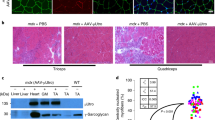
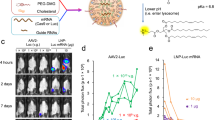
Abstract
Attempts to correct genetic disorders by gene therapy have been hindered by various problems including unwanted immune responses against the gene product. It has been shown that immune responses with DNA vaccines after i.m. injection of antigen-encoding plasmid DNA are primed solely by professional antigen-presenting cells (APC), even though myocytes are the primary type of cell transfected. This possibly involves direct transfection of some APC in regional lymph nodes draining the injected muscle. Here we have used plasmid DNA vaccines that express hepatitis B surface antigen (HBsAg) to evaluate the possibility of abrogating these immune responses by use of a tissue-specific promoter that does not drive expression in APC. We show that HBsAg-specific humoral or cell-mediated responses are not induced in mice when the muscle-specific human muscle creatine kinase promoter is used in place of the ubiquitous cytomegaloviral promoter to drive expression of HBsAg. This may have significance in the field of gene therapy where one aims to achieve stable expression of the desired gene product without interference from the host immune response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ohtsuka Y et al. Dystrophin acts as a transplantation rejection antigen in dystrophin-deficient mice: implication for gene therapy J Immunol 1998 160: 4635–4640
van Ginkel F et al. Adenoviral gene delivery elicits distinct pulmonary-associated T helper cell responses to the vector and to its transgene J Immunol 1997 159: 685–693
Evans G, Morgan R . Genetic induction of immune tolerance to human clotting factor VIII in a mouse model for hemophilia A Proc Natl Acad Sci USA 1998 95: 5734–5739
Kass-Eisler A et al. Circumventing the immune response to adenovirus-mediated gene therapy Gene Therapy 1996 3: 154–162
Wells K et al. Immune responses, not promoter inactivation, are responsible for decreased long-term expression following plasmid gene transfer into skeletal muscle FEBS Lett 1997 407: 164–168
Petrof BJ . Respiratory muscles as a target for adenoviral-mediated gene therapy Eur Respir J 1998 11: 492–497
Corr M, Lee D, Carson D, Tighe H . Gene vaccination with naked plasmid DNA: mechanism of CTL priming J Exp Med 1996 184: 1555–1560
Doe B et al. Induction of cytotoxic T lymphocytes by intramuscular immunization with plasmid DNA is facilitated by bone marrow-derived cells Proc Natl Acad Sci USA 1996 93: 8578–8583
Iwasaki A et al. The dominant role of bone-marrow derived cells in CTL induction following plasmid DNA immunization at different sites J Immunol 1997 159: 11–14
Ulmer JB et al. Generation of MHC class-I restricted T lymphocytes by expression of a viral protein in muscle cells: antigen presentation by non-muscle cells Immunology 1996 89: 59–67
Fu T et al. Priming of cytotoxic T lymphocytes by DNA vaccines: requirement for professional antigen presenting cells and evidence for antigen transfer from myocytes Mol Med 1997 3: 362–371
Davis HL, Michel M-L, Whalen RG . DNA based immunization induces continuous secretion of for hepatitis B surface antigen and high levels of circulating antibody Hum Mol Genet 1993 2: 1847–1851
Davis HL, Mancini M, Michel M-L, Whalen RG . DNA-mediated immunization to hepatitis B surface antigen: longevity of primary response and effect of boost Vaccine 1996 14: 910–915
Davis HL et al. CpG DNA is a potent enhancer of specific immunity in mice immunized with recombinant hepatitis B surface antigen J Immunol 1998 160: 870–876
Davis HL, Brazolot Millan CL, Watkins SC . Immune-mediated destruction of transfected muscle fibers after direct gene transfer with antigen-expressing plasmid DNA Gene Therapy 1997 4: 181–188
Michel ML et al. DNA-mediated immunization to the hepatitis B surface antigen in mice: Aspects of the humoral response mimic hepatitis B viral infection in humans Proc Natl Acad Sci USA 1995 92: 5307–5311
Chattergoon MA, Robinason TM, Boyer JD, Weiner DB . Specific immune induction following DNA-based immunization through in vivo transfection and activation of macrophages/antigen-presenting cells J Immunol 1998 160: 5707–5718
Manickan E et al. Enhancement of immune response to naked DNA vaccine by immunization with transfected dendritic cells J Leuk Biol 1997 61: 125–132
Hohlfeld R, Engel AG . The immunobiology of muscle Immunol Today 1994 15: 269–274
Wolff JA et al. Direct gene transfer into mouse muscle in vivo Science 1990 247: 1465–1468
Wolff JA et al. Long-term persistence of plasmid DNA and foreign gene expression in mouse muscle Hum Mol Genet 1992 1: 363–369
Torres C, Iwasaki A, Barber B, Robinson H . Differential dependence on target site tissue for gene gun and intramuscular DNA immunizations J Immunol 1997 158: 4529–4532
Condon C et al. DNA-based immunization by in vivo transfection of dendritic cells Nat Med 1996 2: 1122–1128
Loirat D et al. Muscle-specific expression of hepatitis B surface antigen: no effect on DNA-raised immune responses Virology 1999 260: 74–83
Loirat D et al. Muscle-specific expression of hepatitis B surface antigen: no effect on DNA-raised immune responses
Krieg AM et al. Sequence motifs in adenoviral DNA block immune activation by stimulatory CpG motifs Proc Natl Acad Sci USA 1998 95: 12631–12636
Davis HL, Whalen RG, Demeneix BA . Direct gene transfer into skeletal muscle in vivo: factors affecting efficiency of transfer and stability of expression Hum Gene Ther 1993 4: 151–159
Brazolot Millan CL et al. CpG DNA can induce strong Th1 humoral and cell-mediated immune responses against hepatitis B surface antigen in young mice Proc Natl Acad Sci USA 1998 95: 15553–15558
McCluskie MJ, Davis HL . CpG DNA is a potent enhancer of systemic and mucosal immune responses against hepatitis B surface antigen with intranasal administration to mice J Immunol 1998 161: 4463–4466
Acknowledgements
We are most grateful to Amanda Boyd, Lorraine Hamblin and Yu Xu for technical assistance. We also thank Dr Yubo Ren for his help with confocal microscopy and Dr Richard Bartlett for providing plasmids (see Materials and methods).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weeratna, R., Wu, T., Efler, S. et al. Designing gene therapy vectors: avoiding immune responses by using tissue-specific promoters. Gene Ther 8, 1872–1878 (2001). https://doi.org/10.1038/sj.gt.3301602
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301602
Keywords
This article is cited by
-
State of play and clinical prospects of antibody gene transfer
Journal of Translational Medicine (2017)
-
Comparative analysis of macrophage associated vectors for use in genetic vaccine
Genetic Vaccines and Therapy (2011)
-
Construction and analysis of compact muscle-specific promoters for AAV vectors
Gene Therapy (2008)
-
Long-term expression of full-length human dystrophin in transgenic mdx mice expressing internally deleted human dystrophins
Gene Therapy (2004)
-
Muscle stem cells can act as antigen-presenting cells: implication for gene therapy
Gene Therapy (2004)