q, Wang Y, Worton R . Modulation of the specificity and activity of a cellular promoter in an adenoviral vector. Hum Gene Ther 1997; 8: 403–410.
Bilbao R et al. Transduction efficacy, antitumoral effect, and toxicity of adenovirus-mediated herpes simplex virus thymidine kinase/ganciclovir therapy of hepatocellular carcinoma: the woodchuck animal model. Cancer Gene Ther 2000; 7: 657–662.
Shinoura N et al. Adenovirus-mediated transfer of bax with caspase-8 controlled by myelin basic protein promoter exerts an enhanced cytotoxic effect in gliomas. Cancer Gene Ther 2000; 7: 739–748.
Fechner H et al. Expression of coxsackie adenovirus receptor and alphav-integrin does not correlate with adenovector targeting in vivo indicating anatomical vector barriers. Gene Ther 1999; 6: 1520–1535.
Addison CL, Hitt M, Kunsken D, Graham FL . Comparison of the human versus murine cytomegalovirus immediate early gene promoters for transgene expression by adenoviral vectors. J Gen Virol 1997; 78: 1653–1661.
Jiang BH, Agani F, Passaniti A, Semenza GL . V-SRC induces expression of hypoxia-inducible factor 1 (HIF-1) and transcription of genes encoding vascular endothelial growth factor and enolase 1: involvement of HIF-1 in tumor progression. Cancer Res 1997; 57: 5328–5335.
Maxwell PH et al. Hypoxia-inducible factor-1 modulates gene expression in solid tumors and influences both angiogenesis and tumor growth. Proc Natl Acad Sci USA 1997; 94: 8104–8109.
Wiesener MS et al. Constitutive activation of hypoxia-inducible genes related to overexpression of hypoxia-inducible factor-1alpha in clear cell renal carcinomas. Cancer Res 2001; 61: 5215–5222.
Birner P et al. Overexpression of hypoxia-inducible factor 1alpha is a marker for an unfavorable prognosis in early-stage invasive cervical cancer. Cancer Res 2000; 60: 4693–4696.
Kan O et al. Direct retroviral delivery of human cytochrome P450 2B6 for gene-directed enzyme prodrug therapy of cancer. Cancer Gene Ther 2001; 8: 473–482.
Waxman DJ, Chen L, Hecht JE, Jounaidi Y . Cytochrome P450-based cancer gene therapy: recent advances and future prospects. Drug Metab Rev 1999; 31: 503–522.
van der Eb MM et al. Severe hepatic dysfunction after adenovirus-mediated transfer of the herpes simplex virus thymidine kinase gene and ganciclovir administration. Gene Ther 1998; 5: 451–458.
Yamamoto M, Alemany R, Adachi Y, Grizzle WE, Curiel DT . Characterization of the cyclooxygenase-2 promoter in an adenoviral vector and its application for the mitigation of toxicity in suicide gene therapy of gastrointestinal cancers. Mol Ther 2001; 3: 385–394.
Brand K et al. Tumor cell-specific transgene expression prevents liver toxicity of the adeno-HSVtk/GCV approach. Gene Ther 1998; 5: 1363–1371.
Reynolds PN et al. Combined transductional and transcriptional targeting improves the specificity of transgene expression in vivo. Nat Biotechnol 2001; 19: 838–842.
Wickham TJ . Targeting adenovirus. Gene Ther 2000; 7: 110–114.
Kruyt FA, Curiel DT . Toward a new generation of conditionally replicating adenoviruses: pairing tumor selectivity with maximal oncolysis. Hum Gene Ther 2002; 13: 485–495.
Rodriguez R et al. Prostate attenuated replication competent adenovirus (ARCA) CN706: a selective cytotoxic for prostate-specific antigen-positive prostate cancer cells. Cancer Res 1997; 57: 2559–2563.
Alemany R et al. Complementary adenoviral vectors for oncolysis. Cancer Gene Ther 1999; 6: 21–25.
Yamamoto M et al. Characterization of the cyclooxygenase-2 promoter in an adenoviral vector and its application for the mitigation of toxicity in suicide gene therapy of gastrointestinal cancers. Mol Ther 2001; 3: 385–394.
Patterson AV et al. Oxygen-sensitive enzyme-prodrug gene therapy for the eradication of radiation-resistant solid tumours. Gene Ther 2002; 9: 946–954.
Shibata T, Giaccia AJ, Brown JM . Hypoxia-inducible regulation of a prodrug-activating enzyme for tumor-specific gene therapy. Neoplasia 2002; 4: 40–48.
He TC et al. A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 1998; 95: 2509–2514.
Acknowledgements
We thank Michael Dennis (CAMR, Salisbury, Wiltshire, UK) and Pierrick Auvray (Oncodesign, France) for the maintenance and management of the animals, and Theresa Wardell (Oxford BioMedica (UK) Ltd) for advice and guidance with the real-time PCR studies.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Binley, K., Askham, Z., Martin, L. et al. Hypoxia-mediated tumour targeting. Gene Ther 10, 540–549 (2003). https://doi.org/10.1038/sj.gt.3301944
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301944
Keywords
This article is cited by
-
A hypoxia- and telomerase-responsive oncolytic adenovirus expressing secretable trimeric TRAIL triggers tumour-specific apoptosis and promotes viral dispersion in TRAIL-resistant glioblastoma
Scientific Reports (2018)
-
Hypoxia-Sensitive Materials for Biomedical Applications
Annals of Biomedical Engineering (2016)
-
Oncolytic adenovirus armed with IL-24 Inhibits the growth of breast cancer in vitro and in vivo
Journal of Experimental & Clinical Cancer Research (2012)
-
Adenoviral targeting of gene expression to tumors
Cancer Gene Therapy (2010)
-
Lister strain vaccinia virus, a potential therapeutic vector targeting hypoxic tumours
Gene Therapy (2010)